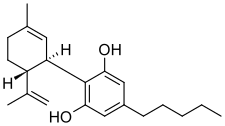
Cannabidiol (CBD) (/kæ.nə.bə.ˈdaɪ.əl/) is a phytocannabinoid discovered in 1940. It is one of 113 identified cannabinoids in cannabis plants, along with tetrahydrocannabinol (THC), and accounts for up to 40% of the plant's extract.[14] As of 2022[update], clinical research on CBD included studies related to the treatment of anxiety, addiction, psychosis, movement disorders, and pain, but there is insufficient high-quality evidence that cannabidiol is effective for these conditions.[15][16][17][18] CBD is also sold as a herbal dietary supplement promoted with unproven claims of particular therapeutic effects.[19]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Epidiolex, Epidyolex |
| Other names | CBD, cannabidiolum, (−)-cannabidiol[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618051 |
| License data |
|
| Pregnancy category |
|
| Addiction liability | Low[3] |
| Routes of administration | Inhalation (smoking, vaping), buccal (aerosol spray),[4][5] oral (solution)[6][7] |
| Drug class | cannabinoid |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | |
| Elimination half-life | 18–32 hours[13] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.215.986 |
| Chemical and physical data | |
| Formula | C21H30O2 |
| Molar mass | 314.469 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 66 °C (151 °F) |
| Solubility in water | Insoluble |
| |
| |
| (verify) | |
Cannabidiol can be taken internally in multiple ways, including by inhaling cannabis smoke or vapor, oral, and as an aerosol spray into the cheek.[4][5] It may be supplied as CBD oil containing only CBD as the active ingredient (excluding THC or terpenes), CBD-dominant hemp extract oil, capsules, dried cannabis, or prescription liquid solution.[7][17] CBD does not have the same psychoactivity as THC,[20][21] and can modulate the psychoactive effects of THC on the body if both are present.[14][20][22][23] CBD heated to 250–300 °C may partially be converted into THC.[24]
In the United States, the cannabidiol drug Epidiolex was approved by the Food and Drug Administration (FDA) in 2018 for the treatment of two epilepsy disorders.[7] While the 2018 United States Farm Bill removed hemp and hemp extracts (including CBD) from the Controlled Substances Act, the marketing and sale of CBD formulations for medical use or as an ingredient in dietary supplements or manufactured foods remains illegal under FDA regulation, as of 2024[update].[25][26]
Medical uses edit
Cannabidiol is the generic name of the drug and its INN.[27]
Research edit
As of 2022[update], there was little evidence for cannabidiol having a neurological effect in humans.[15][16][17][28][29]
In the United States, federal illegality has made it difficult historically to conduct research on CBD.[30]
Epilepsy edit
In the United States, the FDA has indicated only one brand of prescription cannabidiol called Epidiolex for the treatment of seizures associated with Dravet syndrome, Lennox–Gastaut syndrome, or tuberous sclerosis complex in people one year of age and older.[7][31][32][33] While Epidiolex treatment is generally well tolerated, it is associated with minor adverse effects, such as gastrointestinal upset, decreased appetite, lethargy, sleepiness and poor sleep quality.[7][32][34][33]
In the European Union, cannabidiol (Epidyolex) is indicated for use as adjunctive therapy of seizures associated with Lennox–Gastaut syndrome or Dravet syndrome, in conjunction with clobazam, for people two years of age and older.[10] In 2020, the label for Epidiolex in the US was expanded to include seizures associated with tuberous sclerosis complex. Epidiolex/Epidyolex is the first prescription formulation of plant-derived cannabidiol approved by regulatory bodies in the US and Europe.[35]
Other uses edit
Research on other uses for cannabidiol includes several neurological disorders, but the findings have not been confirmed to establish such uses in clinical practice.[13][16][17][20][28][36][37] In October 2019, the FDA issued an advisory warning that the effects of CBD during pregnancy or breastfeeding are unknown, indicating that the safety, doses, interactions with other drugs or foods, and side effects of CBD are not clinically defined, and may pose a risk to the mother and infant.[38]
Many claims are made for the therapeutic benefit of cannabidiol that are not backed by sound evidence. Some claims, such as treatment of cancer, are pseudoscience.[19]
In 2020, the label for Epidiolex in the US was expanded to include treatment of seizures associated with tuberous sclerosis.[31]
Acclaimed for relieving chronic pain, some researchers conclude that the evidence is insufficient to determine the effectiveness of CBD in pain relief, primarily due to the challenging access to pure CBD.[39]
Non-intoxicating effects edit
Cannabidiol does not appear to have any intoxicating effects[40] such as those caused by ∆9-THC in cannabis, but it is under preliminary research for its possible anti-anxiety and anti-psychotic effects.[16][17][21] As the legal landscape and understanding about the differences in medical cannabinoids unfolds, experts are working to distinguish "medical cannabis" (with varying degrees of psychotropic effects and deficits in executive function) from "medical CBD therapies", which would commonly present as having a reduced or non-psychoactive side-effect profile.[17][21][41]
Various strains of "medical cannabis" are found to have a significant variation in the ratios of CBD-to-THC and are known to contain other non-psychotropic cannabinoids.[42] Any psychoactive cannabis, regardless of its CBD content, is derived from the flower (or bud) of the genus Cannabis. As defined by US federal law, non-psychoactive hemp (also commonly termed "industrial hemp"), regardless of its CBD content, is any part of the cannabis plant, whether growing or not, containing a ∆9-tetrahydrocannabinol concentration of no more than 0.3% on a dry-weight basis.[43] In the United States, certain standards are required for legal growing, cultivating, and producing the hemp plant, but there are no federal standards for quality being enforced in the hemp industry. Certain state regulations are in place, but vary state to state.[44] For instance, the Colorado Industrial Hemp Program registers growers of industrial hemp and samples crops to verify that the dry-weight THC concentration does not exceed 0.3%.[43]
Side effects edit
Research indicates that cannabidiol may reduce adverse effects of THC, particularly those causing intoxication and sedation, but only at high doses.[45] Safety studies of cannabidiol showed it is well tolerated, but may cause tiredness, somnolence, sedation, diarrhea, or changes in appetite as common adverse effects with the most common being somnolence and sedation. Side effects of CBD are dose related.[46] Epidiolex documentation lists sleepiness, insomnia and poor quality sleep, decreased appetite, diarrhea, and fatigue.[7][47]
In November 2019, the FDA issued concerns about the safety of cannabidiol, stating that CBD use has potential to cause liver injury, interfere with the mechanisms of prescription drugs, produce gastrointestinal disorders, or affect alertness and mood.[48] Over 2020–23, the FDA updated its safety concerns about CBD,[49] acknowledging the unknown effects of protracted use, how it affects the developing brain, fetus or infants during breastfeeding, whether it interacts with dietary supplements or prescription drugs, whether male fertility is affected, and its possible side effects, such as drowsiness.[50]
As of September 2019[update], 1,085 people contacted US poison control centers about CBD-induced illnesses, doubling the number of cases over the 2018 rate and increasing by 9 times the case numbers of 2017.[51] Of cases reported in 2019, more than 33% received medical attention and 46 people were admitted to a hospital intensive care unit, possibly due to exposure to other products, or drug interactions with CBD.[52]
In 2022, the FDA stated that "scientific studies show possible harm to the male reproductive system, including testicular atrophy, harm to the liver, and interactions with certain medications. The FDA has not found adequate information showing how much CBD can be consumed, and for how long, before causing harm. This is particularly true for vulnerable populations like children and those who are pregnant."[53]
Potential interactions edit
Laboratory evidence indicated that cannabidiol may reduce THC clearance, increasing plasma concentrations which may raise THC availability to receptors and enhance its effect in a dose-dependent manner.[54][55] In vitro, cannabidiol inhibited the activity of voltage-dependent sodium and potassium channels, which may affect neural activity.[56] A recent study using X-ray crystallography showed that CBD binds inside the sodium channel pore at a novel site at the interface of the fenestrations and the central hydrophobic cavity of the channel. Binding at this site blocks the transmembrane-spanning sodium ion translocation pathway, providing a molecular mechanism for channel inhibition, which could contribute to a reduced excitability.[57] A small clinical trial reported that CBD partially inhibited the CYP2C-catalyzed hydroxylation of THC to 11-OH-THC.[58] Little is known about potential drug interactions, but CBD mediates a decrease in clobazam metabolism.[59] Work with human liver microsomes shows that cannabidiol inhibits CYP3A5 and CYP3A4 to some degree.[60]
Veterinary medicine edit
Research edit
The number of research projects and scientific publications on cannabidiol and other cannabinoids in pets surged in the late 2010s; nonetheless, as of December 2020[update], there were no hemp-derived, cannabinoid-rich registered veterinary medicinal products in any of the major regions (see #Legal status).
In the US and other territories there are, however, numerous veterinary nutraceutical products available over the counter (OTC). The lack of clarity in the regulations governing veterinary hemp food supplements allows for products of questionable quality to flood the market,[61][62] which may pose a risk to the wellbeing of pets and owners.
To understand better the benefits of CBD and associated compounds for the quality of life of animals, companies specialized in CBD products for animals have been funding research projects.[63][64][65][66]
Canine osteoarthritis edit
CBD's ability to help regulate the endocannabinoid system[67][68][69] and reduce the release of excitatory neurotransmitters could result in a retrograde inhibitory signal that lessens chronic pain responses. Studies in dogs with chronic pain associated with osteoarthritis showed an increase in level of activity in animals receiving CBD-rich food supplements.[70][71][72][64]
Epilepsy edit
From the results seen in humans with drugs such as Epidiolex and Sativex in scientific studies and reviews,[73] it could be expected that CBD-based products would be helpful to manage seizures in dogs. However, despite the numerous case reports presented by veterinary neurologists supporting the benefits of CBD as adjunctive therapy, as of December 2020[update], published controlled studies have not shown a statistically significant decrease in the number of seizures across the groups receiving CBD.[63][65]
Pharmacokinetics edit
The oral bioavailability of CBD varies greatly across species and it is linked to the presentation and the time of administration.[74][66][75] A 24-hour kinetic examination in dogs showed that the absorption of the cannabidiolic acid (CBDA) does occur, and that this molecule is absorbed least twice as well as CBD post oral ingestion.[74][66][76]
It was found that the major metabolites of CBD in humans (7-OH-CBD and 7-COOH-CBD) are not prevalent in dogs, while 6-OH-CBD was found to be the primary metabolite in dogs receiving a CBD-enriched cannabis-derived herbal extract,[77] suggesting that canine and human CBD metabolic route might be somewhat different.[75]
Pharmacology edit
Possible mechanisms edit
As of 2024, the cellular effects and mechanisms of cannabidiol in vivo are unknown,[7][10] as research to date has been inconclusive and based on laboratory studies.[78] The anticonvulsant effects provided by cannabidiol (Epidiolex) in people with certain forms of epilepsy do not appear to involve cannabinoid receptors.[7] A possible mechanism for the effects of cannabidiol on seizures is by affecting the neuronal movement of calcium in brain structures involved in the excessive electrical activity of seizures.[10]
Pharmacodynamics edit
In vitro, cannabidiol has low affinity for, and acts as a negative allosteric modulator on the CB1 cannabinoid receptor[79][80]
Cannabidiol may be an antagonist of GPR55, a G protein-coupled receptor and putative cannabinoid receptor shown by in vitro studies to be widely distributed in the brain.[78][81][82] Cannabidiol may interact with various neurotransmitters, such as serotonin, dopamine, and GABA.[78][81][83]
Pharmacokinetics edit
The oral bioavailability of cannabidiol is approximately 6% in humans, while its bioavailability via inhalation is 11 to 45% (mean 31%).[11][12] The elimination half-life of cannabidiol in blood is 56 to 61 hours after oral doses twice per day over 7 days.[7] Cannabidiol is metabolized in the liver as well as in the intestines by cytochrome P450 enzymes.[7][81]
Pharmaceutical preparations edit
Nabiximols (brand name Sativex), an oromucosal spray made of a complex botanical mixture containing cannabidiol (CBD), delta-9-tetrahydrocannabinol (THC), and additional cannabinoid and non-cannabinoid constituents from cannabis sativa plants, was approved by Health Canada in 2005, to treat central neuropathic pain in multiple sclerosis, and in 2007, for cancer-related pain.[84] In New Zealand, Sativex is "approved for use as an add-on treatment for symptom improvement in people with moderate to severe spasticity due to multiple sclerosis who have not responded adequately to other anti-spasticity medication."[85]
Epidiolex (Epidyolex in Europe) is an orally administered cannabidiol solution.[7][10] It was approved in 2018 for treatment of two rare forms of childhood epilepsy, Lennox–Gastaut syndrome and Dravet syndrome, and seizures associated with tuberous sclerosis complex.[7][10] In the US, it is approved in these indications for people one year of age and older.[7]
Chemistry edit
At room temperature, cannabidiol is a colorless crystalline solid.[86] In strongly basic media and the presence of air, it is oxidized to a quinone.[87] Under acidic conditions it cyclizes to THC,[88] which also occurs during pyrolysis,[89] and during smoking.[24][90] The synthesis of cannabidiol has been accomplished by several research groups.[91][92][93]
Biosynthesis edit
Cannabis produces CBD through the same metabolic pathway as THC, until the next to last step, where CBDA synthase performs catalysis instead of THCA synthase.[96]
Isomerism edit
| Formal numbering | Terpenoid numbering | Number of stereoisomers | Natural occurrence | Convention on Psychotropic Substances Schedule | Structure | |||
|---|---|---|---|---|---|---|---|---|
| Short name | Chiral centers | Full name | Short name | Chiral centers | ||||
| Δ5-Cannabidiol | 1 and 3 | 2-(6-isopropenyl-3-methyl-5-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ4-Cannabidiol | 1 and 3 | 4 | No | Unscheduled | |
| Δ4-Cannabidiol | 1, 3 and 6 | 2-(6-isopropenyl-3-methyl-4-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ5-Cannabidiol | 1, 3 and 4 | 8 | No | Unscheduled | |
| Δ3-Cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methyl-3-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ6-Cannabidiol | 3 and 4 | 4 | ? | Unscheduled | |
| Δ3,7-Cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methylenecyclohex-1-yl)-5-pentyl-1,3-benzenediol | Δ1,7-Cannabidiol | 3 and 4 | 4 | No | Unscheduled | |
| Δ2-Cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methyl-2-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ1-Cannabidiol | 3 and 4 | 4 | Yes | Unscheduled | |
| Δ1-Cannabidiol | 3 and 6 | 2-(6-isopropenyl-3-methyl-1-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ2-Cannabidiol | 1 and 4 | 4 | No | Unscheduled | |
| Δ6-Cannabidiol | 3 | 2-(6-isopropenyl-3-methyl-6-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ3-Cannabidiol | 1 | 2 | No | Unscheduled | |
Pyrolysis edit
In the typical operating temperature range of e-cigarettes (250–400 °C (482–752 °F)), 25–52% of CBD is transformed into other chemical substances: Δ9-THC, Δ8-THC, cannabinol and cannabichromene as predominant pyrolysates. From a chemical point of view, CBD in e-cigarettes can be considered as a precursor of THC.[24]
Labeling and advertising edit
Studies conducted by the FDA from 2014 through 2019 have determined that a majority of CBD products are not accurately labeled with the amount of CBD they contain.[97] For example, a 2017 analysis of cannabidiol content in oil, tincture, or liquid vape products purchased online in the United States showed that 69% were mislabeled, with 43% having higher and 26% having lower content than stated on product labels.[98][52] In 2020, the FDA conducted a study of 147 CBD products and found that half contained THC.[97][99]
From 2015 to November 2022, the FDA issued dozens of warning letters to American manufacturers of CBD products for false advertising and illegal interstate marketing of CBD as an unapproved drug to treat diseases, such as cancer, osteoarthritis, symptoms of opioid withdrawal, Alzheimer's disease, and pet disorders.[53] Chemical analysis of CBD products found that many did not contain the levels of CBD claimed in advertising.[53]
In December 2020, the Federal Trade Commission initiated a law enforcement crackdown on American companies marketing CBD products as unapproved drugs.[100][101] The warning also applied to hemp CBD capsules and oil that were being marketed illegally while not adhering to the federal definition of a dietary supplement.[101]
History edit
Efforts to isolate the active ingredients in cannabis were made in the 19th century.[102] Cannabidiol was studied in 1940 from Minnesota wild hemp[102] and Egyptian Cannabis indica resin.[103][104] The chemical formula of CBD was proposed from a method for isolating it from wild hemp.[102] Its structure and stereochemistry were determined in 1963.[105]
Plant breeding edit
Selective breeding of cannabis plants has expanded and diversified as commercial and therapeutic markets develop. Some growers in the US succeeded in lowering the proportion of CBD-to-THC to accommodate customers who preferred varietals that were more mind-altering due to the higher THC and lower CBD content.[106] In the US, hemp is classified by the federal government as cannabis containing no more than 0.3% THC by dry weight. This classification was established in the 2018 Farm Bill and was refined to include hemp-sourced extracts, cannabinoids, and derivatives in the definition of hemp.[107]
Society and culture edit
Foods and beverages edit
Food and beverage products containing cannabidiol were widely marketed in the United States as early as 2017.[108] Hemp seed ingredients which do not naturally contain THC or CBD (but which may be contaminated with trace amounts on the outside during harvesting) were declared by the US Food and Drug Administration as generally recognized as safe (GRAS) in December 2018. CBD itself has not been declared GRAS, and under US federal law is illegal to sell as a food, dietary supplement, or animal feed.[49] State laws vary considerably as non-medical cannabis and derived products have been legalized in various jurisdictions.[109] Despite once having a promising market, the industry for CBD stalled out during the COVID-19 pandemic beginning in 2020, and, by 2024, it collapsed due to withdrawal of investors, the absence of a FDA ruling on efficacy and safety, inconsistent state-by-state legislation, and consumer ambivalence.[109]
Similar to energy drinks and protein bars which may contain vitamin or herbal additives, food and beverage items can be infused with CBD as an alternative means of ingesting the substance.[109] In the United States, numerous products are marketed as containing CBD, but in reality contain little or none.[49][110] Some companies marketing CBD-infused food products with claims that are similar to the effects of prescription drugs have received warning letters from the FDA for making unsubstantiated health claims.[49][111] In February 2019, the New York City Department of Health announced plans to fine restaurants that sell food or drinks containing CBD, beginning in October 2019.[112]
Sports edit
Cannabidiol has been used by professional and amateur athletes across disciplines and countries, with the World Anti-Doping Agency removing CBD from its banned substances list. The United States Anti-Doping Agency and United Kingdom-Anti-Doping Agency do not have anti-CBD policies, with the latter stating that, "CBD is not currently listed on the World Anti-Doping Agency Prohibited List. As a result, it is permitted to use in sport, though the intended benefits are unclear and not backed by clinical evidence. All other cannabinoids (including but not limited to cannabis, hashish, marijuana, and THC) are prohibited in-competition. The intention of the regulations is to prohibit cannabinoids that activate the same receptors in the brain as activated by THC."[113][114]
In 2019, the cannabis manufacturer Canopy Growth acquired majority ownership of BioSteel Sports Nutrition, which is developing CBD products under endorsement by numerous professional athletes.[115] The National Hockey League Alumni Association began a project with Canopy Growth to determine if CBD or other cannabis products might improve neurological symptoms and quality of life in head-injured players.[115] Some sports leagues have announced sponsorships with CBD companies, such as Major League Baseball (Charlotte's Web) and Ultimate Fighting Championship (Love Hemp).[116][117] Numerous professional athletes use CBD, primarily for treating pain.[115][118][119]
Legal status edit
Australia edit
Prescription medicine (Schedule 4) for therapeutic use containing two percent (2.0%) or less of other cannabinoids commonly found in cannabis (such as ∆9-THC). A Schedule 4 drug under the SUSMP is a Prescription Only Medicine, or Prescription Animal Remedy – Substances, the use or supply of which should be by or on the order of persons permitted by state or territory legislation to prescribe and should be available from a pharmacist on prescription.[120]
In June 2020, the Australian Therapeutic Goods Administration (TGA) published a consultation on a proposal to pave the way to make "low dose" CBD available to consumer/patients via pharmacists only through moving products from Schedule 4 to 3.[121] Any products sold would need to have their safety, quality and efficacy pre-assessed by the TGA and be formally approved for sale (details to be outlined by TGA). They would be made available to over 18s only, with the maximum daily dose of 60 mg/day, up to 2% THC finished product allowed, 30-day maximum supply, plant-derived or synthetic. This proposal is based on an initial literature review on the safety of low dose CBD published by the TGA in April 2020.[122] Epidyolex was approved for the adjunctive therapy of seizures associated with Lennox–Gastaut syndrome or with Dravet syndrome in September 2020.[2]
Bulgaria edit
In 2020, Bulgaria became the first country in the European Union to allow retail sales of food products and supplements containing CBD, despite the ongoing discussion within the EU about the classification of CBD as a novel food.[123] However, there exists a legal gap because of the lack of a legally-permissible minimum amount of THC in the products containing cannabinoids.[124]
Canada edit
In October 2018, cannabidiol became legal for recreational and medical use by the federal Cannabis Act.[125][126][127] As of August 2019[update], CBD products in Canada could only be sold by authorized retailers or federally licensed medical companies, limiting their access to the general public.[128] Nonetheless, with online delivery services and over 2,600 authorized cannabis retail stores as of October 2021[update], accessibility has steadily increased over time.[129][130] The Canadian government states that CBD products "are subject to all of the rules and requirements that apply to cannabis under the Cannabis Act and its regulations."[125] It requires "a processing licence to manufacture products containing CBD for sale, no matter what the source of the CBD is, and that CBD and products containing CBD, such as cannabis oil, may only be sold by an authorized retailer or licensed seller of medical CBD."[125] Edible CBD products were scheduled to be permitted for sale in Canada on October 17, 2019, for human consumption.[125]
As of August 2020[update], it was still illegal to carry cannabis and cannabis-derived products (including products containing CBD) across the Canadian border. If one carries any amount of cannabis for any purpose (including medical), it needs to be declared to the Canada Border Services Agency. Not declaring it is a serious criminal offence.[131]
Czech Republic edit
As of May 2023, the State Agricultural and Food Inspection of the Czech Republic is putting together broad regulations regarding a ban on CBD products.[132] They will make it illegal to sell products containing cannabidiol and other cannabinoids derived from hemp, as a result of EU Novel Food Regulation. In case of Czech Republic, European Industrial Hemp Association has submitted an official request to the Czech Republic to recognize natural hemp extracts with cannabinoids as traditional food.[133]
European Union edit
In 2019, the European Commission announced that CBD and other cannabinoids would be classified as "novel foods",[134] meaning that CBD products would require authorization under the EU Novel Food Regulation stating that because "this product was not used as a food or food ingredient before May 15, 1997, before it may be placed on the market in the EU as a food or food ingredient, a safety assessment under the Novel Food Regulation is required."[135] The recommendation – applying to CBD extracts, synthesized CBD, and all CBD products, including CBD oil – was scheduled for a final ruling by the European Commission in March 2019.[134] If approved, manufacturers of CBD products would be required to conduct safety tests and prove safe consumption, indicating that CBD products would not be eligible for legal commerce until at least 2021.[134] In December 2020, the European Commission concluded that CBD should not be considered as drug and can be qualified as food.[136]
Cannabidiol is listed in the EU Cosmetics Ingredient Database (CosIng).[137] However, the listing of an ingredient, assigned with an INCI name, in CosIng does not mean it is to be used in cosmetic products or is approved for such use.[137]
Several industrial hemp varieties can be legally cultivated in Western Europe. A variety such as "Fedora 17" has a cannabinoid profile consistently around 1%, with THC less than 0.3%.[138]
Hong Kong edit
In 2022, the HKSAR Government proposed a ban on any use of cannabidiol (including for academic research and by medical professionals) within the Hong Kong territory, making Hong Kong the first jurisdiction in the world to have complete prohibition of cannabidiol, starting from February 1, 2023,[139] in part due to the possible presence of THC which is illegal in Hong Kong, according to a research subsidized by the Hong Kong SAR Government.[140][141][142]
New Zealand edit
In 2017, the New Zealand government made changes to the regulations so that restrictions would be removed, which meant a doctor was able to prescribe cannabidiol to patients.[143]
The passing of the Misuse of Drugs (Medicinal Cannabis) Amendment Act in December 2018 means cannabidiol is no longer a controlled drug in New Zealand, but is a prescription medicine under the Medicines Act, with the restriction that "the tetrahydrocannabinols (THCs) and specified substances within the product must not exceed 2 percent of the total CBD, tetrahydrocannabinol (THC) and other specified substances."[144]
Russian Federation edit
According to a document received in response to an appeal to the Ministry of Internal Affairs of the Russian Federation, measures of state control in the Russian Federation regarding CBD have not been established. However, there is also a response from the Ministry of Health of the Russian Federation indicating that CBD can be considered as an isomer of restricted THC. The "isomer" argument is nonetheless vague, as progesterone, which is freely sold in pharmacies, is also an isomer of THC, all three being C
21H
30O
2.[145] On February 17, 2020, the deputy of the Moscow City Duma Darya Besedina sent an official request to the Prime Minister of the Russian Federation Mikhail Mishustin with a request to eliminate that legal ambiguity by publishing official explanations and, if necessary, making required changes in the corresponding government decree.[146]
Singapore edit
Singapore allows medical cannabis on a case-by-case basis, usually as a last resort drug. Each case is evaluated by the government, and largely comes in the form of Cannabidiol. However, the country is flexible to what is required for patient treatment, despite having some of the strictest drug laws in the world.
Sweden edit
Cannabidiol is classified as a medical product in Sweden.[147] However, in July 2019, Supreme Court of Sweden ruled that CBD oil with any concentration of THC falls under the narcotic control laws.[148]
Switzerland edit
While THC remains illegal, cannabidiol is not subject to the Swiss Narcotic Acts because it does not produce a comparable psychoactive effect.[149] Cannabis products containing less than 1% THC can be sold and purchased legally.[150][151]
Ukraine edit
On 7 April 2021 the Ukrainian government legalised use of isolated cannabidiol. Additionally, it approved Nabiximols, a cannabidiol-containing drug, for medical use.[152]
United Kingdom edit
Cannabidiol, in an oral-mucosal spray formulation combined with delta-9-tetrahydrocannabinol, is a product available by prescription for the relief of severe spasticity due to multiple sclerosis (where other anti-spasmodics have not been effective) in the United Kingdom.[153]
Until 2017, products containing cannabidiol marketed for medical purposes were classed as medicines by the UK regulatory body, the Medicines and Healthcare products Regulatory Agency (MHRA), and could not be marketed without regulatory approval for the medical claims.[154][155] As of 2018[update], cannabis oil is legal to possess, buy, and sell in the UK, providing the product does not contain more than 1 milligram of THC and is not advertised as providing a medicinal benefit.[9] Individual police officers and others who are ill-informed of the exact legislature pertaining to cannabidiol, however, may erroneously consider it of dubious legality, reflecting lack of awareness.[156]
In January 2019, the UK Food Standards Agency indicated it would regard CBD products, including CBD oil, as a novel food having no history of use before May 1997, and stated that such products must have authorisation and proven safety before being marketed.[134][157] The deadline for companies with existing products to submit a full and validated novel foods application with the FSA was 31 March 2021; failure to do so before this date would exclude those companies from selling CBD.[158] New products containing CBD after this deadline would require a fully approved application.[159]
In February 2020, the UK FSA advised vulnerable people, such as pregnant women, breastfeeding mothers, and those already taking medication for other medical concerns not to take CBD. The FSA further recommended that healthy adults should not consume more than 70 mg CBD per day.[158]
United Nations edit
Cannabidiol is scheduled under the Single Convention on Narcotic Drugs as cannabis. International Narcotics Control Board reminds Member States that, at the reconvened sixty-third session of the Commission on Narcotic Drugs, in December 2020, the States members of the Commission rejected the recommendation of WHO that a footnote be added to the entry for cannabis and cannabis resin in Schedule I of the 1961 Convention as amended to exempt from international control preparations containing predominantly CBD and not more than 0.2 per cent of delta-9-THC.[160]
United States edit
As of 2023[update], cannabidiol extracted from marijuana remains a Schedule I Controlled Substance,[49][161][162] and is not approved as a prescription drug or dietary supplement or allowed for interstate commerce in the United States.[48] CBD derived from hemp (with 0.3% THC or lower) is legal to sell as a cosmetics ingredient or for other purposes not regulated by the FDA, but cannot be sold under federal law as an ingredient in food, dietary supplement, or animal feed.[49][163] It is a common misconception that the legal ability to sell hemp (which may contain CBD), and hemp extracts and derivatives (including CBD), makes CBD legal for sale as a supplement or medicine.[163][164]
In September 2018, the GW Pharmaceuticals drug Epidiolex was placed in Schedule V of the Controlled Substances Act by the Drug Enforcement Administration (DEA),[165] following its approval by the FDA for rare types of childhood epilepsy.[7] It was then removed from the Controlled Substances Act by the DEA in April 2020.[166] Epidiolex is available for prescription use in all 50 states.[167]
In 2013, a CNN program that featured Charlotte's Web cannabis brought increased attention to the use of CBD for the treatment of seizure disorders in children.[168][169] A number of states passed laws over the next few years to allow the use of low-THC, high-CBD cannabis oil in such situations.[170] These states were in addition to the states that had already legalized cannabis for medical or recreational use.[170] Many states further relaxed their laws regarding CBD following the passage of the 2018 Farm Bill.[171][172]
The 2014 Farm Bill[173] legalized the sale of "non-viable hemp material" grown within states participating in the Hemp Pilot Program which defined hemp as cannabis containing less than 0.3% of THC.[174] The 2018 Farm Bill removed the hemp plant and all "derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a delta-9 tetrahydrocannabinol concentration of not more than 0.3 percent on a dry weight basis," including CBD, from the Controlled Substances Act, making them legal to manufacture in the United States.[25][175][176] The FDA retains regulatory authority over hemp-derived CBD,[164] while the DEA is not involved in the regulation of legally-compliant hemp and hemp products.[177] The 2018 Farm Bill requires that research and development of CBD for a therapeutic purpose would have to be conducted under notification and reporting to the FDA.[164][50]
See also edit
- Cannabidivarin (CBDV), a homolog of cannabidiol
- Hash oil
- Hemp oil
- List of investigational antipsychotics
- List of investigational analgesics
References edit
- ^ "cannabidiol (CHEBI:69478)". ebi.ac.uk. Archived from the original on May 12, 2021. Retrieved February 12, 2019.
- ^ a b "Epidyolex". Therapeutic Goods Administration (TGA). September 29, 2020. Archived from the original on October 30, 2021. Retrieved September 30, 2020.
- ^ "Federal Register :: Request Access". unblock.federalregister.gov. Retrieved March 10, 2024.
- ^ a b Itin C, Barasch D, Domb AJ, Hoffman A (May 2020). "Prolonged oral transmucosal delivery of highly lipophilic drug cannabidiol". International Journal of Pharmaceutics. 581: 119276. doi:10.1016/j.ijpharm.2020.119276. PMID 32243971. S2CID 214785913.
- ^ a b Itin C, Domb AJ, Hoffman A (October 2019). "A meta-opinion: cannabinoids delivered to oral mucosa by a spray for systemic absorption are rather ingested into gastro-intestinal tract: the influences of fed / fasting states". Expert Opinion on Drug Delivery. 16 (10): 1031–1035. doi:10.1080/17425247.2019.1653852. PMID 31393180. S2CID 199505274.
- ^ "Sativex (Cannabidiol/Tetrahydrocannabinol) Bayer Label" (PDF). bayer.ca. Archived (PDF) from the original on January 16, 2021. Retrieved June 28, 2018.
- ^ a b c d e f g h i j k l m n "Epidiolex – cannabidiol solution". DailyMed. August 26, 2020. Archived from the original on February 25, 2021. Retrieved September 11, 2020.
- ^ Anvisa (July 24, 2023). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published July 25, 2023). Archived from the original on August 27, 2023. Retrieved August 27, 2023.
- ^ a b Stallworth J. "Drug licensing factsheet: cannabis, CBD and other cannabinoids". The Home Office. Archived from the original on January 17, 2021. Retrieved December 10, 2020.
- ^ a b c d e f "Epidyolex EPAR". European Medicines Agency (EMA). June 24, 2019. Archived from the original on August 9, 2021. Retrieved September 11, 2020. Text was copied from this source which is copyrighted by the European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b Perucca E, Bialer M (June 5, 2020). "Critical Aspects Affecting Cannabidiol Oral Bioavailability and Metabolic Elimination, and Related Clinical Implications". CNS Drugs. 34 (8): 795–800. doi:10.1007/s40263-020-00741-5. PMID 32504461. S2CID 219313952.
- ^ a b Scuderi C, Filippis DD, Iuvone T, Blasio A, Steardo A, Esposito G (May 2009). "Cannabidiol in medicine: a review of its therapeutic potential in CNS disorders". Phytotherapy Research (Review). 23 (5): 597–602. doi:10.1002/ptr.2625. PMID 18844286. S2CID 21836765. Archived from the original on April 11, 2021. Retrieved May 22, 2020.
- ^ a b Devinsky O, Cilio MR, Cross H, Fernandez-Ruiz J, French J, Hill C, et al. (June 2014). "Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders". Epilepsia. 55 (6): 791–802. doi:10.1111/epi.12631. PMC 4707667. PMID 24854329.
- ^ a b Campos AC, Moreira FA, Gomes FV, Del Bel EA, Guimarães FS (December 2012). "Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences (Review). 367 (1607): 3364–3378. doi:10.1098/rstb.2011.0389. PMC 3481531. PMID 23108553.
- ^ a b Kirkland AE, Fadus MC, Gruber SA, Gray KM, Wilens TE, Squeglia LM (February 2022). "A scoping review of the use of cannabidiol in psychiatric disorders". Psychiatry Research. 308: 114347. doi:10.1016/j.psychres.2021.114347. PMC 8799523. PMID 34952255.
- ^ a b c d Black N, Stockings E, Campbell G, Tran LT, Zagic D, Hall WD, et al. (December 2019). "Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis". The Lancet. Psychiatry. 6 (12): 995–1010. doi:10.1016/S2215-0366(19)30401-8. PMC 6949116. PMID 31672337.
- ^ a b c d e f VanDolah HJ, Bauer BA, Mauck KF (September 2019). "Clinicians' Guide to Cannabidiol and Hemp Oils". Mayo Clinic Proceedings. 94 (9): 1840–1851. doi:10.1016/j.mayocp.2019.01.003. PMID 31447137.
- ^ Prud'homme M, Cata R, Jutras-Aswad D (January 2015). "Cannabidiol as an Intervention for Addictive Behaviors: A Systematic Review of the Evidence". Substance Abuse. 9: 33–38. doi:10.4137/SART.S25081. PMC 4444130. PMID 26056464.
- ^ a b Novella S (September 30, 2020). "Where Are We With CBD?". Science-Based Medicine. Archived from the original on August 12, 2021. Retrieved October 1, 2020.
- ^ a b c Pisanti S, Malfitano AM, Ciaglia E, Lamberti A, Ranieri R, Cuomo G, et al. (July 2017). "Cannabidiol: State of the art and new challenges for therapeutic applications". Pharmacology & Therapeutics. 175: 133–150. doi:10.1016/j.pharmthera.2017.02.041. PMID 28232276.
- ^ a b c Iseger TA, Bossong MG (March 2015). "A systematic review of the antipsychotic properties of cannabidiol in humans". Schizophrenia Research. 162 (1–3): 153–161. doi:10.1016/j.schres.2015.01.033. PMID 25667194. S2CID 3745655.
- ^ Boggs DL, Nguyen JD, Morgenson D, Taffe MA, Ranganathan M (January 2018). "Clinical and Preclinical Evidence for Functional Interactions of Cannabidiol and Δ9-Tetrahydrocannabinol". Neuropsychopharmacology. 43 (1): 142–154. doi:10.1038/npp.2017.209. PMC 5719112. PMID 28875990.
- ^ Aizpurua-Olaizola O, Soydaner U, Öztürk E, Schibano D, Simsir Y, Navarro P, et al. (February 2016). "Evolution of the Cannabinoid and Terpene Content during the Growth of Cannabis sativa Plants from Different Chemotypes". Journal of Natural Products. 79 (2): 324–331. doi:10.1021/acs.jnatprod.5b00949. hdl:1874/350973. PMID 26836472. Archived from the original on January 5, 2023. Retrieved November 27, 2022.
- ^ a b c Czégény Z, Nagy G, Babinszki B, Bajtel Á, Sebestyén Z, Kiss T, et al. (April 2021). "CBD, a precursor of THC in e-cigarettes". Scientific Reports. 11 (1): 8951. Bibcode:2021NatSR..11.8951C. doi:10.1038/s41598-021-88389-z. PMC 8076212. PMID 33903673.
- ^ a b Mead A (June 14, 2019). "Legal and Regulatory Issues Governing Cannabis and Cannabis-Derived Products in the United States". Frontiers in Plant Science. 10: 697. doi:10.3389/fpls.2019.00697. PMC 6590107. PMID 31263468.
- ^ "FDA Regulation of Cannabis and Cannabis-Derived Products, Including Cannabidiol (CBD). #2. How does the 2018 Farm Bill define hemp? What does it mean for FDA-regulated products?". US Food and Drug Administration (FDA). February 6, 2024. Retrieved February 6, 2024.
- ^ "International Nonproprietary Names for Pharmaceutical Substances (INN)" (PDF). WHO Drug Information. 30 (2): 241. 2016. Archived (PDF) from the original on February 5, 2018. Retrieved February 18, 2023.
- ^ a b Prud'homme M, Cata R, Jutras-Aswad D (2015). "Cannabidiol as an Intervention for Addictive Behaviors: A Systematic Review of the Evidence". Substance Abuse. 9: 33–38. doi:10.4137/SART.S25081. PMC 4444130. PMID 26056464.
- ^ Hoch E, Niemann D, von Keller R, Schneider M, Friemel CM, Preuss UW, et al. (February 2019). "How effective and safe is medical cannabis as a treatment of mental disorders? A systematic review". European Archives of Psychiatry and Clinical Neuroscience. 269 (1): 87–105. doi:10.1007/s00406-019-00984-4. PMC 6595000. PMID 30706168.
- ^ Sanders L (March 27, 2019). "The CBD boom is way ahead of the science". Science News. Retrieved April 24, 2023.
- ^ a b Office of the Commissioner (July 31, 2020). "FDA Approves New Indication for Drug Containing an Active Ingredient Derived from Cannabis to Treat Seizures in Rare Genetic Disease". FDA. Archived from the original on September 29, 2021. Retrieved November 25, 2020.
- ^ a b Stockings E, Zagic D, Campbell G, Weier M, Hall WD, Nielsen S, et al. (July 2018). "Evidence for cannabis and cannabinoids for epilepsy: a systematic review of controlled and observational evidence". Journal of Neurology, Neurosurgery, and Psychiatry. 89 (7): 741–753. doi:10.1136/jnnp-2017-317168. hdl:1959.4/unsworks_50076. PMID 29511052.
- ^ a b Elliott J, DeJean D, Clifford T, Coyle D, Potter BK, Skidmore B, et al. (February 2020). "Cannabis-based products for pediatric epilepsy: An updated systematic review". Seizure. 75: 18–22. doi:10.1016/j.seizure.2019.12.006. PMID 31865133. S2CID 208878465.
- ^ "Cannabis derivative may reduce seizures in some severe drug-resistant epilepsies, but adverse events increase". NIHR Evidence (Plain English summary). June 26, 2018. doi:10.3310/signal-000606. S2CID 242083755. Archived from the original on July 24, 2021. Retrieved March 15, 2022.
- ^ Office of the Commissioner (March 27, 2020). "FDA Approves First Drug Comprised of an Active Ingredient Derived from Marijuana to Treat Rare, Severe Forms of Epilepsy". FDA. Archived from the original on April 23, 2019. Retrieved May 28, 2021.
- ^ Silva TB, Balbino CQ, Weiber AF (May 2015). "The relationship between cannabidiol and psychosis: A review". Annals of Clinical Psychiatry. 27 (2): 134–141. PMID 25954940.
- ^ Blessing EM, Steenkamp MM, Manzanares J, Marmar CR (October 2015). "Cannabidiol as a Potential Treatment for Anxiety Disorders". Neurotherapeutics. 12 (4): 825–836. doi:10.1007/s13311-015-0387-1. PMC 4604171. PMID 26341731.
- ^ "What You Should Know About Using Cannabis, Including CBD, When Pregnant or Breastfeeding". US Food and Drug Administration (FDA). October 16, 2019. Archived from the original on October 17, 2019. Retrieved October 17, 2019.
- ^ Villanueva MR, Joshaghani N, Villa N, Badla O, Goit R, Saddik SE, et al. (July 2022). "Efficacy, Safety, and Regulation of Cannabidiol on Chronic Pain: A Systematic Review". Cureus. 14 (7): e26913. doi:10.7759/cureus.26913. PMC 9288157. PMID 35860716.
- ^ Kicman A, Toczek M (September 2020). "The Effects of Cannabidiol, a Non-Intoxicating Compound of Cannabis, on the Cardiovascular System in Health and Disease". International Journal of Molecular Sciences. 21 (18): 6740. doi:10.3390/ijms21186740. PMC 7554803. PMID 32937917.
- ^ Sachs J, McGlade E, Yurgelun-Todd D (October 2015). "Safety and Toxicology of Cannabinoids". Neurotherapeutics. 12 (4): 735–746. doi:10.1007/s13311-015-0380-8. PMC 4604177. PMID 26269228.
- ^ Izzo AA, Borrelli F, Capasso R, Di Marzo V, Mechoulam R (October 2009). "Non-psychotropic plant cannabinoids: new therapeutic opportunities from an ancient herb". Trends in Pharmacological Sciences. 30 (10): 515–527. doi:10.1016/j.tips.2009.07.006. PMID 19729208.
- ^ a b "Industrial hemp". Department of Agriculture, State of Colorado. 2018. Archived from the original on August 26, 2018. Retrieved September 14, 2018.
- ^ "Cannabinoid Clinical | Cannabinoids Research, Effects, and Uses". CannabinoidClinical.com. Archived from the original on June 26, 2021. Retrieved October 27, 2020.
- ^ Fischer B, Russell C, Sabioni P, van den Brink W, Le Foll B, Hall W, et al. (August 2017). "Lower-Risk Cannabis Use Guidelines: A Comprehensive Update of Evidence and Recommendations". American Journal of Public Health. 107 (8): e1–e12. doi:10.2105/AJPH.2017.303818. PMC 5508136. PMID 28644037.
- ^ Huestis MA, Solimini R, Pichini S, Pacifici R, Carlier J, Busardò FP (September 13, 2019). "Cannabidiol Adverse Effects and Toxicity". Current Neuropharmacology. 17 (10). Bentham Science Publishers Ltd.: 974–989. doi:10.2174/1570159x17666190603171901. PMC 7052834. PMID 31161980.
- ^ "Cannabidiol (CBD): MedlinePlus Supplements". medlineplus.gov. Archived from the original on October 20, 2021. Retrieved January 15, 2021.
- ^ a b "What You Need to Know (And What We're Working to Find Out) About Products Containing Cannabis or Cannabis-derived Compounds, Including CBD". US Food and Drug Administration. March 5, 2020. Archived from the original on October 22, 2021. Retrieved June 23, 2022.
- ^ a b c d e f "FDA Regulation of Cannabis and Cannabis-Derived Products, Including Cannabidiol (CBD)". US Food and Drug Administration (FDA). September 28, 2023. Retrieved October 29, 2023.
- ^ a b Hahn SM (March 5, 2020). "FDA Advances Work Related to Cannabidiol Products with Focus on Protecting Public Health, Providing Market Clarity". US Food and Drug Administration. Archived from the original on September 29, 2021. Retrieved March 6, 2020.
- ^ "Cannabidiol (CBD)". American Association of Poison Control Centers. September 30, 2019. Archived from the original on October 17, 2019. Retrieved October 17, 2019.
- ^ a b MacKeen D (October 16, 2019). "Scam or Not: What Are the Benefits of CBD?". The New York Times. Archived from the original on October 16, 2019. Retrieved October 17, 2019.
- ^ a b c "FDA Warns Companies for Illegally Selling Food and Beverage Products that Contain CBD". US Food and Drug Administration. November 21, 2022. Archived from the original on November 23, 2022. Retrieved November 23, 2022.
These companies are selling CBD containing products that people may confuse for traditional foods or beverages which may result in unintentional consumption or overconsumption of CBD. CBD-containing products in forms that are appealing to children, such as gummies, hard candies and cookies, are especially concerning.
- ^ Bornheim LM, Kim KY, Li J, Perotti BY, Benet LZ (August 1995). "Effect of cannabidiol pretreatment on the kinetics of tetrahydrocannabinol metabolites in mouse brain". Drug Metabolism and Disposition. 23 (8): 825–831. PMID 7493549.
- ^ Klein C, Karanges E, Spiro A, Wong A, Spencer J, Huynh T, et al. (November 2011). "Cannabidiol potentiates Δ9-tetrahydrocannabinol (THC) behavioural effects and alters THC pharmacokinetics during acute and chronic treatment in adolescent rats". Psychopharmacology. 218 (2): 443–457. doi:10.1007/s00213-011-2342-0. PMID 21667074. S2CID 6240926.
- ^ Ghovanloo MR, Shuart NG, Mezeyova J, Dean RA, Ruben PC, Goodchild SJ (October 2018). "Inhibitory effects of cannabidiol on voltage-dependent sodium currents". The Journal of Biological Chemistry. 293 (43): 16546–16558. doi:10.1074/jbc.RA118.004929. PMC 6204917. PMID 30219789.
- ^ Sait LG, Sula A, Ghovanloo MR, Hollingworth D, Ruben PC, Wallace BA (October 2020). "Cannabidiol interactions with voltage-gated sodium channels". eLife. 9. doi:10.7554/eLife.58593. PMC 7641581. PMID 33089780.
- ^ Nadulski T, Pragst F, Weinberg G, Roser P, Schnelle M, Fronk EM, et al. (December 2005). "Randomized, double-blind, placebo-controlled study about the effects of cannabidiol (CBD) on the pharmacokinetics of Delta9-tetrahydrocannabinol (THC) after oral application of THC verses standardized cannabis extract". Therapeutic Drug Monitoring. 27 (6): 799–810. doi:10.1097/01.ftd.0000177223.19294.5c. PMID 16306858. S2CID 12979224.
- ^ Lucas CJ, Galettis P, Schneider J (November 2018). "The pharmacokinetics and the pharmacodynamics of cannabinoids". British Journal of Clinical Pharmacology. 84 (11): 2477–2482. doi:10.1111/bcp.13710. PMC 6177698. PMID 30001569.
- ^ Yamaori S, Ebisawa J, Okushima Y, Yamamoto I, Watanabe K (April 2011). "Potent inhibition of human cytochrome P450 3A isoforms by cannabidiol: role of phenolic hydroxyl groups in the resorcinol moiety". Life Sciences. 88 (15–16): 730–736. doi:10.1016/j.lfs.2011.02.017. PMID 21356216.
- ^ "FDA warns company marketing unapproved cannabidiol products with unsubstantiated claims to treat cancer, Alzheimer's disease, opioid withdrawal, pain and pet anxiety". US Food and Drug Administration (FDA). July 23, 2019. Archived from the original on August 14, 2019. Retrieved July 24, 2019.
Unlike drugs approved by the FDA, the manufacturing process of these products has not been subject to FDA review as part of the drug approval process, and there has been no FDA evaluation of whether these products are effective for their intended use, what the proper dosage is, how they could interact with FDA-approved drugs, or whether they have dangerous side effects or other safety concerns.
- ^ Wakshlag JJ, Cital S, Eaton SJ, Prussin R, Hudalla C (April 15, 2020). "Cannabinoid, Terpene, and Heavy Metal Analysis of 29 Over-the-Counter Commercial Veterinary Hemp Supplements". Veterinary Medicine: Research and Reports. 11: 45–55. doi:10.2147/vmrr.s248712. PMC 7169471. PMID 32346530.
- ^ a b McGrath S, Bartner LR, Rao S, Packer RA, Gustafson DL (June 2019). "Randomized blinded controlled clinical trial to assess the effect of oral cannabidiol administration in addition to conventional antiepileptic treatment on seizure frequency in dogs with intractable idiopathic epilepsy". Journal of the American Veterinary Medical Association. 254 (11): 1301–1308. doi:10.2460/javma.254.11.1301. PMID 31067185. S2CID 148569810.
- ^ a b Verrico CD, Wesson S, Konduri V, Hofferek CJ, Vazquez-Perez J, Blair E, et al. (September 2020). "A randomized, double-blind, placebo-controlled study of daily cannabidiol for the treatment of canine osteoarthritis pain". Pain. 161 (9): 2191–2202. doi:10.1097/j.pain.0000000000001896. PMC 7584779. PMID 32345916.
- ^ a b Mogi C, Fukuyama T (2019). "Cannabidiol as a potential anti-epileptic dietary supplement in dogs with suspected epilepsy: three case reports". Pet Behaviour Science (7): 11–16. doi:10.21071/pbs.v0i7. ISSN 2445-2874. Archived from the original on September 3, 2020. Retrieved December 17, 2020.
- ^ a b c Wakshlag JJ, Schwark WS, Deabold KA, Talsma BN, Cital S, Lyubimov A, et al. (September 4, 2020). "Pharmacokinetics of Cannabidiol, Cannabidiolic Acid, Δ9-Tetrahydrocannabinol, Tetrahydrocannabinolic Acid and Related Metabolites in Canine Serum After Dosing With Three Oral Forms of Hemp Extract". Frontiers in Veterinary Science. 7: 505. doi:10.3389/fvets.2020.00505. PMC 7498943. PMID 33102539.
- ^ Ibrahim MM, Deng H, Zvonok A, Cockayne DA, Kwan J, Mata HP, et al. (September 2003). "Activation of CB2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: pain inhibition by receptors not present in the CNS". Proceedings of the National Academy of Sciences of the United States of America. 100 (18): 10529–10533. Bibcode:2003PNAS..10010529I. doi:10.1073/pnas.1834309100. PMC 193595. PMID 12917492.
- ^ Hohmann AG, Farthing JN, Zvonok AM, Makriyannis A (February 2004). "Selective activation of cannabinoid CB2 receptors suppresses hyperalgesia evoked by intradermal capsaicin". The Journal of Pharmacology and Experimental Therapeutics. 308 (2): 446–453. doi:10.1124/jpet.103.060079. PMID 14610224. S2CID 17663780.
- ^ Jung KM, Piomelli D (2015). "Cannabinoids and Endocannabinoids". In Pfaff DW, Volkow ND (eds.). Neuroscience in the 21st Century. New York: Springer New York. pp. 1–31. doi:10.1007/978-1-4614-6434-1_136-1. ISBN 978-1461464341.
- ^ Gamble LJ, Boesch JM, Frye CW, Schwark WS, Mann S, Wolfe L, et al. (July 23, 2018). "Pharmacokinetics, Safety, and Clinical Efficacy of Cannabidiol Treatment in Osteoarthritic Dogs". Frontiers in Veterinary Science. 5: 165. doi:10.3389/fvets.2018.00165. PMC 6065210. PMID 30083539.
- ^ Brioschi FA, Di Cesare F, Gioeni D, Rabbogliatti V, Ferrari F, D'Urso ES, et al. (August 2020). "Oral Transmucosal Cannabidiol Oil Formulation as Part of a Multimodal Analgesic Regimen: Effects on Pain Relief and Quality of Life Improvement in Dogs Affected by Spontaneous Osteoarthritis". Animals. 10 (9): 1505. doi:10.3390/ani10091505. PMC 7552307. PMID 32858828.
- ^ Kogan L, Hellyer P, Downing R (2020). "Hemp Oil Extract to Treat Canine Osteoarthritis-Related Pain: A Pilot Study". Journal of the American Holistic Veterinary Medical Association. 58: 35–45. Archived from the original on November 1, 2021. Retrieved December 17, 2020.
- ^ Russo E (2001). Handbook of psychotropic herbs: a scientific analysis of herbal remedies for psychiatric conditions. New York: Haworth Herbal Press. ISBN 0789007185. OCLC 43810871.
- ^ a b Deabold KA, Schwark WS, Wolf L, Wakshlag JJ (October 2019). "Single-Dose Pharmacokinetics and Preliminary Safety Assessment with Use of CBD-Rich Hemp Nutraceutical in Healthy Dogs and Cats". Animals. 9 (10): 832. doi:10.3390/ani9100832. PMC 6826847. PMID 31635105.
- ^ a b Vaughn D, Kulpa J, Paulionis L (February 11, 2020). "Preliminary Investigation of the Safety of Escalating Cannabinoid Doses in Healthy Dogs". Frontiers in Veterinary Science. 7: 51. doi:10.3389/fvets.2020.00051. PMC 7029731. PMID 32118071.
- ^ Della Rocca G, Di Salvo A (July 28, 2020). "Hemp in Veterinary Medicine: From Feed to Drug". Frontiers in Veterinary Science. 7: 387. doi:10.3389/fvets.2020.00387. PMC 7399642. PMID 32850997.
- ^ Chicoine A, Illing K, Vuong S, Pinto KR, Alcorn J, Cosford K (September 29, 2020). "Pharmacokinetic and Safety Evaluation of Various Oral Doses of a Novel 1:20 THC:CBD Cannabis Herbal Extract in Dogs". Frontiers in Veterinary Science. 7: 583404. doi:10.3389/fvets.2020.583404. PMC 7550466. PMID 33134364.
- ^ a b c Schouten M, Dalle S, Mantini D, Koppo K (2023). "Cannabidiol and brain function: current knowledge and future perspectives". Frontiers in Pharmacology. 14: 1328885. doi:10.3389/fphar.2023.1328885. PMC 10823027. PMID 38288087.
- ^ Laprairie RB, Bagher AM, Kelly ME, Denovan-Wright EM (2015). "Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor". British Journal of Pharmacology. 172 (20): 4790–4805. doi:10.1111/bph.13250. PMC 4621983. PMID 26218440.
- ^ Nguyen T, Li JX, Thomas BF, Wiley JL, Kenakin TP, Zhang Y (2016). "Allosteric Modulation: An Alternate Approach Targeting the Cannabinoid CB1 Receptor". Medicinal Research Reviews. 37 (3): 441–474. doi:10.1002/med.21418. PMC 5397374. PMID 27879006.
- ^ a b c "Cannabidiol". Drugs.com. July 14, 2023. Retrieved February 12, 2024.
- ^ Gray RA, Whalley BJ (January 2020). "The proposed mechanisms of action of CBD in epilepsy". Epileptic Disorders. 22 (S1): 10–15. doi:10.1684/epd.2020.1135. PMID 32053110.
- ^ Martínez-Aguirre C, et al. (December 2020). "Cannabidiol Acts at 5-HT1A Receptors in the Human Brain: Relevance for Treating Temporal Lobe Epilepsy". Front. Behav. Neurosci. 14: 611278. doi:10.3389/fnbeh.2020.611278. PMC 7770178. PMID 33384591.
- ^ Russo EB (February 2008). "Cannabinoids in the management of difficult to treat pain". Therapeutics and Clinical Risk Management. 4 (1): 245–259. doi:10.2147/TCRM.S1928. PMC 2503660. PMID 18728714.
- ^ "Sativex Oromucosal Spray". Medsafe, New Zealand Medicines and Medical Devices Safety Authority. December 19, 2018. Archived from the original on July 27, 2020. Retrieved April 3, 2019.
- ^ Jones PG, Falvello L, Kennard O, Sheldrick GM, Mechoulam R (1977). "Cannabidiol". Acta Crystallogr. B. 33 (10): 3211–3214. Bibcode:1977AcCrB..33.3211J. doi:10.1107/S0567740877010577.
- ^ Mechoulam R, Ben-Zvi Z, Gaoni Y (August 1968). "Hashish – 13. On the nature of the Beam test". Tetrahedron. 24 (16): 5615–5624. doi:10.1016/0040-4020(68)88159-1. PMID 5732891.
- ^ Gaoni Y, Mechoulam R (1966). "Hashish – VII The isomerization of cannabidiol to tetrahydrocannabinols". Tetrahedron. 22 (4): 1481–1488. doi:10.1016/S0040-4020(01)99446-3.
- ^ Küppers FJ, Bercht CA, Salemink CA, Lousberg RC, Terlouw JK, Heerma W (1975). "Cannabis – XV: Pyrolysis of cannabidiol. Structure elucidation of four pyrolytic products". Tetrahedron. 31 (13–14): 1513–1516. doi:10.1016/0040-4020(75)87002-5.
- ^ Quarles W, Ellman G, Jones R (1973). "Toxicology of marijuana: conditions for conversion of cannabidiol to THC upon smoking". Clinical Toxicology. 6 (2): 211–216. doi:10.3109/15563657308990520. PMID 4715204.
- ^ Petrzilka T, Haefliger W, Sikemeier C, Ohloff G, Eschenmoser A (March 1967). "[Synthesis and optical rotation of the (-)-cannabidiols]". Helvetica Chimica Acta. 50 (2): 719–723. doi:10.1002/hlca.19670500235. PMID 5587099.
- ^ Gaoni Y, Mechoulam R (1985). "Boron trifluoride etherate on alumuna – a modified Lewis acid reagent. An improved synthesis of cannabidiol". Tetrahedron Letters. 26 (8): 1083–1086. doi:10.1016/S0040-4039(00)98518-6.
- ^ Kobayashi Y, Takeuchi A, Wang YG (June 2006). "Synthesis of cannabidiols via alkenylation of cyclohexenyl monoacetate". Organic Letters. 8 (13): 2699–2702. doi:10.1021/ol060692h. PMID 16774235.
- ^ Gaoni Y, Mechoulam R (January 1966). "Hashish – VII: The isomerization of cannabidiol to tetrahydrocannabinols". Tetrahedron. 22 (4): 1481–1488. doi:10.1016/S0040-4020(01)99446-3.
- ^ Taura F, Sirikantaramas S, Shoyama Y, Yoshikai K, Shoyama Y, Morimoto S (June 2007). "Cannabidiolic-acid synthase, the chemotype-determining enzyme in the fiber-type Cannabis sativa". FEBS Letters. 581 (16): 2929–2934. doi:10.1016/j.febslet.2007.05.043. PMID 17544411. S2CID 20253070.
- ^ Marks MD, Tian L, Wenger JP, Omburo SN, Soto-Fuentes W, He J, et al. (2009). "Identification of candidate genes affecting Delta9-tetrahydrocannabinol biosynthesis in Cannabis sativa". Journal of Experimental Botany. 60 (13): 3715–3726. doi:10.1093/jxb/erp210. PMC 2736886. PMID 19581347.
- ^ a b "Sampling Study of the Current Cannabidiol Marketplace to Determine the Extent That Products are Mislabeled or Adulterated Report in Response to Further Consolidated Appropriations Act, 2020" (PDF). United States Food and Drug Administration. July 2020. Archived (PDF) from the original on June 23, 2021. Retrieved January 30, 2021.
- ^ Bonn-Miller MO, Loflin MJ, Thomas BF, Marcu JP, Hyke T, Vandrey R (November 2017). "Labeling Accuracy of Cannabidiol Extracts Sold Online". JAMA. 318 (17): 1708–1709. doi:10.1001/jama.2017.11909. PMC 5818782. PMID 29114823.
- ^ Evans DG (2020). "Medical Fraud, Mislabeling, Contamination: All Common in CBD Products". Missouri Medicine. 117 (5): 394–399. PMC 7723146. PMID 33311737.
- ^ "FTC Announces Crackdown on Deceptively Marketed CBD Products". US Federal Trade Commission (FTC). December 17, 2020. Archived from the original on June 22, 2021. Retrieved January 12, 2021.
- ^ a b Ashley DD, Engle MK (October 22, 2019). "Warning letter: Rooted Apothecary LLC". Office of Compliance, Center for Drug Evaluation and Research, US Food and Drug Administration; US Federal Trade Commission. Archived from the original on December 12, 2019. Retrieved October 23, 2019.
- ^ a b c Adams R, Hunt M, Clark JH (1940). "Structure of cannabidiol, a product isolated from the marihuana extract of Minnesota wild hemp". Journal of the American Chemical Society. 62 (1): 196–200. doi:10.1021/ja01858a058. ISSN 0002-7863.
- ^ Jacob A, Todd AR (1940). "Cannabidiol and cannabol, constituents of Cannabis indica resin". Nature. 145 (3670): 350. Bibcode:1940Natur.145..350J. doi:10.1038/145350a0. ISSN 0028-0836. S2CID 4106662.
- ^ Work TS, Bergel F, Todd AR (January 1939). "The active principles of Cannabis indica resin. I". The Biochemical Journal. 33 (1): 123–127. doi:10.1042/bj0330123. PMC 1264344. PMID 16746878.
- ^ Mechoulam R, Shvo Y (December 1963). "Hashish. I. The structure of cannabidiol". Tetrahedron. 19 (12): 2073–2078. doi:10.1016/0040-4020(63)85022-x. PMID 5879214.
- ^ Romney L (September 13, 2012). "On the frontier of medical pot to treat boy's epilepsy". Los Angeles Times. Archived from the original on October 21, 2018. Retrieved April 16, 2020.
- ^ "7 U.S. Code § 5940 – Legitimacy of industrial hemp research". LII / Legal Information Institute. Archived from the original on December 22, 2020. Retrieved November 27, 2018.
- ^ "Billboard featuring hemp leaf raises questions about new beverage for sale in Cincinnati | WLWT5". WLWT5. September 29, 2017. Archived from the original on December 26, 2020. Retrieved September 29, 2017.
- ^ a b c Creswell, Julie (February 28, 2024). "Companies were big on CBD. Not anymore". The New York Times. Retrieved March 25, 2024.
- ^ "Warning Letters and Test Results for Cannabidiol-Related Products". US Food and Drug Administration (FDA). November 2, 2017. Archived from the original on January 26, 2018. Retrieved January 2, 2018.
- ^ Fox A, Ravitz JR, Leongini EM, Malkin BJ (November 20, 2017). "Companies Marketing CBD Products Be Warned: FDA Is Watching". Lexology. Archived from the original on August 3, 2020. Retrieved December 14, 2017.
- ^ La Vito A, Franck T (February 15, 2019). "New York City plans to fine restaurants using CBD in food and drinks". CNBC. Archived from the original on November 7, 2020. Retrieved February 19, 2019.
- ^ "Athlete Advisory Note: Cannabidiol (CBD)". United Kingdom Anti-Doping Agency. Archived from the original on October 9, 2019. Retrieved October 9, 2019.
- ^ "Athletes: 6 things to know about cannabidiol". US Anti-Doping Agency. October 23, 2018. Archived from the original on October 11, 2021. Retrieved February 17, 2020.
- ^ a b c Donna S (October 2, 2019). "Canopy cannabis company buys ex-NHL player's sports nutrition business". CTV Business. The Canadian Press. Archived from the original on November 26, 2020. Retrieved February 17, 2020.
BioSteel's brand ambassadors also include well-known athletes across major sports leagues in North America, which could be beneficial as the company's attempt to push regulated CBD nutrition products into the mainstream health and wellness segments
- ^ "Major League Baseball, pioneering CBD brand Charlotte's Web strike groundbreaking deal" (Press release). Major League Baseball. October 12, 2022.
- ^ "UFC Names Love Hemp Official Global CBD Partner" (Press release). Ultimate Fighting Championship. March 18, 2021.
- ^ Loudin A (December 7, 2019). "As more pro athletes use cannabis for aches and pain, the more they run afoul of rules". The Washington Post. Archived from the original on November 24, 2020. Retrieved February 17, 2020.
- ^ Harrison S (September 28, 2019). "Lots of athletes say CBD Is a better painkiller. Is It?". Wired. Archived from the original on July 12, 2021. Retrieved February 17, 2020.
- ^ "Poisons Standard June 2017". Legislation.gov.au. Archived from the original on December 13, 2020. Retrieved December 4, 2016.
- ^ Australian Government Department of Health Therapeutic Goods Administration (April 24, 2020). "Consultation: Proposed amendments to the Poisons Standard – Joint ACMS/ACCS meetings, June 2020". Therapeutic Goods Administration (TGA). Archived from the original on May 11, 2021. Retrieved November 25, 2020.
- ^ "Safety of low dose cannabidiol" (PDF). Government of Australia. Therapeutic Goods Administration. April 2020. Archived (PDF) from the original on September 7, 2021. Retrieved November 25, 2020.
- ^ Hasse J. "This EU country has become the first to allow free sale Of CBD". Forbes. Archived from the original on October 6, 2021. Retrieved February 21, 2020.
- ^ Todorova SE (July 29, 2019). "Growing cannabis in Bulgaria: Legal but still stigmatised". Lexology. Archived from the original on April 11, 2021. Retrieved September 3, 2020.
- ^ a b c d "Cannabidiol (CBD)". Government of Canada. August 13, 2019. Archived from the original on September 18, 2019. Retrieved October 11, 2019.
- ^ "Health products containing cannabis or for use with cannabis: Guidance for the Cannabis Act, the Food and Drugs Act, and related regulations". Government of Canada. July 11, 2018. Archived from the original on October 19, 2018. Retrieved October 19, 2018.
- ^ "Cannabis Legalization and Regulation". Department of Justice, Electronic Communications. Government of Canada. June 20, 2018. Archived from the original on August 24, 2021. Retrieved November 24, 2018.
- ^ "'It should be available': Natural health food stores hope to cash in on CBD craze". CBC News. August 6, 2019. Archived from the original on October 12, 2019. Retrieved October 11, 2019.
- ^ Conway J (September 17, 2021). "Number of cannabis stores in Canada as of June 2021, by region". Statista. Archived from the original on May 2, 2022. Retrieved May 2, 2022.
- ^ Armstrong M (October 13, 2021). "Cannabis store openings in Canada only slightly affected the number of users". The Conversation. Archived from the original on May 2, 2022. Retrieved May 2, 2022.
- ^ Canada Health (June 20, 2018). "Cannabis and Canadian borders". aem. Archived from the original on August 14, 2021. Retrieved September 3, 2020.
- ^ "Ministerstvo zemědělství informuje o chystaném zákazu uvádění na trh produktů obsahujících kanabidiol (CBD) a jiné kanabinoidy (eAGRI)". eagri.cz (in Czech). Retrieved May 8, 2023.
- ^ Milosz F (May 6, 2023). "EIHA reacts about CBD and hemp extracts". CannabizEU. Archived from the original on May 6, 2023. Retrieved May 8, 2023.
- ^ a b c d Chu W (January 31, 2019). "Updated EC ruling for CBD classes supplement ingredient as Novel Food". NutraIngredients.com, William Reed Business Media Ltd. Archived from the original on November 1, 2021. Retrieved January 1, 2019.
- ^ "Cannabinoids, searched in the EU Novel food catalogue (v.1.1)". European Commission. January 1, 2019. Archived from the original on April 3, 2019. Retrieved February 1, 2019.
- ^ "European Commission reverses course, says CBD should not be regulated as a narcotic". Hemp Industry Daily. December 2, 2020. Archived from the original on October 21, 2021. Retrieved December 4, 2020.
- ^ a b "CosIng – Cosmetics – Cannabidiol". European Commission. Archived from the original on January 13, 2019. Retrieved December 4, 2016.
- ^ Fournier G, Beherec O, Bertucelli S (2003). "Intérêt du rapport Δ-9-THC / CBD dans le contrôle des cultures de chanvre industriel" [The advantage of the Δ-9-THC / CBD ratio in the control of industrial hemp crops]. Annales de Toxicologie Analytique (in French). 15 (4): 250–259. doi:10.1051/ata/2003003.
- ^ "Orders to amend Dangerous Drugs Ordinance and Control of Chemicals Ordinance to be gazetted on October 21 and cannabidiol to become dangerous drug". Hong Kong SAR Government. Archived from the original on October 20, 2022. Retrieved October 20, 2022.
- ^ "What you need to know about a proposed ban on CBD products in Hong Kong". South China Morning Post. June 11, 2022. Archived from the original on September 25, 2022. Retrieved September 25, 2022.
- ^ Leung H (August 21, 2022). "Hong Kong's zero-tolerance approach to drugs leaves budding CBD industry high and dry". Hong Kong Free Press HKFP. Archived from the original on April 20, 2023. Retrieved September 25, 2022.
- ^ Legislative Council Panel on Security (June 7, 2022). "Proposed Control of Cannabidiol through Legislation" (PDF). Government of Hong Kong. Archived (PDF) from the original on September 28, 2022. Retrieved October 24, 2022.
- ^ "Doctors now able to prescribe cannabidiol". radionz.co.nz. June 2, 2017. Archived from the original on June 1, 2017. Retrieved June 2, 2017.
- ^ "CBD products". www.health.govt.nz. Archived from the original on February 4, 2019. Retrieved March 10, 2019.
- ^ "Является ли вещество CBD (каннабидиол) разрешенным к использованию на территории России? – Правовед.RU". Archived from the original on November 1, 2021. Retrieved February 19, 2021.
- ^ "Журнал переписки депутата Бесединой". Archived from the original on February 19, 2021. Retrieved February 19, 2021.
- ^ "CBD products should follow the drug laws". Swedish Medical Products Agency. April 4, 2018. Archived from the original on July 24, 2018. Retrieved July 31, 2018.
- ^ Arnold M (July 30, 2019). "Sweden Joins Italy In Path To Defining CBD Oil Regulations". Cannabis Industry Journal. Archived from the original on November 8, 2020. Retrieved September 3, 2020.
- ^ "Cannabis: What is allowed, what is not allowed in Switzerland? – www.ch.ch". www.ch.ch. Archived from the original on January 21, 2021. Retrieved September 3, 2020.
- ^ Kohler S, Benz AS, Pruschy D, et al. (Vischer AG) (April 11, 2019). "Trends in the Swiss Cannabis Regulation | Lexology". www.lexology.com. Archived from the original on January 17, 2021. Retrieved September 3, 2020.
- ^ "Cannabis à faible teneur en THC et CBD" (in French). BAG.Admin.ch. Archived from the original on March 26, 2017. Retrieved May 20, 2017.
- ^ Cross D (April 9, 2021). "В Україні легалізували використання медичного канабісу, але не всього" [Ukraine has legalized the use of medical cannabis, but not all of it]. УП.Життя (UP.Life) (in Ukrainian). Archived from the original on April 9, 2021. Retrieved April 10, 2021.
- ^ "Sativex Oromucosal Spray – Summary of Product Characteristics (SmPC)". (emc). August 25, 2020. Archived from the original on February 4, 2021. Retrieved September 11, 2020.
- ^ "MHRA statement on products containing Cannabidiol (CBD)". Gov.uk. December 14, 2016. Archived from the original on May 10, 2019. Retrieved December 14, 2016.
- ^ "UK Classifies CBD Oil as a Medicinal Ingredient". BuyCBD.net. Archived from the original on November 1, 2021. Retrieved September 3, 2020.
- ^ Ukaegbu O, Smith J, Hall D, Frain T, Abbasian C. Staff awareness of the use of cannabidiol (CBD): a trust-wide survey study in the UK. Journal of Cannabis Research. 2021 Dec;3(1):1-0.
- ^ Gunn L, Haigh L (January 29, 2019). "British watchdog deems CBD a novel food, seeks to curtail sale on UK market". Nutrition Insight, CNS Media BV. Archived from the original on February 2, 2019. Retrieved January 1, 2019.
- ^ a b "Cannabidiol (CBD) guidance – Business guidance on cannabidiol (CBD) as a novel food". UK Food Standards Agency. September 24, 2020. Archived from the original on October 30, 2021. Retrieved December 8, 2020.
- ^ Marsat N (July 7, 2020). "CBD and Novel Foods regulation in the UK: regulation then and now". Health Europa. Archived from the original on October 6, 2020. Retrieved December 10, 2020.
- ^ "Reports published by the International Narcotics Control Board for 2021" (PDF). Archived (PDF) from the original on November 30, 2022. Retrieved December 1, 2022.
- ^ Mead A (May 2017). "The legal status of cannabis (marijuana) and cannabidiol (CBD) under US law". Epilepsy & Behavior. 70 (Pt B): 288–291. doi:10.1016/j.yebeh.2016.11.021. PMID 28169144.
- ^ Conaway KM (December 20, 2018). "Text – H.R.2 – 115th Congress (2017-2018): Agriculture Improvement Act of 2018". Congress.gov. Archived from the original on October 6, 2021. Retrieved May 1, 2019.
- ^ a b Bellamy J (May 9, 2019). "FDA: No CBD in dietary supplements or foods for now, but let's talk". Science-Based Medicine. Archived from the original on August 17, 2020. Retrieved June 15, 2019.
- ^ a b c Abernethy A, Schiller L (July 17, 2019). "FDA is Committed to Sound, Science-based Policy on CBD". US Food and Drug Administration (FDA). Archived from the original on November 14, 2019. Retrieved October 17, 2019.
- ^ "DEA reschedules Epidiolex, marijuana-derived drug, paving the way for it to hit the market". CNBC. September 27, 2018. Archived from the original on April 26, 2021. Retrieved October 3, 2018.
- ^ Wood S (April 6, 2020). "DEA relaxes rules for the only federally approved drug derived from marijuana". The Philadelphia Inquirer. Archived from the original on June 9, 2020. Retrieved April 23, 2023.
- ^ Tinker B (November 2, 2018). "First FDA-approved cannabis-based drug now available in the US". CNN. Retrieved April 23, 2023.
- ^ Maa E, Figi P (June 2014). "The case for medical marijuana in epilepsy". Epilepsia. 55 (6): 783–786. doi:10.1111/epi.12610. PMID 24854149. S2CID 4849160.
- ^ Saundra Y (August 7, 2013). "Marijuana stops child's severe seizures". CNN. Archived from the original on May 14, 2018. Retrieved May 14, 2018.
- ^ a b "State Medical Marijuana Laws". National Conference of State Legislatures. April 27, 2018. Archived from the original on December 11, 2018. Retrieved May 14, 2018.
- ^ Schuman B, Fisher J, Radke B, Faldetta G (October 22, 2020). "A Survey of State CBD & Hemp Regulation Since The 2018 Farm Bill". Cannabis Industry Journal. Retrieved April 24, 2023.
- ^ Gebhart F (September 13, 2022). "The Evolution of the CBD Regulatory Landscape". Drug Topics Journal. Retrieved April 24, 2023.
- ^ "The 2014 Farm Bill". thefarmbill.com. Archived from the original on October 30, 2021. Retrieved November 27, 2018.
- ^ Zhang M. "No, CBD Is Not 'Legal in All 50 States'". Forbes. Archived from the original on December 5, 2020. Retrieved November 27, 2018.
- ^ 7 U.S.C. § 1639o
- ^ 21 U.S.C. § 802
- ^ "DEA announces steps necessary to improve access to marijuana research". United States Drug Enforcement Administration. August 26, 2019. Archived from the original on October 18, 2019. Retrieved October 18, 2019.
Further reading edit
- Williams A (October 27, 2018). "Why Is CBD Everywhere?". The New York Times. ISSN 0362-4331. Archived from the original on October 22, 2021. Retrieved November 17, 2018.
- Dunning B (May 18, 2021). "Skeptoid #780: CBD for Everything: Cannabidiol is sold as an additive to just about every kind of product you can imagine. Why?". Skeptoid. Retrieved May 21, 2022.
External links edit
- "Cannabidiol". Drug Information Portal. U.S. National Library of Medicine.[failed verification]