Introduction
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during reactions with other substances. Chemistry also addresses the nature of chemical bonds in chemical compounds.
In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both basic and applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the properties of the soil on the Moon (cosmochemistry), how medications work (pharmacology), and how to collect DNA evidence at a crime scene (forensics).
Chemistry has existed under various names since ancient times. It has evolved, and now chemistry encompasses various areas of specialisation, or subdisciplines, that continue to increase in number and interrelate to create further interdisciplinary fields of study. The applications of various fields of chemistry are used frequently for economic purposes in the chemical industry. (Full article...)
Selected article
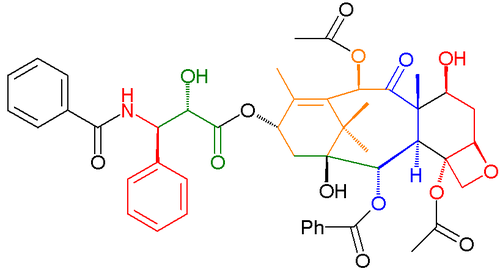
Enzymes are molecules that manipulate other molecules — the enzymes' substrates. These target molecules bind to an enzyme's active site and are transformed into products through a series of steps known as the enzymatic mechanism. Some enzymes bind multiple substrates and/or release multiple products, such as a protease cleaving one protein substrate into two polypeptide products. Others join substrates together, such as DNA polymerase linking a nucleotide to DNA. Although these mechanisms are often a complex series of steps, there is typically one rate-determining step that determines the overall kinetics. This rate-determining step may be a chemical reaction or a conformational change of the enzyme or substrates, such as those involved in the release of product(s) from the enzyme.
Subcategories
- Analytical chemistry: Chromatography, Spectroscopy
- Biochemistry: Molecular biology
- Crystal Chemistry
- Environmental chemistry: Geochemistry
- Inorganic chemistry: Inorganic reactions
- Materials science: Nanotechnology, Glass, Ceramics
- Medicinal chemistry
- Metallurgy
- Nuclear chemistry
- Organic chemistry: Functional groups, Organic compounds, Organic reactions
- Organometallic chemistry
- Pharmacy
- Physical chemistry: Electrochemistry, Quantum chemistry
- Polymer chemistry
- Supramolecular chemistry
- Theoretical chemistry: Computational chemistry
History and Philosophy of Chemistry

Many chemists have an interest in the history of chemistry. Those with philosophical interests will be interested that the philosophy of chemistry has quite recently developed along a path somewhat different from the general philosophy of science.
Other articles that might interest you are:
There is a Wikipedia Project on the History of Science.
Chemistry Resources

Wikipedia:WikiProject Chemicals/Data is a collection of links and references that are useful for chemistry-related works. This includes free online chemical databases, publications, patents, computer programs, and various tools.
unit-conversion.info A good place to figure out what equals what.
General Chemistry Online Clear text and comprehensive coverage of general chemistry topics by Fred Senese, Dept. of Chemistry Frostburg State University
General Chemistry Demonstration at Purdue Video clips (and descriptions) of lecture demonstrations.
Chemistry Webercises Directory A large listing of chemistry resources maintained by Steven Murov, Emeritus Chemistry Professor Modesto Junior College.
MathMol MathMol (Mathematics and Molecules) is a good starting point for those interested in the field of molecular modeling.
ABC-Chemistry A directory of free full-text journals in chemistry, biochemistry and related subjects.
The Element Song A goofy little song about all of the elements.
Selected image
Selected biography
Techniques used by chemists
Equipment used by chemists

Chemistry in society
Chemistry in industry

Companies: AstraZeneca - Bayer - BP - BASF - Bristol-Myers Squibb - DowDuPont - Evonik Industries - ExxonMobil - Linde plc - Mitsubishi - Monsanto - Nestlé - OSI - Shell - Sigma-Aldrich - Sasol - Total - GlaxoSmithKline - Teva
WikiProjects
Topics
Periodic Table
| Group | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hydrogen & alkali metals |
Alkaline earth metals | Triels | Tetrels | Pnictogens | Chalcogens | Halogens | Noble gases | ||||||||||||
| Period |
|||||||||||||||||||
| 2 | |||||||||||||||||||
| 3 | |||||||||||||||||||
| 4 | |||||||||||||||||||
| 5 | |||||||||||||||||||
| 6 | |||||||||||||||||||
| 7 | |||||||||||||||||||
- Ca: 40.078 — Abridged value (uncertainty omitted here)[2]
- Po: [209] — mass number of the most stable isotope
Related portals
Associated Wikimedia
The following Wikimedia Foundation sister projects provide more on this subject:
-
Commons
Free media repository -
Wikibooks
Free textbooks and manuals -
Wikidata
Free knowledge base -
Wikinews
Free-content news -
Wikiquote
Collection of quotations -
Wikisource
Free-content library -
Wikiversity
Free learning tools -
Wiktionary
Dictionary and thesaurus
Sources
- ^ Meija, Juris; et al. (2016). "Atomic weights of the elements 2013 (IUPAC Technical Report)". Pure and Applied Chemistry. 88 (3): 265–91. doi:10.1515/pac-2015-0305.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (2022-05-04). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.