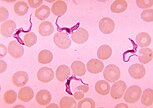
The eukaryotes (/juːˈkærioʊts, -əts/ yoo-KARR-ee-ohts, -əts) constitute the domain of Eukarya or Eukaryota, organisms whose cells have a membrane-bound nucleus. All animals, plants, fungi, and many unicellular organisms are eukaryotes. They constitute a major group of life forms alongside the two groups of prokaryotes: the Bacteria and the Archaea. Eukaryotes represent a small minority of the number of organisms, but given their generally much larger size, their collective global biomass is much larger than that of prokaryotes.
| Eukaryota Temporal range: Statherian–Present
| |
|---|---|
| Scientific classification | |
| Domain: | Eukaryota (Chatton, 1925) Whittaker & Margulis, 1978 |
| Supergroups and kingdoms[2] | |
| Synonyms | |
The eukaryotes seemingly emerged within the Asgard archaea, and are closely related to the Heimdallarchaeia.[5] This implies that there are only two domains of life, Bacteria and Archaea, with eukaryotes incorporated among the Archaea. Eukaryotes first emerged during the Paleoproterozoic, likely as flagellated cells. The leading evolutionary theory is they were created by symbiogenesis between an anaerobic Asgard archaean and an aerobic proteobacterium, which formed the mitochondria. A second episode of symbiogenesis with a cyanobacterium created the plants, with chloroplasts.
Eukaryotic cells contain membrane-bound organelles such as the nucleus, the endoplasmic reticulum, and the Golgi apparatus. Eukaryotes may be either unicellular or multicellular. In comparison, prokaryotes are typically unicellular. Unicellular eukaryotes are sometimes called protists. Eukaryotes can reproduce both asexually through mitosis and sexually through meiosis and gamete fusion (fertilization).
Diversity edit
Eukaryotes are organisms that range from microscopic single cells, such as picozoans under 3 micrometres across,[6] to animals like the blue whale, weighing up to 190 tonnes and measuring up to 33.6 metres (110 ft) long,[7] or plants like the coast redwood, up to 120 metres (390 ft) tall.[8] Many eukaryotes are unicellular; the informal grouping called protists includes many of these, with some multicellular forms like the giant kelp up to 200 feet (61 m) long.[9] The multicellular eukaryotes include the animals, plants, and fungi, but again, these groups too contain many unicellular species.[10] Eukaryotic cells are typically much larger than those of prokaryotes—the bacteria and the archaea—having a volume of around 10,000 times greater.[11][12] Eukaryotes represent a small minority of the number of organisms, but, as many of them are much larger, their collective global biomass (468 gigatons) is far larger than that of prokaryotes (77 gigatons), with plants alone accounting for over 81% of the total biomass of Earth.[13]
- Eukaryotes range in size from single cells to organisms weighing many tons
The eukaryotes are a diverse lineage, consisting mainly of microscopic organisms.[14] Multicellularity in some form has evolved independently at least 25 times within the eukaryotes.[15][16] Complex multicellular organisms, not counting the aggregation of amoebae to form slime molds, have evolved within only six eukaryotic lineages: animals, symbiomycotan fungi, brown algae, red algae, green algae, and land plants.[17] Eukaryotes are grouped by genomic similarities, so that groups often lack visible shared characteristics.[14]
Distinguishing features edit
Nucleus edit
The defining feature of eukaryotes is that their cells have nuclei. This gives them their name, from the Greek εὖ (eu, "well" or "good") and κάρυον (karyon, "nut" or "kernel", here meaning "nucleus").[18] Eukaryotic cells have a variety of internal membrane-bound structures, called organelles, and a cytoskeleton which defines the cell's organization and shape. The nucleus stores the cell's DNA, which is divided into linear bundles called chromosomes;[19] these are separated into two matching sets by a microtubular spindle during nuclear division, in the distinctively eukaryotic process of mitosis.[20]
Biochemistry edit
Eukaryotes differ from prokaryotes in multiple ways, with unique biochemical pathways such as sterane synthesis.[21] The eukaryotic signature proteins have no homology to proteins in other domains of life, but appear to be universal among eukaryotes. They include the proteins of the cytoskeleton, the complex transcription machinery, the membrane-sorting systems, the nuclear pore, and some enzymes in the biochemical pathways.[22]
Internal membranes edit
Eukaryote cells include a variety of membrane-bound structures, together forming the endomembrane system.[23] Simple compartments, called vesicles and vacuoles, can form by budding off other membranes. Many cells ingest food and other materials through a process of endocytosis, where the outer membrane invaginates and then pinches off to form a vesicle.[24] Some cell products can leave in a vesicle through exocytosis.[25]
The nucleus is surrounded by a double membrane known as the nuclear envelope, with nuclear pores that allow material to move in and out.[26] Various tube- and sheet-like extensions of the nuclear membrane form the endoplasmic reticulum, which is involved in protein transport and maturation. It includes the rough endoplasmic reticulum, covered in ribosomes which synthesize proteins; these enter the interior space or lumen. Subsequently, they generally enter vesicles, which bud off from the smooth endoplasmic reticulum.[27] In most eukaryotes, these protein-carrying vesicles are released and further modified in stacks of flattened vesicles (cisternae), the Golgi apparatus.[28]
Vesicles may be specialized; for instance, lysosomes contain digestive enzymes that break down biomolecules in the cytoplasm.[29]
Mitochondria edit
Mitochondria are organelles in eukaryotic cells. The mitochondrion is commonly called "the powerhouse of the cell",[30] for its function providing energy by oxidising sugars or fats to produce the energy-storing molecule ATP.[31][32] Mitochondria have two surrounding membranes, each a phospholipid bilayer; the inner of which is folded into invaginations called cristae where aerobic respiration takes place.[33]
Mitochondria contain their own DNA, which has close structural similarities to bacterial DNA, from which it originated, and which encodes rRNA and tRNA genes that produce RNA which is closer in structure to bacterial RNA than to eukaryote RNA.[34]
Some eukaryotes, such as the metamonads Giardia and Trichomonas, and the amoebozoan Pelomyxa, appear to lack mitochondria, but all contain mitochondrion-derived organelles, like hydrogenosomes or mitosomes, having lost their mitochondria secondarily.[35] They obtain energy by enzymatic action in the cytoplasm.[36][35]
Plastids edit
Plants and various groups of algae have plastids as well as mitochondria. Plastids, like mitochondria, have their own DNA and are developed from endosymbionts, in this case cyanobacteria. They usually take the form of chloroplasts which, like cyanobacteria, contain chlorophyll and produce organic compounds (such as glucose) through photosynthesis. Others are involved in storing food. Although plastids probably had a single origin, not all plastid-containing groups are closely related. Instead, some eukaryotes have obtained them from others through secondary endosymbiosis or ingestion.[37] The capture and sequestering of photosynthetic cells and chloroplasts, kleptoplasty, occurs in many types of modern eukaryotic organisms.[38][39]
Cytoskeletal structures edit
The cytoskeleton provides stiffening structure and points of attachment for motor structures that enable the cell to move, change shape, or transport materials. The motor structures are microfilaments of actin and actin-binding proteins, including α-actinin, fimbrin, and filamin are present in submembranous cortical layers and bundles. Motor proteins of microtubules, dynein and kinesin, and myosin of actin filaments, provide dynamic character of the network.[40][41]
Many eukaryotes have long slender motile cytoplasmic projections, called flagella, or multiple shorter structures called cilia. These organelles are variously involved in movement, feeding, and sensation. They are composed mainly of tubulin, and are entirely distinct from prokaryotic flagella. They are supported by a bundle of microtubules arising from a centriole, characteristically arranged as nine doublets surrounding two singlets. Flagella may have hairs (mastigonemes), as in many Stramenopiles. Their interior is continuous with the cell's cytoplasm.[42][43]
Centrioles are often present, even in cells and groups that do not have flagella, but conifers and flowering plants have neither. They generally occur in groups that give rise to various microtubular roots. These form a primary component of the cytoskeleton, and are often assembled over the course of several cell divisions, with one flagellum retained from the parent and the other derived from it. Centrioles produce the spindle during nuclear division.[44]
Cell wall edit
The cells of plants, algae, fungi and most chromalveolates, but not animals, are surrounded by a cell wall. This is a layer outside the cell membrane, providing the cell with structural support, protection, and a filtering mechanism. The cell wall also prevents over-expansion when water enters the cell.[45]
The major polysaccharides making up the primary cell wall of land plants are cellulose, hemicellulose, and pectin. The cellulose microfibrils are linked together with hemicellulose, embedded in a pectin matrix. The most common hemicellulose in the primary cell wall is xyloglucan.[46]
Sexual reproduction edit
Eukaryotes have a life cycle that involves sexual reproduction, alternating between a haploid phase, where only one copy of each chromosome is present in each cell, and a diploid phase, with two copies of each chromosome in each cell. The diploid phase is formed by fusion of two haploid gametes, such as eggs and spermatozoa, to form a zygote; this may grow into a body, with its cells dividing by mitosis, and at some stage produce haploid gametes through meiosis, a division that reduces the number of chromosomes and creates genetic variability.[47] There is considerable variation in this pattern. Plants have both haploid and diploid multicellular phases.[48] Eukaryotes have lower metabolic rates and longer generation times than prokaryotes, because they are larger and therefore have a smaller surface area to volume ratio.[49]
The evolution of sexual reproduction may be a primordial characteristic of eukaryotes. Based on a phylogenetic analysis, Dacks and Roger have proposed that facultative sex was present in the group's common ancestor.[50] A core set of genes that function in meiosis is present in both Trichomonas vaginalis and Giardia intestinalis, two organisms previously thought to be asexual.[51][52] Since these two species are descendants of lineages that diverged early from the eukaryotic evolutionary tree, core meiotic genes, and hence sex, were likely present in the common ancestor of eukaryotes.[51][52] Species once thought to be asexual, such as Leishmania parasites, have a sexual cycle.[53] Amoebae, previously regarded as asexual, are anciently sexual; present-day asexual groups likely arose recently.[54]
Evolution edit
History of classification edit
In antiquity, the two lineages of animals and plants were recognized by Aristotle and Theophrastus. The lineages were given the taxonomic rank of Kingdom by Linnaeus in the 18th century. Though he included the fungi with plants with some reservations, it was later realized that they are quite distinct and warrant a separate kingdom.[56] The various single-cell eukaryotes were originally placed with plants or animals when they became known. In 1818, the German biologist Georg A. Goldfuss coined the word protozoa to refer to organisms such as ciliates,[57] and this group was expanded until Ernst Haeckel made it a kingdom encompassing all single-celled eukaryotes, the Protista, in 1866.[58][59][60] The eukaryotes thus came to be seen as four kingdoms:
The protists were at that time thought to be "primitive forms", and thus an evolutionary grade, united by their primitive unicellular nature.[59] Understanding of the oldest branchings in the tree of life only developed substantially with DNA sequencing, leading to a system of domains rather than kingdoms as top level rank being put forward by Carl Woese, Otto Kandler, and Mark Wheelis in 1990, uniting all the eukaryote kingdoms in the domain "Eucarya", stating, however, that "'eukaryotes' will continue to be an acceptable common synonym".[3][61] In 1996, the evolutionary biologist Lynn Margulis proposed to replace Kingdoms and Domains with "inclusive" names to create a "symbiosis-based phylogeny", giving the description "Eukarya (symbiosis-derived nucleated organisms)".[4]
Phylogeny edit
By 2014, a rough consensus started to emerge from the phylogenomic studies of the previous two decades.[10][62] The majority of eukaryotes can be placed in one of two large clades dubbed Amorphea (similar in composition to the unikont hypothesis) and the Diphoda (formerly bikonts), which includes plants and most algal lineages. A third major grouping, the Excavata, has been abandoned as a formal group as it is paraphyletic.[2] The proposed phylogeny below includes only one group of excavates (Discoba),[63] and incorporates the 2021 proposal that picozoans are close relatives of rhodophytes.[64] The Provora are a group of microbial predators discovered in 2022.[1]
| Eukaryotes |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2200 mya |
One view of the great kingdoms and their stem groups.[63][65][66][14] The Metamonada are hard to place, being sister possibly to Discoba, possibly to Malawimonada.[14]
Origin of eukaryotes edit
The origin of the eukaryotic cell, or eukaryogenesis, is a milestone in the evolution of life, since eukaryotes include all complex cells and almost all multicellular organisms. The last eukaryotic common ancestor (LECA) is the hypothetical origin of all living eukaryotes,[68] and was most likely a biological population, not a single individual.[69] The LECA is believed to have been a protist with a nucleus, at least one centriole and flagellum, facultatively aerobic mitochondria, sex (meiosis and syngamy), a dormant cyst with a cell wall of chitin or cellulose, and peroxisomes.[70][71][72]
An endosymbiotic union between a motile anaerobic archaean and an aerobic alphaproteobacterium gave rise to the LECA and all eukaryotes, with mitochondria. A second, much later endosymbiosis with a cyanobacterium gave rise to the ancestor of plants, with chloroplasts.[67]
The presence of eukaryotic biomarkers in archaea points towards an archaeal origin. The genomes of Asgard archaea have plenty of Eukaryotic signature protein genes, which play a crucial role in the development of the cytoskeleton and complex cellular structures characteristic of eukaryotes. In 2022, cryo-electron tomography demonstrated that Asgard archaea have a complex actin-based cytoskeleton, providing the first direct visual evidence of the archaeal ancestry of eukaryotes.[73]
Fossils edit
The timing of the origin of eukaryotes is hard to determine but the discovery of Qingshania magnificia, the earliest multicelluar eukaryote from North China which lived during 1.635 billion years ago, suggests that the crown group eukaryotes would have originated from the late Paleoproterozoic (Statherian); the earliest unequivocal unicellular eukaryotes which lived during approximately 1.65 billion years ago are also discovered from North China: Tappania plana, Shuiyousphaeridium macroreticulatum, Dictyosphaera macroreticulata, Germinosphaera alveolata, and Valeria lophostriata.[74]
Some acritarchs are known from at least 1.65 billion years ago, and a fossil, Grypania, which may be an alga, is as much as 2.1 billion years old.[75][76] The "problematic"[77] fossil Diskagma has been found in paleosols 2.2 billion years old.[77]
Structures proposed to represent "large colonial organisms" have been found in the black shales of the Palaeoproterozoic such as the Francevillian B Formation, in Gabon, dubbed the "Francevillian biota" which is dated at 2.1 billion years old.[78][79] However, the status of these structures as fossils is contested, with other authors suggesting that they might represent pseudofossils.[80] The oldest fossils than can unambiguously be assigned to eukaryotes are from the Ruyang Group of China, dating to approximately 1.8-1.6 billion years ago.[81] Fossils that are clearly related to modern groups start appearing an estimated 1.2 billion years ago, in the form of red algae, though recent work suggests the existence of fossilized filamentous algae in the Vindhya basin dating back perhaps to 1.6 to 1.7 billion years ago.[82]
The presence of steranes, eukaryotic-specific biomarkers, in Australian shales previously indicated that eukaryotes were present in these rocks dated at 2.7 billion years old,[21][83] but these Archaean biomarkers have been rebutted as later contaminants.[84] The oldest valid biomarker records are only around 800 million years old.[85] In contrast, a molecular clock analysis suggests the emergence of sterol biosynthesis as early as 2.3 billion years ago.[86] The nature of steranes as eukaryotic biomarkers is further complicated by the production of sterols by some bacteria.[87][88]
Whenever their origins, eukaryotes may not have become ecologically dominant until much later; a massive increase in the zinc composition of marine sediments 800 million years ago has been attributed to the rise of substantial populations of eukaryotes, which preferentially consume and incorporate zinc relative to prokaryotes, approximately a billion years after their origin (at the latest).[89]
See also edit
References edit
- ^ a b Tikhonenkov DV, Mikhailov KV, Gawryluk RM, et al. (December 2022). "Microbial predators form a new supergroup of eukaryotes". Nature. 612 (7941): 714–719. Bibcode:2022Natur.612..714T. doi:10.1038/s41586-022-05511-5. PMID 36477531. S2CID 254436650.
- ^ a b Adl SM, Bass D, Lane CE, et al. (January 2019). "Revisions to the Classification, Nomenclature, and Diversity of Eukaryotes". The Journal of Eukaryotic Microbiology. 66 (1): 4–119. doi:10.1111/jeu.12691. PMC 6492006. PMID 30257078.
- ^ a b Woese CR, Kandler O, Wheelis ML (June 1990). "Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya". Proceedings of the National Academy of Sciences of the United States of America. 87 (12): 4576–4579. Bibcode:1990PNAS...87.4576W. doi:10.1073/pnas.87.12.4576. PMC 54159. PMID 2112744.
- ^ a b Margulis L (6 February 1996). "Archaeal-eubacterial mergers in the origin of Eukarya: phylogenetic classification of life". Proceedings of the National Academy of Sciences. 93 (3): 1071–1076. Bibcode:1996PNAS...93.1071M. doi:10.1073/pnas.93.3.1071. PMC 40032. PMID 8577716.
- ^ Eme, Laura; Tamarit, Daniel; Caceres, Eva F.; Stairs, Courtney W.; De Anda, Valerie; Schön, Max E.; Seitz, Kiley W.; Dombrowski, Nina; Lewis, William H.; Homa, Felix; Saw, Jimmy H.; Lombard, Jonathan; Nunoura, Takuro; Li, Wen-Jun; Hua, Zheng-Shuang; Chen, Lin-Xing; Banfield, Jillian F.; John, Emily St; Reysenbach, Anna-Louise; Stott, Matthew B.; Schramm, Andreas; Kjeldsen, Kasper U.; Teske, Andreas P.; Baker, Brett J.; Ettema, Thijs J. G. (29 June 2023). "Inference and reconstruction of the heimdallarchaeial ancestry of eukaryotes". Nature. 618 (7967): 992–999. Bibcode:2023Natur.618..992E. doi:10.1038/s41586-023-06186-2. ISSN 1476-4687. PMC 10307638. PMID 37316666.
- ^ Seenivasan R, Sausen N, Medlin LK, Melkonian M (26 March 2013). "Picomonas judraskeda Gen. Et Sp. Nov.: The First Identified Member of the Picozoa Phylum Nov., a Widespread Group of Picoeukaryotes, Formerly Known as 'Picobiliphytes'". PLOS ONE. 8 (3): e59565. Bibcode:2013PLoSO...859565S. doi:10.1371/journal.pone.0059565. PMC 3608682. PMID 23555709.
- ^ Wood G (1983). The Guinness Book of Animal Facts and Feats. Enfield, Middlesex : Guinness Superlatives. ISBN 978-0-85112-235-9.
- ^ Earle CJ, ed. (2017). "Sequoia sempervirens". The Gymnosperm Database. Archived from the original on 1 April 2016. Retrieved 15 September 2017.
- ^ van den Hoek C, Mann D, Jahns H (1995). Algae An Introduction to Phycology. Cambridge: Cambridge University Press. ISBN 0-521-30419-9. Archived from the original on 10 February 2023. Retrieved 7 April 2023.
- ^ a b Burki F (May 2014). "The eukaryotic tree of life from a global phylogenomic perspective". Cold Spring Harbor Perspectives in Biology. 6 (5): a016147. doi:10.1101/cshperspect.a016147. PMC 3996474. PMID 24789819.
- ^ DeRennaux B (2001). "Eukaryotes, Origin of". Encyclopedia of Biodiversity. Vol. 2. Elsevier. pp. 329–332. doi:10.1016/b978-0-12-384719-5.00174-x. ISBN 9780123847201.
- ^ Yamaguchi M, Worman CO (2014). "Deep-sea microorganisms and the origin of the eukaryotic cell" (PDF). Japanese Journal of Protozoology. 47 (1, 2): 29–48. Archived from the original (PDF) on 9 August 2017.
- ^ Bar-On, Yinon M.; Phillips, Rob; Milo, Ron (17 May 2018). "The biomass distribution on Earth". Proceedings of the National Academy of Sciences. 115 (25): 6506–6511. Bibcode:2018PNAS..115.6506B. doi:10.1073/pnas.1711842115. ISSN 0027-8424. PMC 6016768. PMID 29784790.
- ^ a b c d Burki F, Roger AJ, Brown MW, Simpson AG (2020). "The New Tree of Eukaryotes". Trends in Ecology & Evolution. 35 (1). Elsevier BV: 43–55. doi:10.1016/j.tree.2019.08.008. ISSN 0169-5347. PMID 31606140. S2CID 204545629.
- ^ Grosberg RK, Strathmann RR (2007). "The evolution of multicellularity: A minor major transition?" (PDF). Annu Rev Ecol Evol Syst. 38: 621–654. doi:10.1146/annurev.ecolsys.36.102403.114735. Archived (PDF) from the original on 14 March 2023. Retrieved 8 April 2023.
- ^ Parfrey L, Lahr D (2013). "Multicellularity arose several times in the evolution of eukaryotes" (PDF). BioEssays. 35 (4): 339–347. doi:10.1002/bies.201200143. PMID 23315654. S2CID 13872783. Archived (PDF) from the original on 25 July 2014. Retrieved 8 April 2023.
- ^ Popper ZA, Michel G, Hervé C, Domozych DS, Willats WG, Tuohy MG, Kloareg B, Stengel DB (2011). "Evolution and diversity of plant cell walls: From algae to flowering plants". Annual Review of Plant Biology. 62: 567–590. doi:10.1146/annurev-arplant-042110-103809. hdl:10379/6762. PMID 21351878. S2CID 11961888.
- ^ Harper, Douglas. "eukaryotic". Online Etymology Dictionary.
- ^ Bonev B, Cavalli G (14 October 2016). "Organization and function of the 3D genome". Nature Reviews Genetics. 17 (11): 661–678. doi:10.1038/nrg.2016.112. hdl:2027.42/151884. PMID 27739532. S2CID 31259189.
- ^ O'Connor, Clare (2008). "Chromosome Segregation: The Role of Centromeres". Nature Education. Retrieved 18 February 2024.
eukar
- ^ a b Brocks JJ, Logan GA, Buick R, Summons RE (August 1999). "Archean molecular fossils and the early rise of eukaryotes". Science. 285 (5430): 1033–1036. Bibcode:1999Sci...285.1033B. CiteSeerX 10.1.1.516.9123. doi:10.1126/science.285.5430.1033. PMID 10446042.
- ^ Hartman H, Fedorov A (February 2002). "The origin of the eukaryotic cell: a genomic investigation". Proceedings of the National Academy of Sciences of the United States of America. 99 (3): 1420–5. Bibcode:2002PNAS...99.1420H. doi:10.1073/pnas.032658599. PMC 122206. PMID 11805300.
- ^ Linka M, Weber AP (2011). "Evolutionary Integration of Chloroplast Metabolism with the Metabolic Networks of the Cells". In Burnap RL, Vermaas WF (eds.). Functional Genomics and Evolution of Photosynthetic Systems. Springer. p. 215. ISBN 978-94-007-1533-2. Archived from the original on 29 May 2016. Retrieved 27 October 2015.
- ^ Marsh M (2001). Endocytosis. Oxford University Press. p. vii. ISBN 978-0-19-963851-2.
- ^ Stalder D, Gershlick DC (November 2020). "Direct trafficking pathways from the Golgi apparatus to the plasma membrane". Seminars in Cell & Developmental Biology. 107: 112–125. doi:10.1016/j.semcdb.2020.04.001. PMC 7152905. PMID 32317144.
- ^ Hetzer MW (March 2010). "The nuclear envelope". Cold Spring Harbor Perspectives in Biology. 2 (3): a000539. doi:10.1101/cshperspect.a000539. PMC 2829960. PMID 20300205.
- ^ "Endoplasmic Reticulum (Rough and Smooth)". British Society for Cell Biology. Archived from the original on 24 March 2019. Retrieved 12 November 2017.
- ^ "Golgi Apparatus". British Society for Cell Biology. Archived from the original on 13 November 2017. Retrieved 12 November 2017.
- ^ "Lysosome". British Society for Cell Biology. Archived from the original on 13 November 2017. Retrieved 12 November 2017.
- ^ Saygin D, Tabib T, Bittar HE, et al. (July 1957). "Transcriptional profiling of lung cell populations in idiopathic pulmonary arterial hypertension". Pulmonary Circulation. 10 (1): 131–144. Bibcode:1957SciAm.197a.131S. doi:10.1038/scientificamerican0757-131. PMC 7052475. PMID 32166015.
- ^ Voet D, Voet JC, Pratt CW (2006). Fundamentals of Biochemistry (2nd ed.). John Wiley and Sons. pp. 547, 556. ISBN 978-0471214953.
- ^ Mack S (1 May 2006). "Re: Are there eukaryotic cells without mitochondria?". madsci.org. Archived from the original on 24 April 2014. Retrieved 24 April 2014.
- ^ Zick M, Rabl R, Reichert AS (January 2009). "Cristae formation-linking ultrastructure and function of mitochondria". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 1793 (1): 5–19. doi:10.1016/j.bbamcr.2008.06.013. PMID 18620004.
- ^ Watson J, Hopkins N, Roberts J, Steitz JA, Weiner A (1988). "28: The Origins of Life". Molecular Biology of the Gene (Fourth ed.). Menlo Park, California: The Benjamin/Cummings Publishing Company, Inc. p. 1154. ISBN 978-0-8053-9614-0.
- ^ a b Karnkowska A, Vacek V, Zubáčová Z, et al. (May 2016). "A Eukaryote without a Mitochondrial Organelle". Current Biology. 26 (10): 1274–1284. Bibcode:2016CBio...26.1274K. doi:10.1016/j.cub.2016.03.053. PMID 27185558.
- ^ Davis JL (13 May 2016). "Scientists Shocked To Discover Eukaryote With NO Mitochondria". IFL Science. Archived from the original on 17 February 2019. Retrieved 13 May 2016.
- ^ Sato N (2006). "Origin and Evolution of Plastids: Genomic View on the Unification and Diversity of Plastids". In Wise RR, Hoober JK (eds.). The Structure and Function of Plastids. Advances in Photosynthesis and Respiration. Vol. 23. Springer Netherlands. pp. 75–102. doi:10.1007/978-1-4020-4061-0_4. ISBN 978-1-4020-4060-3.
- ^ Minnhagen S, Carvalho WF, Salomon PS, Janson S (September 2008). "Chloroplast DNA content in Dinophysis (Dinophyceae) from different cell cycle stages is consistent with kleptoplasty". Environ. Microbiol. 10 (9): 2411–7. Bibcode:2008EnvMi..10.2411M. doi:10.1111/j.1462-2920.2008.01666.x. PMID 18518896.
- ^ Bodył A (February 2018). "Did some red alga-derived plastids evolve via kleptoplastidy? A hypothesis". Biological Reviews of the Cambridge Philosophical Society. 93 (1): 201–222. doi:10.1111/brv.12340. PMID 28544184. S2CID 24613863.
- ^ Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (1 January 2002). "Molecular Motors". Molecular Biology of the Cell (4th ed.). New York: Garland Science. ISBN 978-0-8153-3218-3. Archived from the original on 8 March 2019. Retrieved 6 April 2023.
- ^ Sweeney HL, Holzbaur EL (May 2018). "Motor Proteins". Cold Spring Harbor Perspectives in Biology. 10 (5): a021931. doi:10.1101/cshperspect.a021931. PMC 5932582. PMID 29716949.
- ^ Bardy SL, Ng SY, Jarrell KF (February 2003). "Prokaryotic motility structures". Microbiology. 149 (Pt 2): 295–304. doi:10.1099/mic.0.25948-0. PMID 12624192.
- ^ Silflow CD, Lefebvre PA (December 2001). "Assembly and motility of eukaryotic cilia and flagella. Lessons from Chlamydomonas reinhardtii". Plant Physiology. 127 (4): 1500–7. doi:10.1104/pp.010807. PMC 1540183. PMID 11743094.
- ^ Vorobjev IA, Nadezhdina ES (1987). The centrosome and its role in the organization of microtubules. International Review of Cytology. Vol. 106. pp. 227–293. doi:10.1016/S0074-7696(08)61714-3. ISBN 978-0-12-364506-7. PMID 3294718.
- ^ Howland JL (2000). The Surprising Archaea: Discovering Another Domain of Life. Oxford: Oxford University Press. pp. 69–71. ISBN 978-0-19-511183-5.
- ^ Fry SC (1989). "The Structure and Functions of Xyloglucan". Journal of Experimental Botany. 40 (1): 1–11. doi:10.1093/jxb/40.1.1.
- ^ Hamilton MB (2009). Population genetics. Wiley-Blackwell. p. 55. ISBN 978-1-4051-3277-0.
- ^ Taylor TN, Kerp H, Hass H (2005). "Life history biology of early land plants: Deciphering the gametophyte phase". Proceedings of the National Academy of Sciences of the United States of America. 102 (16): 5892–5897. doi:10.1073/pnas.0501985102. PMC 556298. PMID 15809414.
- ^ Lane N (June 2011). "Energetics and genetics across the prokaryote-eukaryote divide". Biology Direct. 6 (1): 35. doi:10.1186/1745-6150-6-35. PMC 3152533. PMID 21714941.
- ^ Dacks J, Roger AJ (June 1999). "The first sexual lineage and the relevance of facultative sex". Journal of Molecular Evolution. 48 (6): 779–783. Bibcode:1999JMolE..48..779D. doi:10.1007/PL00013156. PMID 10229582. S2CID 9441768.
- ^ a b Ramesh MA, Malik SB, Logsdon JM (January 2005). "A phylogenomic inventory of meiotic genes; evidence for sex in Giardia and an early eukaryotic origin of meiosis". Current Biology. 15 (2): 185–191. Bibcode:2005CBio...15..185R. doi:10.1016/j.cub.2005.01.003. PMID 15668177. S2CID 17013247.
- ^ a b Malik SB, Pightling AW, Stefaniak LM, Schurko AM, Logsdon JM (August 2007). Hahn MW (ed.). "An expanded inventory of conserved meiotic genes provides evidence for sex in Trichomonas vaginalis". PLOS ONE. 3 (8): e2879. Bibcode:2008PLoSO...3.2879M. doi:10.1371/journal.pone.0002879. PMC 2488364. PMID 18663385.
- ^ Akopyants NS, Kimblin N, Secundino N, Patrick R, Peters N, Lawyer P, Dobson DE, Beverley SM, Sacks DL (April 2009). "Demonstration of genetic exchange during cyclical development of Leishmania in the sand fly vector". Science. 324 (5924): 265–268. Bibcode:2009Sci...324..265A. doi:10.1126/science.1169464. PMC 2729066. PMID 19359589.
- ^ Lahr DJ, Parfrey LW, Mitchell EA, Katz LA, Lara E (July 2011). "The chastity of amoebae: re-evaluating evidence for sex in amoeboid organisms". Proceedings: Biological Sciences. 278 (1715): 2081–2090. doi:10.1098/rspb.2011.0289. PMC 3107637. PMID 21429931.
- ^ Patrick J. Keeling; Yana Eglit (21 November 2023). "Openly available illustrations as tools to describe eukaryotic microbial diversity". PLOS Biology. 21 (11): e3002395. doi:10.1371/JOURNAL.PBIO.3002395. ISSN 1544-9173. PMC 10662721. PMID 37988341. Wikidata Q123558544.
- ^ Moore RT (1980). "Taxonomic proposals for the classification of marine yeasts and other yeast-like fungi including the smuts". Botanica Marina. 23: 361–373.
- ^ Goldfuß (1818). "Ueber die Classification der Zoophyten" [On the classification of zoophytes]. Isis, Oder, Encyclopädische Zeitung von Oken (in German). 2 (6): 1008–1019. Archived from the original on 24 March 2019. Retrieved 15 March 2019. From p. 1008: "Erste Klasse. Urthiere. Protozoa." (First class. Primordial animals. Protozoa.) [Note: each column of each page of this journal is numbered; there are two columns per page.]
- ^ Scamardella JM (1999). "Not plants or animals: a brief history of the origin of Kingdoms Protozoa, Protista and Protoctista" (PDF). International Microbiology. 2 (4): 207–221. PMID 10943416. Archived from the original (PDF) on 14 June 2011.
- ^ a b Rothschild LJ (1989). "Protozoa, Protista, Protoctista: what's in a name?". Journal of the History of Biology. 22 (2): 277–305. doi:10.1007/BF00139515. PMID 11542176. S2CID 32462158. Archived from the original on 4 February 2020. Retrieved 4 February 2020.
- ^ Whittaker RH (January 1969). "New concepts of kingdoms or organisms. Evolutionary relations are better represented by new classifications than by the traditional two kingdoms". Science. 163 (3863): 150–60. Bibcode:1969Sci...163..150W. CiteSeerX 10.1.1.403.5430. doi:10.1126/science.163.3863.150. PMID 5762760.
- ^ Knoll AH (1992). "The Early Evolution of Eukaryotes: A Geological Perspective". Science. 256 (5057): 622–627. Bibcode:1992Sci...256..622K. doi:10.1126/science.1585174. PMID 1585174.
Eucarya, or eukaryotes
- ^ Burki F, Kaplan M, Tikhonenkov DV, et al. (January 2016). "Untangling the early diversification of eukaryotes: a phylogenomic study of the evolutionary origins of Centrohelida, Haptophyta and Cryptista". Proceedings: Biological Sciences. 283 (1823): 20152802. doi:10.1098/rspb.2015.2802. PMC 4795036. PMID 26817772.
- ^ a b Brown MW, Heiss AA, Kamikawa R, Inagaki Y, Yabuki A, Tice AK, Shiratori T, Ishida KI, Hashimoto T, Simpson A, Roger A (19 January 2018). "Phylogenomics Places Orphan Protistan Lineages in a Novel Eukaryotic Super-Group". Genome Biology and Evolution. 10 (2): 427–433. doi:10.1093/gbe/evy014. PMC 5793813. PMID 29360967.
- ^ Schön ME, Zlatogursky VV, Singh RP, et al. (2021). "Picozoa are archaeplastids without plastid". Nature Communications. 12 (1): 6651. bioRxiv 10.1101/2021.04.14.439778. doi:10.1038/s41467-021-26918-0. PMC 8599508. PMID 34789758. S2CID 233328713. Archived from the original on 2 February 2024. Retrieved 20 December 2021.
- ^ Schön ME, Zlatogursky VV, Singh RP, et al. (2021). "Picozoa are archaeplastids without plastid". Nature Communications. 12 (1): 6651. bioRxiv 10.1101/2021.04.14.439778. doi:10.1038/s41467-021-26918-0. PMC 8599508. PMID 34789758. S2CID 233328713.
- ^ Tikhonenkov DV, Mikhailov KV, Gawryluk RM, et al. (December 2022). "Microbial predators form a new supergroup of eukaryotes". Nature. 612 (7941): 714–719. doi:10.1038/s41586-022-05511-5. PMID 36477531. S2CID 254436650.
- ^ a b Latorre A, Durban A, Moya A, Pereto J (2011). "The role of symbiosis in eukaryotic evolution". In Gargaud M, López-Garcìa P, Martin H (eds.). Origins and Evolution of Life: An astrobiological perspective. Cambridge: Cambridge University Press. pp. 326–339. ISBN 978-0-521-76131-4. Archived from the original on 24 March 2019. Retrieved 27 August 2017.
- ^ Gabaldón T (October 2021). "Origin and Early Evolution of the Eukaryotic Cell". Annual Review of Microbiology. 75 (1): 631–647. doi:10.1146/annurev-micro-090817-062213. PMID 34343017. S2CID 236916203.
- ^ O'Malley MA, Leger MM, Wideman JG, Ruiz-Trillo I (March 2019). "Concepts of the last eukaryotic common ancestor". Nature Ecology & Evolution. 3 (3): 338–344. Bibcode:2019NatEE...3..338O. doi:10.1038/s41559-019-0796-3. hdl:10261/201794. PMID 30778187. S2CID 67790751.
- ^ Leander BS (May 2020). "Predatory protists". Current Biology. 30 (10): R510–R516. Bibcode:2020CBio...30.R510L. doi:10.1016/j.cub.2020.03.052. PMID 32428491. S2CID 218710816.
- ^ Strassert JF, Irisarri I, Williams TA, Burki F (March 2021). "A molecular timescale for eukaryote evolution with implications for the origin of red algal-derived plastids". Nature Communications. 12 (1): 1879. Bibcode:2021NatCo..12.1879S. doi:10.1038/s41467-021-22044-z. PMC 7994803. PMID 33767194.
- ^ Koumandou VL, Wickstead B, Ginger ML, van der Giezen M, Dacks JB, Field MC (2013). "Molecular paleontology and complexity in the last eukaryotic common ancestor". Critical Reviews in Biochemistry and Molecular Biology. 48 (4): 373–396. doi:10.3109/10409238.2013.821444. PMC 3791482. PMID 23895660.
- ^ Rodrigues-Oliveira T, Wollweber F, Ponce-Toledo RI, et al. (2023). "Actin cytoskeleton and complex cell architecture in an Asgard archaean". Nature. 613 (7943): 332–339. Bibcode:2023Natur.613..332R. doi:10.1038/s41586-022-05550-y. hdl:20.500.11850/589210. PMC 9834061. PMID 36544020.
- ^ Miao, L.; Yin, Z.; Knoll, A. H.; Qu, Y.; Zhu, M. (2024). "1.63-billion-year-old multicellular eukaryotes from the Chuanlinggou Formation in North China". Science Advances. 10 (4): eadk3208. Bibcode:2024SciA...10K3208M. doi:10.1126/sciadv.adk3208. PMC 10807817. PMID 38266082.
- ^ Han TM, Runnegar B (July 1992). "Megascopic eukaryotic algae from the 2.1-billion-year-old negaunee iron-formation, Michigan". Science. 257 (5067): 232–5. Bibcode:1992Sci...257..232H. doi:10.1126/science.1631544. PMID 1631544.
- ^ Knoll AH, Javaux EJ, Hewitt D, Cohen P (June 2006). "Eukaryotic organisms in Proterozoic oceans". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 361 (1470): 1023–1038. doi:10.1098/rstb.2006.1843. PMC 1578724. PMID 16754612.
- ^ a b c Retallack GJ, Krull ES, Thackray GD, Parkinson DH (2013). "Problematic urn-shaped fossils from a Paleoproterozoic (2.2 Ga) paleosol in South Africa". Precambrian Research. 235: 71–87. Bibcode:2013PreR..235...71R. doi:10.1016/j.precamres.2013.05.015.
- ^ El Albani A, Bengtson S, Canfield DE, et al. (July 2010). "Large colonial organisms with coordinated growth in oxygenated environments 2.1 Gyr ago". Nature. 466 (7302): 100–104. Bibcode:2010Natur.466..100A. doi:10.1038/nature09166. PMID 20596019. S2CID 4331375.
- ^ El Albani, Abderrazak (2023). "A search for life in Palaeoproterozoic marine sediments using Zn isotopes and geochemistry" (PDF). Earth and Planetary Science Letters. 623: 118169. Bibcode:2023E&PSL.61218169E. doi:10.1016/j.epsl.2023.118169. S2CID 258360867.
- ^ Ossa Ossa, Frantz; Pons, Marie-Laure; Bekker, Andrey; Hofmann, Axel; Poulton, Simon W.; et al. (2023). "Zinc enrichment and isotopic fractionation in a marine habitat of the c. 2.1 Ga Francevillian Group: A signature of zinc utilization by eukaryotes?" (PDF). Earth and Planetary Science Letters. 611: 118147. Bibcode:2023E&PSL.61118147O. doi:10.1016/j.epsl.2023.118147.
- ^ Fakhraee, Mojtaba; Tarhan, Lidya G.; Reinhard, Christopher T.; Crowe, Sean A.; Lyons, Timothy W.; Planavsky, Noah J. (May 2023). "Earth's surface oxygenation and the rise of eukaryotic life: Relationships to the Lomagundi positive carbon isotope excursion revisited". Earth-Science Reviews. 240: 104398. Bibcode:2023ESRv..24004398F. doi:10.1016/j.earscirev.2023.104398. S2CID 257761993.
- ^ Bengtson S, Belivanova V, Rasmussen B, Whitehouse M (May 2009). "The controversial "Cambrian" fossils of the Vindhyan are real but more than a billion years older". Proceedings of the National Academy of Sciences of the United States of America. 106 (19): 7729–7734. Bibcode:2009PNAS..106.7729B. doi:10.1073/pnas.0812460106. PMC 2683128. PMID 19416859.
- ^ Ward P (9 February 2008). "Mass extinctions: the microbes strike back". New Scientist. pp. 40–43. Archived from the original on 8 July 2008. Retrieved 27 August 2017.
- ^ French KL, Hallmann C, Hope JM, Schoon PL, Zumberge JA, Hoshino Y, Peters CA, George SC, Love GD, Brocks JJ, Buick R, Summons RE (May 2015). "Reappraisal of hydrocarbon biomarkers in Archean rocks". Proceedings of the National Academy of Sciences of the United States of America. 112 (19): 5915–5920. Bibcode:2015PNAS..112.5915F. doi:10.1073/pnas.1419563112. PMC 4434754. PMID 25918387.
- ^ Brocks JJ, Jarrett AJ, Sirantoine E, Hallmann C, Hoshino Y, Liyanage T (August 2017). "The rise of algae in Cryogenian oceans and the emergence of animals". Nature. 548 (7669): 578–581. Bibcode:2017Natur.548..578B. doi:10.1038/nature23457. PMID 28813409. S2CID 205258987.
- ^ Gold DA, Caron A, Fournier GP, Summons RE (March 2017). "Paleoproterozoic sterol biosynthesis and the rise of oxygen". Nature. 543 (7645): 420–423. Bibcode:2017Natur.543..420G. doi:10.1038/nature21412. hdl:1721.1/128450. PMID 28264195. S2CID 205254122.
- ^ Wei JH, Yin X, Welander PV (24 June 2016). "Sterol Synthesis in Diverse Bacteria". Frontiers in Microbiology. 7: 990. doi:10.3389/fmicb.2016.00990. PMC 4919349. PMID 27446030.
- ^ Hoshino Y, Gaucher EA (June 2021). "Evolution of bacterial steroid biosynthesis and its impact on eukaryogenesis". Proceedings of the National Academy of Sciences of the United States of America. 118 (25): e2101276118. Bibcode:2021PNAS..11801276H. doi:10.1073/pnas.2101276118. PMC 8237579. PMID 34131078.
- ^ Isson TT, Love GD, Dupont CL, et al. (June 2018). "Tracking the rise of eukaryotes to ecological dominance with zinc isotopes". Geobiology. 16 (4): 341–352. Bibcode:2018Gbio...16..341I. doi:10.1111/gbi.12289. PMID 29869832.