Thiamine, also known as thiamin and vitamin B1, is a vitamin, an essential micronutrient for humans and animals.[3][4] It is found in food and commercially synthesized to be a dietary supplement or medication.[1][5] Phosphorylated forms of thiamine are required for some metabolic reactions, including the breakdown of glucose and amino acids.[1]
 | |
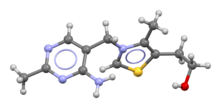 Skeletal formula and ball-and-stick model of the thiamine cation | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈθaɪ.əmɪn/ THY-ə-min |
| Other names | Vitamin B1, aneurine, thiamin |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | By mouth, intravenous, intramuscular[1] |
| Drug class | Vitamin |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 3.7% to 5.3% (Thiamine hydrochloride)[2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEBI |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| Chemical and physical data | |
| Formula | C12H17N4OS+ |
| Molar mass | 265.36 g·mol−1 |
| 3D model (JSmol) |
|
| |
| |
Food sources of thiamine include whole grains, legumes, and some meats and fish.[1][6] Grain processing removes much of the vitamin content, so in many countries cereals and flours are enriched with thiamine.[1] Supplements and medications are available to treat and prevent thiamine deficiency and the disorders that result from it such as beriberi and Wernicke encephalopathy. They are also used to treat maple syrup urine disease and Leigh syndrome. Supplements and medications are typically taken by mouth, but may also be given by intravenous or intramuscular injection.[7]
Thiamine supplements are generally well tolerated. Allergic reactions, including anaphylaxis, may occur when repeated doses are given by injection.[7][8] Thiamine is on the World Health Organization's List of Essential Medicines.[9] It is available as a generic medication, and in some countries as a non-prescription dietary supplement.[7]
Definition
editThiamine is one of the B vitamins and is also known as vitamin B1.[3][4] It is a cation that is usually supplied as a chloride salt. It is soluble in water, methanol and glycerol, but practically insoluble in less polar organic solvents.[10][11] In the body, thiamine can form derivatives; the most well-characterized of which is thiamine pyrophosphate (TPP), a coenzyme in the catabolism of sugars and amino acids.[3]
The chemical structure consists of an aminopyrimidine and a thiazolium ring linked by a methylene bridge. The thiazole is substituted with methyl and hydroxyethyl side chains. Thiamine is stable at acidic pH, but it is unstable in alkaline solutions and from exposure to heat.[10][11] It reacts strongly in Maillard-type reactions.[10] Oxidation yields the fluorescent derivative thiochrome, which can be used to determine the amount of the vitamin present in biological samples.[12]
Deficiency
editWell-known disorders caused by thiamine deficiency include beriberi, Wernicke–Korsakoff syndrome, optic neuropathy, Leigh's disease, African seasonal ataxia (or Nigerian seasonal ataxia), and central pontine myelinolysis.[13] Symptoms include malaise, weight loss, irritability and confusion.[10][14][15]
In Western countries, chronic alcoholism is a risk factor for deficiency. Also at risk are older adults, persons with HIV/AIDS or diabetes, and those who have had bariatric surgery.[1] Varying degrees of thiamine insufficiency have been associated with the long-term use of diuretics.[16][17]
Biological functions
editFive natural thiamine phosphate derivatives are known: thiamine monophosphate (ThMP), thiamine pyrophosphate (TPP), thiamine triphosphate (ThTP), adenosine thiamine diphosphate (AThDP) and adenosine thiamine triphosphate (AThTP). They are involved in many cellular processes.[18] The best-characterized form is TPP, a coenzyme in the catabolism of sugars and amino acids. While its role is well-known, the non-coenzyme action of thiamine and derivatives may be realized through binding to proteins which do not use that mechanism.[19] No physiological role is known for the monophosphate except as an intermediate in cellular conversion of thiamine to the di- and triphosphates.[20]
Thiamine pyrophosphate
editThiamine pyrophosphate (TPP), also called thiamine diphosphate (ThDP), participates as a coenzyme in metabolic reactions, including those in which polarity inversion takes place.[21] Its synthesis is catalyzed by the enzyme thiamine diphosphokinase according to the reaction thiamine + ATP → TPP + AMP (EC 2.7.6.2). TPP is a coenzyme for several enzymes that catalyze the transfer of two-carbon units and in particular the dehydrogenation (decarboxylation and subsequent conjugation with coenzyme A) of 2-oxoacids (alpha-keto acids). The mechanism of action of TPP as a coenzyme relies on its ability to form an ylide.[22] Examples include:
- Present in most species
- Present in some species:
- pyruvate decarboxylase (in yeast)
- several additional bacterial enzymes
The enzymes transketolase, pyruvate dehydrogenase (PDH), and 2-oxoglutarate dehydrogenase (OGDH) are important in carbohydrate metabolism. PDH links glycolysis to the citric acid cycle. OGDH catalyzes the overall conversion of 2-oxoglutarate (alpha-ketoglutarate) to succinyl-CoA and CO2 during the citric acid cycle. The reaction catalyzed by OGDH is a rate-limiting step in the citric acid cycle. The cytosolic enzyme transketolase is central to the pentose phosphate pathway, a major route for the biosynthesis of the pentose sugars deoxyribose and ribose. The mitochondrial PDH and OGDH are part of biochemical pathways that result in the generation of adenosine triphosphate (ATP), which is the main energy transfer molecule for the cell. In the nervous system, PDH is also involved in the synthesis of myelin and the neurotransmitter acetylcholine.[11]
Thiamine triphosphate
editThTP is implicated in chloride channel activation in the neurons of mammals and other animals, although its role is not well understood.[20] ThTP has been found in bacteria, fungi and plants, suggesting that it has other cellular roles.[23] In Escherichia coli, it is implicated in the response to amino acid starvation.[24]
Adenosine derivatives
editAThDP exists in small amounts in vertebrate liver, but its role remains unknown.[24]
AThTP is present in E. coli, where it accumulates as a result of carbon starvation. In this bacterium, AThTP may account for up to 20% of total thiamine. It also exists in lesser amounts in yeast, roots of higher plants and animal tissue.[24]
Medical uses
editDuring pregnancy, thiamine is sent to the fetus via the placenta. Pregnant women have a greater requirement for the vitamin than other adults, especially during the third trimester. Pregnant women with hyperemesis gravidarum are at an increased risk of thiamine deficiency due to losses when vomiting.[25] In lactating women, thiamine is delivered in breast milk even if it results in thiamine deficiency in the mother.[4][26]
Thiamine is important not only for mitochondrial membrane development, but also for synaptic membrane function.[27] It has also been suggested that a deficiency hinders brain development in infants and may be a cause of sudden infant death syndrome.[20]
Dietary recommendations
edit| US National Academy of Medicine | |
| Age group | RDA (mg/day) |
|---|---|
| Infants 0–6 months | 0.2* |
| Infants 6–12 months | 0.3* |
| 1–3 years | 0.5 |
| 4–8 years | 0.6 |
| 9–13 years | 0.9 |
| Females 14–18 years | 1.0 |
| Males 14+ years | 1.2 |
| Females 19+ years | 1.1 |
| Pregnant/lactating females 14–50 | 1.4 |
| * Adequate intake for infants, as an RDA has yet to be established[4] | |
| European Food Safety Authority | |
| Age group | Adequate intake (mg/MJ)[28] |
| All persons 7 months+ | 0.1 |
| Neither the US National Academy of Medicine nor the European Food Safety Authority have determined the tolerable upper intake level for thiamine[4] | |
The US National Academy of Medicine updated the Estimated Average Requirements (EARs) and Recommended Dietary Allowances (RDAs) for thiamine in 1998. The EARs for thiamine for women and men aged 14 and over are 0.9 mg/day and 1.1 mg/day, respectively; the RDAs are 1.1 and 1.2 mg/day, respectively. RDAs are higher than EARs to provide adequate intake levels for individuals with higher than average requirements. The RDA during pregnancy and for lactating females is 1.4 mg/day. For infants up to the age of 12 months, the Adequate Intake (AI) is 0.2–0.3 mg/day and for children aged 1–13 years the RDA increases with age from 0.5 to 0.9 mg/day.[4]
The European Food Safety Authority (EFSA) refers to the collective set of information as Dietary Reference Values, with Population Reference Intakes (PRIs) instead of RDAs, and Average Requirements instead of EARs. For women (including those pregnant or lactating), men and children the PRI is 0.1 mg thiamine per megajoule (MJ) of energy in their diet. As the conversion is 1 MJ = 239 kcal, an adult consuming 2390 kilocalories ought to be consuming 1.0 mg thiamine. This is slightly lower than the US RDA.[29]
Neither the National Academy of Medicine nor EFSA have set an upper intake level for thiamine, as there is no human data for adverse effects from high doses.[4][28]
Safety
editThiamine is generally well tolerated and non-toxic when administered orally.[7] There are rare reports of adverse side effects when thiamine is given intravenously, including allergic reactions, nausea, lethargy, and impaired coordination.[28][3]
Labeling
editFor US food and dietary supplement labeling purposes, the amount in a serving is expressed as a percent of Daily Value. Since May 27, 2016, the Daily Value has been 1.2 mg, in line with the RDA.[30][31]
Sources
editThiamine is found in a wide variety of processed and whole foods,[18] including lentils, peas, whole grains, pork, and nuts.[6][32] A typical daily prenatal vitamin product contains around 1.5 mg of thiamine.[33]
Food fortification
editSome countries require or recommend fortification of grain foods such as wheat, rice or maize (corn) because processing lowers vitamin content.[34] As of February 2022, 59 countries, mostly in North and Sub-Saharan Africa, require food fortification of wheat, rice or maize with thiamine or thiamine mononitrate. The amounts stipulated range from 2.0 to 10.0 mg/kg.[35] An additional 18 countries have a voluntary fortification program. For example, the Indian government recommends 3.5 mg/kg for "maida" (white) and "atta" (whole wheat) flour.[36]
Synthesis
editBiosynthesis
editThiamine biosynthesis occurs in bacteria, some protozoans, plants, and fungi.[37][38] The thiazole and pyrimidine moieties are biosynthesized separately and are then combined to form ThMP by the action of thiamine-phosphate synthase.
The pyrimidine ring system is formed in a reaction catalysed by phosphomethylpyrimidine synthase (ThiC), an enzyme in the radical SAM superfamily of iron–sulfur proteins, which use S-adenosyl methionine as a cofactor.[39][40]
The starting material is 5-aminoimidazole ribotide, which undergoes a rearrangement reaction via radical intermediates which incorporate the blue, green and red fragments shown into the product.[41][42]
The thiazole ring is formed in a reaction catalysed by thiazole synthase (EC 2.8.1.10).[39] The ultimate precursors are 1-deoxy-D-xylulose 5-phosphate, 2-iminoacetate and a sulfur carrier protein called ThiS. An additional protein, ThiG, is also required to bring together all the components of the ring at the enzyme active site.[43]
The final step to form ThMP involves decarboxylation of the thiazole intermediate, which reacts with the pyrophosphate derivative of phosphomethylpyrimidine, itself a product of a kinase, phosphomethylpyrimidine kinase.[39]
The biosynthetic pathways differ among organisms. In E. coli and other enterobacteriaceae, ThMP is phosphorylated to the cofactor TPP by a thiamine-phosphate kinase (ThMP + ATP → TPP + ADP).[39] In most bacteria and in eukaryotes, ThMP is hydrolyzed to thiamine and then pyrophosphorylated to TPP by thiamine diphosphokinase (thiamine + ATP → TPP + AMP).[44]
The biosynthetic pathways are regulated by riboswitches.[3] If there is sufficient thiamine present in the cell then the thiamine binds to the mRNAs for the enzymes that are required in the pathway and prevents their translation. If there is no thiamine present then there is no inhibition, and the enzymes required for the biosynthesis are produced. The specific riboswitch, the TPP riboswitch, is the only known riboswitch found in both eukaryotic and prokaryotic organisms.[45]
Laboratory synthesis
editIn the first total synthesis in 1936, ethyl 3-ethoxypropanoate was treated with ethyl formate to give an intermediate dicarbonyl compound which when reacted with acetamidine formed a substituted pyrimidine. Conversion of its hydroxyl group to an amino group was carried out by nucleophilic aromatic substitution, first to the chloride derivative using phosphorus oxychloride, followed by treatment with ammonia. The ethoxy group was then converted to a bromo derivative using hydrobromic acid. In the final stage, thiamine (as its dibromide salt) was formed in an alkylation reaction using 4-methyl-5-(2-hydroxyethyl)thiazole.[46]: 7 [47]
Industrial synthesis
editMerck & Co. adapted the 1936 laboratory-scale synthesis, allowing them to manufacture thiamine in Rahway in 1937.[47] However, an alternative route using the intermediate Grewe diamine (5-(aminomethyl)-2-methyl-4-pyrimidinamine), first published in 1937,[48] was investigated by Hoffman La Roche and competitive manufacturing processes followed. Efficient routes to the diamine have continued to be of interest.[47][49] In the European Economic Area, thiamine is registered under REACH regulation and between 100 and 1,000 tonnes per annum are manufactured or imported there.[50]
Synthetic analogues
editMany vitamin B1 analogues, such as Benfotiamine, fursultiamine, and sulbutiamine, are synthetic derivatives of thiamine. Most were developed in Japan in the 1950s and 1960s as forms that were intended to improve absorption compared to thiamine.[51] Some are approved for use in some countries as a drug or non-prescription dietary supplement for treatment of diabetic neuropathy or other health conditions.[52][53][54]
Absorption, metabolism and excretion
editIn the upper small intestine, thiamine phosphate esters present in food are hydrolyzed by alkaline phosphatase enzymes. At low concentrations, the absorption process is carrier-mediated. At higher concentrations, absorption also occurs via passive diffusion.[3] Active transport can be inhibited by alcohol consumption or by folate deficiency.[10]
The majority of thiamine in serum is bound to proteins, mainly albumin. Approximately 90% of total thiamine in blood is in erythrocytes. A specific binding protein called thiamine-binding protein has been identified in rat serum and is believed to be a hormone-regulated carrier protein important for tissue distribution of thiamine.[14] Uptake of thiamine by cells of the blood and other tissues occurs via active transport and passive diffusion.[10] Two members of the family of transporter proteins encoded by the genes SLC19A2 and SLC19A3 are capable of thiamine transport.[20] In some tissues, thiamine uptake and secretion appear to be mediated by a Na+-dependent transporter and a transcellular proton gradient.[14]
Human storage of thiamine is about 25 to 30 mg, with the greatest concentrations in skeletal muscle, heart, brain, liver, and kidneys. ThMP and free (unphosphorylated) thiamine are present in plasma, milk, cerebrospinal fluid, and, it is presumed, all extracellular fluid. Unlike the highly phosphorylated forms of thiamine, ThMP and free thiamine are capable of crossing cell membranes. Calcium and magnesium have been shown to affect the distribution of thiamine in the body and magnesium deficiency has been shown to aggravate thiamine deficiency.[20] Thiamine contents in human tissues are less than those of other species.[14][55]
Thiamine and its metabolites (2-methyl-4-amino-5-pyrimidine carboxylic acid, 4-methyl-thiazole-5-acetic acid, and others) are excreted principally in the urine.[3]
Interference
editThe bioavailability of thiamine in foods can be interfered with in a variety of ways. Sulfites, added to foods as a preservative,[56] will attack thiamine at the methylene bridge, cleaving the pyrimidine ring from the thiazole ring. The rate of this reaction is increased under acidic conditions.[14] Thiamine is degraded by thermolabile thiaminases present in some species of fish, shellfish and other foods.[10] The pupae of an African silk worm, Anaphe venata, is a traditional food in Nigeria. Consumption leads to thiamine deficiency.[57] Older literature reported that in Thailand, consumption of fermented, uncooked fish caused thiamine deficiency, but either abstaining from eating the fish or heating it first reversed the deficiency.[58] In ruminants, intestinal bacteria synthesize thiamine and thiaminases. The bacterial thiaminases are cell surface enzymes that must dissociate from the cell membrane before being activated; the dissociation can occur in ruminants under acidotic conditions. In dairy cows, over-feeding with grain causes subacute ruminal acidosis and increased ruminal bacteria thiaminase release, resulting in thiamine deficiency.[59]
From reports on two small studies conducted in Thailand, chewing slices of areca nut wrapped in betel leaves and chewing tea leaves reduced food thiamine bioavailability by a mechanism that may involve tannins.[58][60]
Bariatric surgery for weight loss is known to interfere with vitamin absorption.[61] A meta-analysis reported that 27% of people who underwent bariatric surgeries experience vitamin B1 deficiency.[62]
History
editThiamine was the first of the water-soluble vitamins to be isolated.[63] The earliest observations in humans and in chickens had shown that diets of primarily polished white rice caused beriberi, but did not attribute it to the absence of a previously unknown essential nutrient.[64][65]
In 1884, Takaki Kanehiro, a surgeon general in the Imperial Japanese Navy, rejected the previous germ theory for beriberi and suggested instead that the disease was due to insufficiencies in the diet.[64] Switching diets on a navy ship, he discovered that replacing a diet of white rice only with one also containing barley, meat, milk, bread, and vegetables, nearly eliminated beriberi on a nine-month sea voyage. However, Takaki had added many foods to the successful diet and he incorrectly attributed the benefit to increased protein intake, as vitamins were unknown at the time. The Navy was not convinced of the need for such an expensive program of dietary improvement, and many men continued to die of beriberi, even during the Russo-Japanese war of 1904–5. Not until 1905, after the anti-beriberi factor had been discovered in rice bran (removed by polishing into white rice) and in barley bran, was Takaki's experiment rewarded. He was made a baron in the Japanese peerage system, after which he was affectionately called "Barley Baron".[64]
The specific connection to grain was made in 1897 by Christiaan Eijkman, a military doctor in the Dutch East Indies, who discovered that fowl fed on a diet of cooked, polished rice developed paralysis that could be reversed by discontinuing rice polishing.[65] He attributed beriberi to the high levels of starch in rice being toxic. He believed that the toxicity was countered in a compound present in the rice polishings.[66] An associate, Gerrit Grijns, correctly interpreted the connection between excessive consumption of polished rice and beriberi in 1901: He concluded that rice contains an essential nutrient in the outer layers of the grain that is removed by polishing.[67] Eijkman was eventually awarded the Nobel Prize in Physiology and Medicine in 1929, because his observations led to the discovery of vitamins.
In 1910, a Japanese agricultural chemist of Tokyo Imperial University, Umetaro Suzuki, isolated a water-soluble thiamine compound from rice bran, which he named aberic acid. (He later renamed it Orizanin.) He described the compound as not only an anti-beriberi factor, but also as being essential to human nutrition; however, this finding failed to gain publicity outside of Japan, because a claim that the compound was a new finding was omitted in translation of his publication from Japanese to German.[63] In 1911 a Polish biochemist Casimir Funk isolated the antineuritic substance from rice bran (the modern thiamine) that he called a "vitamine" (on account of its containing an amino group).[68][69] However, Funk did not completely characterize its chemical structure. Dutch chemists, Barend Coenraad Petrus Jansen and his closest collaborator Willem Frederik Donath, went on to isolate and crystallize the active agent in 1926,[70] whose structure was determined by Robert Runnels Williams, in 1934. Thiamine was named by the Williams team as a portmanteau of "thio" (meaning sulfur-containing) and "vitamin". The term "vitamin" coming indirectly, by way of Funk, from the amine group of thiamine itself (although by this time, vitamins were known to not always be amines, for example, vitamin C). Thiamine was also synthesized by the Williams group in 1936.[71]
Sir Rudolph Peters, in Oxford, used pigeons to understand how thiamine deficiency results in the pathological-physiological symptoms of beriberi. Pigeons fed exclusively on polished rice developed opisthotonos, a condition characterized by head retraction. If not treated, the animals died after a few days. Administration of thiamine after opisthotonos was observed led to a complete cure within 30 minutes. As no morphological modifications were seen in the brain of the pigeons before and after treatment with thiamine, Peters introduced the concept of a biochemical-induced injury.[72] In 1937, Lohmann and Schuster showed that the diphosphorylated thiamine derivative, TPP, was a cofactor required for the oxidative decarboxylation of pyruvate.[73]
- Some contributors to the discovery of thiamine
References
edit- ^ a b c d e f "Thiamin Fact Sheets for Health Professionals". Office of Dietary Supplements. 11 February 2016. Archived from the original on 30 December 2016. Retrieved 30 December 2016.
- ^ Smithline HA, Donnino M, Greenblatt DJ (February 2012). "Pharmacokinetics of high-dose oral thiamine hydrochloride in healthy subjects". BMC Clinical Pharmacology. 12 (1): 4. doi:10.1186/1472-6904-12-4. PMC 3293077. PMID 22305197.
- ^ a b c d e f g Bettendorff L (2020). "Thiamine". In Marriott BP, Birt DF, Stallings VA, Yates AA (eds.). Present Knowledge in Nutrition, Eleventh Edition. London, United Kingdom: Academic Press (Elsevier). pp. 171–88. ISBN 978-0-323-66162-1.
- ^ a b c d e f g Institute of Medicine (1998). "Thiamin". Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: The National Academies Press. pp. 58–86. ISBN 978-0-309-06554-2. Archived from the original on 16 July 2015. Retrieved 29 August 2017.
- ^ "Thiamine: MedlinePlus Drug Information". medlineplus.gov. Archived from the original on 28 April 2018. Retrieved 30 April 2018.
- ^ a b "Thiamin". Micronutrient Information Center, Linus Pauling Institute, Oregon State University. 2013. Archived from the original on 2 February 2017. Retrieved 2 February 2022.
- ^ a b c d American Society of Health-System Pharmacists. "Thiamine Hydrochloride". Drugsite Trust (Drugs.com). Archived from the original on 9 August 2020. Retrieved 17 April 2018.
- ^ Kliegman RM, Stanton B (2016). Nelson Textbook of Pediatrics. Elsevier Health Sciences. p. 322. ISBN 9781455775668.
There are no cases of adverse effects of excess thiamine... A few isolated cases of puritis...
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ a b c d e f g Mahan LK, Escott-Stump S, eds. (2000). Krause's food, nutrition, & diet therapy (10th ed.). Philadelphia: W.B. Saunders Company. ISBN 978-0-7216-7904-4.
- ^ a b c Butterworth RF (2006). "Thiamin". In Shils ME, Shike M, Ross AC, Caballero B, Cousins RJ (eds.). Modern Nutrition in Health and Disease (10th ed.). Baltimore: Lippincott Williams & Wilkins.
- ^ Bettendorff L, Wins P (2013). "Biochemistry of Thiamine and Thiamine Phosphate Compounds". Encyclopedia of Biological Chemistry. pp. 202–9. doi:10.1016/B978-0-12-378630-2.00102-X. ISBN 9780123786319.
- ^ McCandless D (2010). Thiamine Deficiency and Associate Clinical Disorders. New York, NY: Humana Press. pp. 157–9. ISBN 978-1-60761-310-7.
- ^ a b c d e Combs Jr GF (2008). The Vitamins: Fundamental Aspects in Nutrition and Health (3rd ed.). Ithaca, NY: Elsevier Academic Press. ISBN 978-0-12-183493-7.
- ^ Smith TJ, Johnson CR, Koshy R, Hess SY, Qureshi UA, Mynak ML, et al. (August 2021). "Thiamine deficiency disorders: a clinical perspective". Annals of the New York Academy of Sciences. 1498 (1): 9–28. Bibcode:2021NYASA1498....9S. doi:10.1111/nyas.14536. PMC 8451766. PMID 33305487.
- ^ Katta N, Balla S, Alpert MA (July 2016). "Does Long-Term Furosemide Therapy Cause Thiamine Deficiency in Patients with Heart Failure? A Focused Review". The American Journal of Medicine. 129 (7): 753.e7–753.e11. doi:10.1016/j.amjmed.2016.01.037. PMID 26899752.
- ^ Gomes F, Bergeron G, Bourassa MW, Fischer PR (August 2021). "Thiamine deficiency unrelated to alcohol consumption in high-income countries: a literature review". Annals of the New York Academy of Sciences. 1498 (1): 46–56. Bibcode:2021NYASA1498...46G. doi:10.1111/nyas.14569. PMC 8451800. PMID 33576090.
- ^ a b Fitzpatrick TB, Chapman LM (August 2020). "The importance of thiamine (vitamin B1) in plant health: From crop yield to biofortification". The Journal of Biological Chemistry. 295 (34): 12002–13. doi:10.1074/jbc.REV120.010918. PMC 7443482. PMID 32554808.
- ^ Mkrtchyan G, Aleshin V, Parkhomenko Y, Kaehne T, Di Salvo ML, Parroni A, et al. (July 2015). "Molecular mechanisms of the non-coenzyme action of thiamin in brain: biochemical, structural and pathway analysis". Scientific Reports. 5: 12583. Bibcode:2015NatSR...512583M. doi:10.1038/srep12583. PMC 4515825. PMID 26212886.
- ^ a b c d e Lonsdale D (March 2006). "A review of the biochemistry, metabolism and clinical benefits of thiamin(e) and its derivatives". Evidence-Based Complementary and Alternative Medicine. 3 (1): 49–59. doi:10.1093/ecam/nek009. PMC 1375232. PMID 16550223.
- ^ Boluda CJ, Juncá C, Soto E, de la Cruz D, Peña A (13 December 2019). "Umpolung in reactions catalyzed by thiamine pyrophosphate dependent enzymes". Ciencia, Ambiente y Clima (in Spanish). 2 (2): 27–42. doi:10.22206/cac.2019.v2i2.pp27-42. ISSN 2636-2333. S2CID 213836801. Archived from the original on 1 December 2022. Retrieved 1 December 2022.
- ^ Ciszak EM, Korotchkina LG, Dominiak PM, Sidhu S, Patel MS (June 2003). "Structural basis for flip-flop action of thiamin pyrophosphate-dependent enzymes revealed by human pyruvate dehydrogenase". The Journal of Biological Chemistry. 278 (23): 21240–21246. doi:10.1074/jbc.M300339200. hdl:2060/20030106063. PMID 12651851.
- ^ Makarchikov AF, Lakaye B, Gulyai IE, Czerniecki J, Coumans B, Wins P, et al. (July 2003). "Thiamine triphosphate and thiamine triphosphatase activities: from bacteria to mammals". Cellular and Molecular Life Sciences. 60 (7): 1477–88. doi:10.1007/s00018-003-3098-4. PMC 11146050. PMID 12943234. S2CID 25400487.
- ^ a b c Bettendorff L (November 2021). "Update on Thiamine Triphosphorylated Derivatives and Metabolizing Enzymatic Complexes". Biomolecules. 11 (11): 1645. doi:10.3390/biom11111645. PMC 8615392. PMID 34827643.
- ^ Oudman E, Wijnia JW, Oey M, van Dam M, Painter RC, Postma A (May 2019). "Wernicke's encephalopathy in hyperemesis gravidarum: A systematic review". European Journal of Obstetrics, Gynecology, and Reproductive Biology. 236: 84–93. doi:10.1016/j.ejogrb.2019.03.006. hdl:1874/379566. PMID 30889425. S2CID 84184482.
- ^ Butterworth RF (December 2001). "Maternal thiamine deficiency: still a problem in some world communities". The American Journal of Clinical Nutrition. 74 (6): 712–3. doi:10.1093/ajcn/74.6.712. PMID 11722950.
- ^ Kloss O, Eskin NA, Suh M (April 2018). "Thiamin deficiency on fetal brain development with and without prenatal alcohol exposure". Biochemistry and Cell Biology. 96 (2): 169–77. doi:10.1139/bcb-2017-0082. hdl:1807/87775. PMID 28915355.
- ^ a b c Tolerable Upper Intake Levels For Vitamins And Minerals (PDF), European Food Safety Authority, 2006, archived (PDF) from the original on 16 March 2016
- ^ "Overview on Dietary Reference Values for the EU population as derived by the EFSA Panel on Dietetic Products, Nutrition and Allergies" (PDF). 2017. Archived (PDF) from the original on 28 August 2017.
- ^ "Federal Register May 27, 2016 Food Labeling: Revision of the Nutrition and Supplement Facts Labels. FR page 33982" (PDF). Archived (PDF) from the original on 8 August 2016.
- ^ "Daily Value Reference of the Dietary Supplement Label Database (DSLD)". Dietary Supplement Label Database (DSLD). Archived from the original on 7 April 2020. Retrieved 6 February 2022.
- ^ "Thiamin content per 100 grams; select food subset, abridged list by food groups". United States Department of Agriculture, Agricultural Research Service, USDA Branded Food Products Database v.3.6.4.1. 17 January 2017. Archived from the original on 2 February 2017. Retrieved 27 January 2017.
- ^ Kominiarek MA, Rajan P (November 2016). "Nutrition Recommendations in Pregnancy and Lactation". The Medical Clinics of North America. 100 (6): 1199–215. doi:10.1016/j.mcna.2016.06.004. PMC 5104202. PMID 27745590.
- ^ "What nutrients are added to flour and rice in fortification?". Food Fortification Initiative. 2021. Archived from the original on 8 October 2021. Retrieved 8 October 2021.
- ^ "Map: Count of Nutrients In Fortification Standards". Global Fortification Data Exchange. Archived from the original on 11 April 2019. Retrieved 11 October 2021.
- ^ "Direction under Section 16(5) of Foods Safety and Standards Act, 2006 regarding Operationalisation of Food Safety & Standards (Fortification of Foods) Regulations, 2017 relating to standards for fortification of food" (PDF). Food Safety & Standards Authority of India (FSSAI). 19 May 2017. Archived (PDF) from the original on 17 December 2021. Retrieved 1 February 2022.
- ^ Webb ME, Marquet A, Mendel RR, Rébeillé F, Smith AG (October 2007). "Elucidating biosynthetic pathways for vitamins and cofactors". Natural Product Reports. 24 (5): 988–1008. doi:10.1039/b703105j. PMID 17898894.
- ^ Begley TP, Chatterjee A, Hanes JW, Hazra A, Ealick SE (April 2008). "Cofactor biosynthesis--still yielding fascinating new biological chemistry". Current Opinion in Chemical Biology. 12 (2): 118–25. doi:10.1016/j.cbpa.2008.02.006. PMC 2677635. PMID 18314013.
- ^ a b c d Caspi R (14 September 2011). "Pathway: superpathway of thiamine diphosphate biosynthesis I". MetaCyc Metabolic Pathway Database. Archived from the original on 1 February 2022. Retrieved 1 February 2022.
- ^ Holliday GL, Akiva E, Meng EC, Brown SD, Calhoun S, Pieper U, et al. (2018). "Atlas of the Radical SAM Superfamily: Divergent Evolution of Function Using a "Plug and Play" Domain". Radical SAM Enzymes. Methods in Enzymology. Vol. 606. pp. 1–71. doi:10.1016/bs.mie.2018.06.004. ISBN 9780128127940. PMC 6445391. PMID 30097089.
- ^ Chatterjee A, Hazra AB, Abdelwahed S, Hilmey DG, Begley TP (November 2010). "A "radical dance" in thiamin biosynthesis: mechanistic analysis of the bacterial hydroxymethylpyrimidine phosphate synthase". Angewandte Chemie. 49 (46): 8653–6. doi:10.1002/anie.201003419. PMC 3147014. PMID 20886485.
- ^ Mehta AP, Abdelwahed SH, Fenwick MK, Hazra AB, Taga ME, Zhang Y, et al. (August 2015). "Anaerobic 5-Hydroxybenzimidazole Formation from Aminoimidazole Ribotide: An Unanticipated Intersection of Thiamin and Vitamin B₁₂ Biosynthesis". Journal of the American Chemical Society. 137 (33): 10444–7. doi:10.1021/jacs.5b03576. PMC 4753784. PMID 26237670.
- ^ Begley TP (February 2006). "Cofactor biosynthesis: an organic chemist's treasure trove". Natural Product Reports. 23 (1): 15–25. doi:10.1039/b207131m. PMID 16453030.
- ^ Caspi R (23 September 2011). "Pathway: superpathway of thiamine diphosphate biosynthesis III (eukaryotes)". MetaCyc Metabolic Pathway Database. Archived from the original on 14 November 2022. Retrieved 14 November 2022.
- ^ Bocobza SE, Aharoni A (October 2008). "Switching the light on plant riboswitches". Trends in Plant Science. 13 (10): 526–33. Bibcode:2008TPS....13..526B. doi:10.1016/j.tplants.2008.07.004. PMID 18778966.
- ^ Tylicki A, Łotowski Z, Siemieniuk M, Ratkiewicz A (February 2018). "Thiamine and selected thiamine antivitamins - biological activity and methods of synthesis". Bioscience Reports. 38 (1). doi:10.1042/BSR20171148. PMC 6435462. PMID 29208764.
- ^ a b c Eggersdorfer M, Laudert D, Létinois U, McClymont T, Medlock J, Netscher T, et al. (December 2012). "One hundred years of vitamins-a success story of the natural sciences". Angewandte Chemie. 51 (52): 12960–90. doi:10.1002/anie.201205886. PMID 23208776.
- ^ Todd AR, Bergel F (1937). "73. Aneurin. Part VII. A synthesis of aneurin". Journal of the Chemical Society (Resumed): 364. doi:10.1039/JR9370000364.
- ^ Jiang M, Liu M, Huang H, Chen F (2021). "Fully Continuous Flow Synthesis of 5-(Aminomethyl)-2-methylpyrimidin-4-amine: A Key Intermediate of Vitamin B1". Organic Process Research & Development. 25 (10): 2331–7. doi:10.1021/acs.oprd.1c00253. S2CID 242772232.
- ^ "Substance Infocard". echa.europa.eu. Archived from the original on 20 April 2021. Retrieved 11 May 2022.
- ^ Bettendorff L (2014). "Chapter 7 - Thiamine". In Zempleni J, Suttie JW, Gregory JF, Stover PJ (eds.). Handbook of vitamins (5th ed.). Hoboken: CRC Press. pp. 267–324. ISBN 9781466515574.
- ^ Zaheer A, Zaheer F, Saeed H, Tahir Z, Tahir MW (April 2021). "A Review of Alternative Treatment Options in Diabetic Polyneuropathy". Cureus. 13 (4). e14600. doi:10.7759/cureus.14600. PMC 8139599. PMID 34040901.
- ^ McCarty MF, Inoguchi T (2008). "11. Targeting Oxidant Stress as a Strategy for Preventing Vascular Complications of Diabetes and Metabolic Syndrome". In Pasupuleti VK, Anderson JW (eds.). Nutraceuticals, glycemic health and type 2 diabetes (1st ed.). Ames, Iowa: Wiley-Blackwell/IFT Press. p. 213. ISBN 9780813804286.
- ^ Lonsdale D (September 2004). "Thiamine tetrahydrofurfuryl disulfide: a little known therapeutic agent". Medical Science Monitor. 10 (9): RA199–203. PMID 15328496. Archived from the original on 25 September 2012. Retrieved 17 July 2022.
- ^ Bettendorff L, Mastrogiacomo F, Kish SJ, Grisar T (January 1996). "Thiamine, thiamine phosphates, and their metabolizing enzymes in human brain". Journal of Neurochemistry. 66 (1): 250–8. doi:10.1046/j.1471-4159.1996.66010250.x. PMID 8522961. S2CID 7161882.
- ^ McGuire M, Beerman KA (2007). Nutritional Sciences: From Fundamentals to Foods. California: Thomas Wadsworth.
- ^ Nishimune T, Watanabe Y, Okazaki H, Akai H (2000). "Thiamin is decomposed due to Anaphe spp. entomophagy in seasonal ataxia patients in Nigeria". J. Nutr. 130 (6): 1625–8. doi:10.1093/jn/130.6.1625. PMID 10827220.
- ^ a b Vimokesant SL, Hilker DM, Nakornchai S, Rungruangsak K, Dhanamitta S (December 1975). "Effects of betel nut and fermented fish on the thiamin status of northeastern Thais". Am J Clin Nutr. 28 (12): 1458–63. doi:10.1093/ajcn/28.12.1458. PMID 803009.
- ^ Pan X, Nan X, Yang L, Jiang L, Xiong B (September 2018). "Thiamine status, metabolism and application in dairy cows: a review". Br J Nutr. 120 (5): 491–9. doi:10.1017/S0007114518001666. PMID 29986774. S2CID 51606809.
- ^ Vimokesant S, Kunjara S, Rungruangsak K, Nakornchai S, Panijpan B (1982). "Beriberi caused by antithiamin factors in food and its prevention". Ann N Y Acad Sci. 378 (1): 123–36. Bibcode:1982NYASA.378..123V. doi:10.1111/j.1749-6632.1982.tb31191.x. PMID 7044221. S2CID 40854060.
- ^ Nunes R, Santos-Sousa H, Vieira S, Nogueiro J, Bouça-Machado R, Pereira A, et al. (March 2022). "Vitamin B Complex Deficiency After Roux-en-Y Gastric Bypass and Sleeve Gastrectomy-a Systematic Review and Meta-Analysis". Obes Surg. 32 (3): 873–91. doi:10.1007/s11695-021-05783-2. PMID 34982396. S2CID 245655046.
- ^ Bahardoust M, Eghbali F, Shahmiri SS, Alijanpour A, Yarigholi F, Valizadeh R, et al. (September 2022). "B1 Vitamin Deficiency After Bariatric Surgery, Prevalence, and Symptoms: a Systematic Review and Meta-analysis". Obes Surg. 32 (9): 3104–12. doi:10.1007/s11695-022-06178-7. PMID 35776243. S2CID 250149680.
- ^ a b Suzuki U, Shimamura T (1911). "Active constituent of rice grits preventing bird polyneuritis". Tokyo Kagaku Kaishi. 32: 4–7, 144–6, 335–58. doi:10.1246/nikkashi1880.32.4. Archived from the original on 21 June 2020. Retrieved 2 May 2018.
- ^ a b c McCollum EV (1957). A History of Nutrition. Cambridge, Massachusetts: Riverside Press, Houghton Mifflin.
- ^ a b Eijkman C (1897). "Eine Beriberiähnliche Krankheit der Hühner" [A disease of chickens which is similar to beriberi]. Archiv für Pathologische Anatomie und Physiologie und für Klinische Medicin. 148 (3): 523–532. doi:10.1007/BF01937576. S2CID 38445999. Archived from the original on 9 August 2020. Retrieved 4 July 2019.
- ^ "The Nobel Prize and the Discovery of Vitamins". nobelprize.org. Archived from the original on 16 January 2018. Retrieved 1 May 2018.
- ^ Grijns G (1901). "Over polyneuritis gallinarum" [On polyneuritis gallinarum]. Geneeskundig Tijdschrift voor Nederlandsch-Indië (Medical Journal for the Dutch East Indies). 41 (1): 3–11. Archived from the original on 29 August 2021. Retrieved 5 February 2020.
- ^ Funk C (December 1911). "On the chemical nature of the substance which cures polyneuritis in birds induced by a diet of polished rice". The Journal of Physiology. 43 (5): 395–400. doi:10.1113/jphysiol.1911.sp001481. PMC 1512869. PMID 16993097.
- ^ Funk C (1912). "The etiology of the deficiency diseases. Beri-beri, polyneuritis in birds, epidemic dropsy, scurvy, experimental scurvy in animals, infantile scurvy, ship beri-beri, pellagra". Journal of State Medicine. 20: 341–68. Archived from the original on 4 July 2020. Retrieved 5 February 2020. The word "vitamine" is coined on p. 342: "It is now known that all these diseases, with the exception of pellagra, can be prevented and cured by the addition of certain preventative substances; the deficient substances, which are of the nature of organic bases, we will call "vitamines"; and we will speak of a beri-beri or scurvy vitamine, which means a substance preventing the special disease."
- ^ Jansen BC, Donath WF (1926). "On the isolation of antiberiberi vitamin". Proc. Kon. Ned. Akad. Wet. 29: 1390–400.
- ^ Williams RR, Cline JK (1936). "Synthesis of Vitamin B1". Journal of the American Chemical Society. 58 (8): 1504–5. doi:10.1021/ja01299a505.
- ^ Peters RA (1936). "The biochemical lesion in vitamin B1 deficiency. Application of modern biochemical analysis in its diagnosis". Lancet. 230 (5882): 1161–4. doi:10.1016/S0140-6736(01)28025-8.
- ^ Lohmann K, Schuster P (1937). "Untersuchungen über die Cocarboxylase". Biochem. Z. 294: 188–214.