The nucleoid (meaning nucleus-like) is an irregularly shaped region within the prokaryotic cell that contains all or most of the genetic material.[1][2][3] The chromosome of a typical prokaryote is circular, and its length is very large compared to the cell dimensions, so it needs to be compacted in order to fit. In contrast to the nucleus of a eukaryotic cell, it is not surrounded by a nuclear membrane. Instead, the nucleoid forms by condensation and functional arrangement with the help of chromosomal architectural proteins and RNA molecules as well as DNA supercoiling. The length of a genome widely varies (generally at least a few million base pairs) and a cell may contain multiple copies of it.
There is not yet a high-resolution structure known of a bacterial nucleoid, however key features have been researched in Escherichia coli as a model organism. In E. coli, the chromosomal DNA is on average negatively supercoiled and folded into plectonemic loops, which are confined to different physical regions, and rarely diffuse into each other. These loops spatially organize into megabase-sized regions called macrodomains, within which DNA sites frequently interact, but between which interactions are rare. The condensed and spatially organized DNA forms a helical ellipsoid that is radially confined in the cell. The 3D structure of the DNA in the nucleoid appears to vary depending on conditions and is linked to gene expression so that the nucleoid architecture and gene transcription are tightly interdependent, influencing each other reciprocally.
Background edit
In many bacteria, the chromosome is a single covalently closed (circular) double-stranded DNA molecule that encodes the genetic information in a haploid form. The size of the DNA varies from 500,000 to several million base pairs (bp) encoding from 500 to several thousand genes depending on the organism.[2] The chromosomal DNA is present in cells in a highly compact, organized form called the nucleoid (meaning nucleus-like), which is not encased by a nuclear membrane as in eukaryotic cells.[6] The isolated nucleoid contains 80% DNA, 10% protein, and 10% RNA by weight.[7][8]
The gram-negative bacterium Escherichia coli is a model system for nucleoid research into how chromosomal DNA becomes the nucleoid, the factors involved therein, what is known about its structure, and how some of the DNA structural aspects influence gene expression.[2][3]
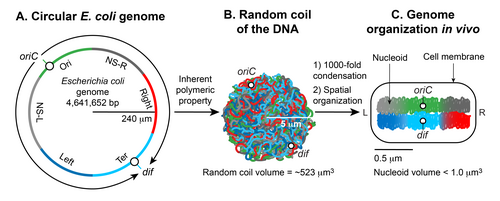
There are two essential aspects of nucleoid formation; condensation of a large DNA into a small cellular space and functional organization of DNA in a three-dimensional form. The haploid circular chromosome in E. coli consists of ~ 4.6 x 106 bp. If DNA is relaxed in the B form, it would have a circumference of ~1.5 millimeters (0.332 nm x 4.6 x 106). However, a large DNA molecule such as the E. coli chromosomal DNA does not remain a straight rigid molecule in a suspension.[5] Brownian motion will generate curvature and bends in DNA. The maximum length up to which a double-helical DNA remains straight by resisting the bending enforced by Brownian motion is ~50 nm or 150 bp, which is called the persistence length. Thus, pure DNA becomes substantially condensed without any additional factors; at thermal equilibrium, it assumes a random coil form.[4][5] The random coil of E. coli chromosomal DNA would occupy a volume (4/3 π r3) of ~ 523 µm3, calculated from the radius of gyration (Rg = (√N a)/√6) where a is the Kuhn length (2 x persistence length), and N is the number of Kuhn length segments in the DNA (total length of the DNA divided by a).[5] Although DNA is already condensed in the random coil form, it still cannot assume the volume of the nucleoid which is less than a micron. Thus, the inherent property of DNA is not sufficient: additional factors must help condense DNA further on the order of ~103 (volume of the random coil divided by the nucleoid volume). The second essential aspect of nucleoid formation is the functional arrangement of DNA. Chromosomal DNA is not only condensed but also functionally organized in a way that is compatible with DNA transaction processes such as replication, recombination, segregation, and transcription.[9][10][11] Almost five decades of research beginning in 1971,[7] has shown that the final form of the nucleoid arises from a hierarchical organization of DNA. At the smallest scale (1 kb or less), nucleoid-associated DNA architectural proteins condense and organize DNA by bending, looping, bridging or wrapping DNA. At a larger scale (10 kb or larger), DNA forms plectonemic loops, a braided form of DNA induced by supercoiling. At the megabase scale, the plectonemic loops coalesce into six spatially organized domains (macrodomains), which are defined by more frequent physical interactions among DNA sites within the same macrodomain than between different macrodomains.[12] Long- and short-range DNA-DNA connections formed within and between the macrodomains contribute to condensation and functional organization. Finally, the nucleoid is a helical ellipsoid with regions of highly condensed DNA at the longitudinal axis.[13][14][15]
Condensation and organization edit
Nucleoid-associated proteins (NAPs) edit
In eukaryotes, genomic DNA is condensed in the form of a repeating array of DNA-protein particles called nucleosomes.[16][17][18]
A nucleosome consists of ~146 bp of DNA wrapped around an octameric complex of the histone proteins. Although bacteria do not have histones, they possess a group of DNA binding proteins referred to as nucleoid-associated proteins (NAPs) that are functionally analogous to histones in a broad sense. NAPs are highly abundant and constitute a significant proportion of the protein component of nucleoid.[19]
A distinctive characteristic of NAPs is their ability to bind DNA in both a specific (either sequence- or structure-specific) and non-sequence specific manner. As a result, NAPs are dual function proteins.[20] The specific binding of NAPs is mostly involved in gene-specific transcription, DNA replication, recombination, and repair.[9][10][11] At the peak of their abundance, the number of molecules of many NAPs is several orders of magnitude higher than the number of specific binding sites in the genome.[20] Therefore, it is reasoned that NAPs bind to the chromosomal DNA mostly in the non-sequence specific mode and it is this mode that is crucial for chromosome compaction. It is noteworthy that so-called non-sequence specific binding of a NAP may not be completely random. There could be low-sequence specificity and or structural specificity due to sequence-dependent DNA conformation or DNA conformation created by other NAPs.[18]
Although molecular mechanisms of how NAPs condense DNA in vivo are not well understood, based on the extensive in vitro studies it appears that NAPs participate in chromosome compaction via the following mechanisms: NAPs induce and stabilize bends in DNA, thus aid in DNA condensation by reducing the persistence length.[20] NAPs condense DNA by bridging, wrapping, and bunching that could occur between nearby DNA segments or distant DNA segments of the chromosome. Another mechanism by which NAPs participate in chromosome compaction is by constraining negative supercoils in DNA thus contributing to the topological organization of the chromosome.[20]
There are at least 12 NAPs identified in E. coli,[20] the most extensively studied of which are HU, IHF, H-NS, and Fis. Their abundance and DNA binding properties and effect on DNA condensation and organization are summarized in the tables below.[20]
| Protein | Molecular mass (kDa) | Native functional unit | Abundance1 in growth phase | Abundance1 in stationary phase |
|---|---|---|---|---|
| HUα and HUβ | ~ 9 | Homo- and hetero-dimer | 55,000 (23) | 30,000 (12.5) |
| IHFα and IHFβ | ~ 11 | Heterodimer | 12,000 (5) | 55,000 (23) |
| H-NS | ~ 15 | Homodimer | 20,000 (8) | 15,000 (6) |
| Fis | ~ 11 | Homodimer | 60,000 (25) | Undetectable |
| Dps | ~ 19 | Dodecamer | 6,000 (0.4) | 180,000 (12.5) |
1 Abundance (molecules/cell) data were taken from;[21] The number in the parenthesis is micromolar concentration calculated using the following formula: (number of native functional units/Avogadro number) x (1/cell volume in liter) x 103. Cell volume in liter ( 2 x 10−15) was determined by assuming volume of the E. coli cell to be 2 μm3.[21]
| Protein | Binding motif | Specific DNA binding affinity1 | Random DNA binding affinity1 |
|---|---|---|---|
| HU | A structural motif defined by bends and kinks in DNA[22][23] | 7.5 x 10−9[24] | 4.0 x 10−7[24] |
| IHF | WATCAANNNNTTR[25] | 1.5 x 10−9[26] | 1.7 x 10−6[26] |
| H-NS | TCGATAAATT[27] | 10-15 x 10−9[28] | 6 x 10−8[28] |
| Fis | GNTYAAAWTTTRANC[29] | 0.2-1.0 x 10−9[29][30] | >8.0 x 10−6[30] |
| Dps | ND | ND | 1.65 x 10−7[31] |
| MatP | GTGACRNYGTCAC[32] | 8.0 x 10−9 | ND |
| MukBEF | ND | ND | ND |
1 Binding affinity refers to equilibrium dissociation constant (Kd) in molar units (M). ND = not determined
HU edit
Histone-like protein from E. coli strain U93 (HU) is an evolutionarily conserved protein in bacteria.[33][34] HU exists in E. coli as homo- and heterodimers of two subunits HUα and HUβ sharing 69% amino acid identity.[35] Although it is referred to as a histone-like protein, close functional relatives of HU in eukaryotes are high-mobility group (HMG) proteins, and not histones.[36][37] HU is a non-sequence specific DNA binding protein. It binds with low-affinity to any linear DNA. However, it preferentially binds with high-affinity to a structurally distorted DNA.[38][39][40][41][42][24] Examples of distorted DNA substrates include cruciform DNA, bulged DNA, dsDNA containing a single-stranded break such as nicks, gaps, or forks. Furthermore, HU specifically binds and stabilizes a protein-mediated DNA loop.[43] In the structurally specific DNA binding mode, HU recognizes a common structural motif defined by bends or kinks created by distortion,[22][44][23] whereas it binds to a linear DNA by locking the phosphate backbone.[45] While the high-affinity structurally-specific binding is required for specialized functions of HU such as site-specific recombination, DNA repair, DNA replication initiation, and gene regulation,[9][10][11] it appears that the low-affinity general binding is involved in DNA condensation.[45] In chromatin-immunoprecipitation coupled with DNA sequencing (ChIP-Seq), HU does not reveal any specific binding events.[46] Instead, it displays a uniform binding across the genome presumably reflecting its mostly weak, non-sequence specific binding, thus masking the high-affinity binding in vivo.[46]
In strains lacking HU, the nucleoid is "decondensed", consistent with a role of HU in DNA compaction.[47] The following in vitro studies suggest possible mechanisms of how HU might condense and organize DNA in vivo. Not only HU stably binds to distorted DNA with bends, it induces flexible bends even in a linear DNA at less than 100 nM concentration. In contrast, HU shows the opposite architectural effect on DNA at higher physiologically relevant concentrations.[45][9][10][11][47][48] It forms rigid nucleoprotein filaments causing the straitening of DNA and not the bending. The filaments can further form a DNA network (DNA bunching) expandable both laterally and medially because of the HU-HU multimerization triggered by the non-sequence-specific DNA binding.[45]
How are these behaviors of HU relevant inside the cell? The formation of filaments requires high-density binding of HU on DNA, one HU dimer per 9-20 bp DNA. But there is only one HU dimer every ~150 bp of the chromosomal DNA based on the estimated abundance of 30,000 HU dimers per cell (4600000 bp /30,000).[21] This indicates that the flexible bends are more likely to occur in vivo. The flexible bending would cause condensation due to a reduction in the persistence length of DNA as shown by magnetic tweezers experiments, which allow studying condensation of a single DNA molecule by a DNA binding protein.[48][49] However, because of the cooperativity, the rigid filaments and networks could form in some regions in the chromosome. The filament formation alone does not induce condensation,[48] but DNA networking or bunching can substantially contribute to condensation by bringing distant or nearby chromosome segments together.[45]
IHF edit
Integration host factor (IHF) is structurally almost identical to HU[51] but behaves differently from HU in many aspects. Unlike HU, which preferentially binds to a structural motif regardless of the sequence, IHF preferentially binds to a specific DNA sequence even though the specificity arises through the sequence-dependent DNA structure and deformability. The specific binding of IHF at cognate sites bends DNA sharply by >160-degree.[51] An occurrence of the cognate sequence motif is about 3000 in the E. coli genome.[46] The estimated abundance of IHF in the growth phase is about 6000 dimers per cell. Assuming that one IHF dimer binds to a single motif and nucleoid contains more than one genome equivalent during the exponential growth phase, most of the IHF molecules would occupy specific sites in the genome and likely only condense DNA by inducing sharp bending.[46]
Besides preferential binding to a specific DNA sequence, IHF also binds to DNA in a non-sequence specific manner with the affinities similar to HU. A role of the non-specific binding of IHF in DNA condensation appears to be critical in the stationary phase because the IHF abundance increases by five-fold in the stationary phase and the additional IHF dimers would likely bind the chromosomal DNA non-specifically.[21][52][53] Unlike HU, IHF does not form thick rigid filaments at higher concentrations. Instead, its non-specific binding also induces DNA bending albeit the degree of bending is much smaller than that at specific sites and is similar to the flexible bending induced by HU in a linear DNA at low concentrations.[54] In vitro, the bending induced by non-specific binding of IHF can cause DNA condensation and promotes the formation of higher-order nucleoprotein complexes depending on the concentrations of potassium chloride and magnesium chloride.[54] The higher-order DNA organization by IHF in vivo is as yet unclear.[54]
H-NS edit
A distinguishable feature of histone-like or heat-stable nucleoid structuring protein (H-NS)[55][56][57][58] from other NAPs is the ability to switch from the homodimeric form at relatively low concentrations (<1 x 10−5 M) to an oligomeric state at higher levels.[59][60] Because of oligomerization properties, H-NS spreads laterally along AT-rich DNA in a nucleation reaction, where high-affinity sites function as nucleation centers.[61][62][28] The spreading of H-NS on DNA results in two opposite outcomes depending on the magnesium concentration in the reaction. At low magnesium concentration (< 2 mM), H-NS forms rigid nucleoprotein filaments whereas it forms inter- and intra-molecular bridges at higher magnesium concentrations (> 5 mM).[63][64][65][66][67] The formation of rigid filaments results in straightening of DNA with no condensation whereas the bridging causes substantial DNA folding.[66] Analysis of H-NS binding in the genome by ChIP-Seq assays provided indirect evidence for the spreading of H-NS on DNA in vivo. H-NS binds selectively to 458 regions in the genome.[50] Although H-NS has been demonstrated to prefer curved DNA formed by repeated A-tracks in DNA sequences[61][68] the basis of the selective binding is the presence of a conserved sequence motif found in AT-rich regions.[27] More importantly, the frequent occurrence of the sequence motif within an H-NS binding region that can re-enforce the cooperative protein-protein interactions, and the unusually long length of the binding region are consistent with the spreading of the protein. Whether the filament formation or DNA bridging is prevalent in vivo depends on the physiological concentration of magnesium inside the cell.[66][69] If the magnesium concentration is uniformly low (< 5 mM), H-NS would form rigid nucleoprotein filaments in vivo.[66] Alternatively, if there is an uneven distribution of magnesium in the cell, it could promote both DNA bridging and stiffening but in different regions of the nucleoid.[66]
Furthermore, H-NS is best known as a global gene silencer that preferentially inhibits transcription of horizontally transferred genes and it is the rigid filament that leads to gene silencing.[70][71] Taken together, it appears that the formation of rigid filaments is the most likely outcome of H-NS-DNA interactions in vivo that leads to gene silencing but does not induce DNA condensation. Consistently, the absence of H-NS does not change the nucleoid volume.[72] However, it is possible that E. coli experiences high-magnesium concentration under some environmental conditions. In such conditions, H-NS can switch from its filament inducing form to the bridge inducing form that contributes to DNA condensation and organization.[66]
Fis edit
Factor for Inversion Stimulation (Fis) is a sequence specific DNA binding protein that binds to specific DNA sequences containing a 15-bp symmetric motif.[29][30][73] Like IHF, Fis induces DNA bending at cognate sites. The ability to bend DNA is apparent in the structure of Fis homodimer. A Fis homodimer possesses two helix-turn-helix (HTH) motifs, one from each monomer. An HTH motif typically recognizes the DNA major groove. However, the distance between the DNA recognition helices of the two HTH motifs in the Fis homodimer is 25 Å, that is ~ 8 Å shorter than the pitch of a canonical B-DNA, indicating that the protein must bend or twist DNA to bind stably.[74][75] Consistently, the crystal structure of Fis-DNA complexes shows that the distance between the recognition helices remains unchanged whereas DNA curves in the range of 60-75 degree.[30] There are 1464 Fis binding regions distributed across the E. coli genome and a binding motif, identified computationally, matches with the known 15-bp motif.[50][76] Specific binding of Fis at such sites would induce bends in DNA, thus contribute to DNA condensation by reducing persistence length of DNA. Furthermore, many Fis binding sites occur in tandem such as those in the stable RNA promoters, e.g., P1 promoter of rRNA operon rrnB. The coherent bending by Fis at the tandem sites is likely to create a DNA micro-loop that can further contribute to DNA condensation.[77]
Besides high-affinity specific binding to cognate sites, Fis can bind to a random DNA sequence. The non-specific DNA binding is significant because Fis is as abundant as HU in the growth phase. Therefore, most of Fis molecules are expected to bind DNA in a non-sequence specific manner. Magnetic tweezers experiments show that this non-specific binding of Fis can contribute to DNA condensation and organization.[78][79] Fis causes mild condensation of a single DNA molecule at <1 mM, but induces substantial folding through the formation of DNA loops of an average size of ~800 bp at >1 mM. The loops in magnetic tweezers experiments are distinct from the micro-loops created by coherent DNA bending at cognate sites, as they require the formation of high-density DNA-protein complexes achieved by sequence-independent binding. Although, occurrence of such loops in vivo remains to be demonstrated, high-density binding of Fis may occur in vivo through concerted action of both specific and non-specific binding. The in-tandem occurrence of specific sites might initiate a nucleation reaction similar to that of H-NS, and then non-specific binding would lead to the formation of localized high-density Fis arrays. The bridging between these localized regions can create large DNA loops.[79] Fis is exclusively present in the growth phase and not in the stationary phase.[80][81] Thus, any role in chromosomal condensation by Fis must be specific to growing cells.[81]
Nucleoid-associated RNAs (naRNAs) edit
Early studies examining the effect of RNase A treatment on isolated nucleoids indicated that RNA participated in the stabilization of the nucleoid in the condensed state.[82] Moreover, treatment with RNase A disrupted the DNA fibers into thinner fibers, as observed by an atomic force microscopy of the nucleoid using the “on-substrate lysis procedure”.[83] These findings demonstrated the participation of RNA in the nucleoid structure, but the identity of the RNA molecule(s) remained unknown until recently.[47] Most of the studies on HU focused on its DNA binding.[83] However, HU also binds to dsRNA and RNA-DNA hybrids with a lower affinity similar to that with a linear dsDNA.[84] Moreover, HU preferentially binds to RNA containing secondary structures and an RNA-DNA hybrid in which the RNA contains a nick or overhang.[84][85] The binding affinities of HU with these RNA substrates are similar to those with which it binds to distorted DNA. An immunoprecipitation of HU-bound RNA coupled to reverse transcription and microarray (RIP-Chip) study as well as an analysis of RNA from purified intact nucleoids identified nucleoid-associated RNA molecules that interact with HU.[47] Several of them are non-coding RNAs, and one such RNA named naRNA4 (nucleoid-associated RNA 4), is encoded in a repetitive extragenic palindrome (REP325). In a strain lacking REP325, the nucleoid is decondensed as it is in a strain lacking HU.[47] naRNA4 most likely participate in DNA condensation by connecting DNA segments in the presence of HU.[86] Recent studies provide insights into the molecular mechanism of how naRNA4 establishes DNA-DNA connections. The RNA targets regions of DNA containing cruciform structures and forms an RNA-DNA complex that is critical for establishing DNA-DNA connections.[87] Surprisingly, although HU helps in the formation of the complex, it is not present in the final complex, indicating its potential role as a catalyst (chaperone). The nature of the RNA-DNA complex remains puzzling because the formation of the complex does not involve extensive Watson/Crick base pairing but is sensitive to RNase H, which cleaves RNA in an RNA-DNA hybrid and the complex binds to an antibody specific to RNA-DNA hybrids.[47][83][84]
Supercoiling edit
Because of its helical structure, a double-stranded DNA molecule becomes topologically constrained in the covalently closed circular form which eliminates the rotation of the free ends.[88] The number of times the two strands cross each other in a topologically constrained DNA is called the linking number (Lk), which is equivalent to the number of helical turns or twists in a circular molecule.[89] The Lk of a topological DNA remains invariant, no matter how the DNA molecule is deformed, as long as neither strand is broken.[90][91]
The Lk of DNA in the relaxed form is defined as Lk0. For any DNA, Lk0 can be calculated by dividing the length (in bp) of the DNA by the number of bp per helical turn. This is equal to 10.4 bp for the relaxed B-form DNA. Any deviation from Lk0 causes supercoiling in DNA. A decrease in the linking number (Lk<Lk0) creates negative supercoiling whereas an increase in the linking number (Lk>Lk0) creates positive supercoiling.[92][90]
The supercoiled state (when Lk is not equal to Lk0) results in a transition in DNA structure that can manifest as a change in the number of twists (negative <10.4 bp/turn, positive >10.4 bp per turn) and/or in the formation of writhes, called supercoils. Thus, Lk is mathematically defined as a sign dependent sum of the two geometric parameters, twist and writhe. A quantitative measure of supercoiling that is independent of the size of DNA molecules is the supercoiling density (σ) where σ =∆Lk/Lk0.[91]
Writhes can adopt two structures; plectoneme and solenoid or toroid. A plectonemic structure arises from the interwinding of the helical axis. Toroidal supercoils originate when DNA forms several spirals, around an axis and not intersecting with each other, like those in a telephone cord.[90] The writhes in the plectonemes form are right- and left-handed in positively or negatively supercoiled DNA, respectively. The handedness of the toroidal supercoils is opposite to those of plectonemes. Both plectonemes and toroidal supercoils can be either in a free form or restrained in a bound form with proteins. The best example of the bound toroidal supercoiling in biology is the eukaryotic nucleosome in which DNA wraps around histones.[17]
Plectonemic supercoils in E. coli edit
In most bacteria, DNA is present in supercoiled form. The circular nature of the E. coli chromosome makes it topologically constrained molecule that is mostly negatively supercoiled with an estimated average supercoiling density (σ) of -0.05.[93] In the eukaryotic chromatin, DNA is found mainly in the toroidal form that is restrained and defined by histones through the formation of nucleosomes. In contrast, in the E. coli nucleoid, about half of the chromosomal DNA is organized in the form of free, plectonemic supercoils.[94][95][96] The remaining DNA is restrained in either the plectonemic form or alternative forms, including but not limited to the toroidal form, by interaction with proteins such as NAPs. Thus, plectonemic supercoils represent effective supercoiling of the E. coli genome that is responsible for its condensation and organization. Both plectonemic and toroidal supercoiling aid in DNA condensation. It is noteworthy that because of branching of plectonemic structures, it provides less DNA condensation than does the toroidal structure. A same size DNA molecule with equal supercoiling densities is more compact in a toroidal form than in a plectonemic form. In addition to condensing DNA, supercoiling aids in DNA organization. It promotes disentanglement of DNA by reducing the probability of catenation.[97] Supercoiling also helps bring two distant sites of DNA in proximity thereby promoting a potential functional interaction between different segments of DNA.[91]
Sources of supercoiling in E. coli edit
Three factors contribute to generating and maintaining chromosomal DNA supercoiling in E. coli: (i) activities of topoisomerases, (ii) the act of transcription, and (iii) NAPs.[95]
Topoisomerases edit
Topoisomerases are a particular category of DNA metabolic enzymes that create or remove supercoiling by breaking and then re-ligating DNA strands.[98] E. coli possesses four topoisomerases. DNA gyrase introduces negative supercoiling in the presence of ATP and it removes positive supercoiling in the absence of ATP.[99] Across all forms of life, DNA gyrase is the only topoisomerase that can create negative supercoiling and it is because of this unique ability that bacterial genomes possess free negative supercoils; DNA gyrase is found in all bacteria but absent from higher eukaryotes. In contrast, Topo I opposes DNA gyrase by relaxing the negatively supercoiled DNA.[100][101] There is genetic evidence to suggest that a balance between the opposing activities of DNA gyrase and Topo I are responsible for maintaining a steady-state level of average negative superhelicity in E. coli.[100][102] Both enzymes are essential for E. coli survival. A null strain of topA, the gene encoding Topo I, survives only because of the presence of suppressor mutations in the genes encoding DNA gyrase.[100][102] These mutations result in reduced gyrase activity, suggesting that excess negative supercoiling due to the absence of Topo I is compensated by reduced negative supercoiling activity of DNA gyrase. Topo III is dispensable in E. coli and is not known to have any role in supercoiling in E. coli.[103] The primary function of Topo IV is to resolve sister chromosomes. However, it has been shown to also contribute to the steady-state level of negative supercoiling by relaxing negative supercoiling together with Topo I.[104][105]
| Topoisomerase | Type | Function | Single- or double-stranded cleavage |
|---|---|---|---|
| Topoisomerase I | IA | Removes (-) supercoiling | SS |
| Topoisomerase III | IA | Removes (-) supercoiling | SS |
| Topoisomerase IV | IIA | Removes (-) supercoiling | DS |
| DNA gyrase | IIA | Creates (-) supercoiling and removes (+) supercoiling | DS |
Transcription edit
A twin supercoiling domain model proposed by Liu and Wang argued that unwinding of DNA double helix during transcription induces supercoiling in DNA as shown in.[106] According to their model, transcribing RNA polymerase (RNAP) sliding along DNA forces the DNA to rotate on its helical axis. A hindrance in the free rotation of DNA might arise due to a topological constraint, causing the DNA in front of RNAP to become over-twisted (positively supercoiled) and the DNA behind RNAP would become under-twisted (negatively supercoiled). It has been found that a topological constraint is not needed because RNAP generates sufficient torque that causes supercoiling even in a linear DNA template.[107] If DNA is already negatively supercoiled, this action relaxes existing negative supercoils before causing a buildup of positive supercoils ahead of RNAP and introduces more negative supercoils behind RNAP. In principle, DNA gyrase and Topo I should remove excess positive and negative supercoils respectively but if the RNAP elongation rate exceeds the turnover of the two enzymes, transcription contributes to the steady-state level of supercoiling.[107]
Control of supercoiling by NAPs edit
In the eukaryotic chromatin, DNA is rarely present in the free supercoiled form because nucleosomes restrain almost all negative supercoiling through tight binding of DNA to histones. Similarly, in E. coli, nucleoprotein complexes formed by NAPs restrain half of the supercoiling density of the nucleoid.[93][96] In other words, if a NAP dissociates from a nucleoprotein complex, the DNA would adopt the free, plectonemic form. DNA binding of HU, Fis, and H-NS has been experimentally shown to restrain negative supercoiling in a relaxed but topologically constrained DNA.[108][109][110][111][112] They can do so either by changing the helical pitch of DNA or generating toroidal writhes by DNA bending and wrapping. Alternatively, NAPs can preferentially bind to and stabilize other forms of the underwound DNA such as cruciform structures and branched plectonemes. Fis has been reported to organize branched plectonemes through its binding to cross-over regions and HU preferentially binds to cruciform structures.[112]
NAPs also regulate DNA supercoiling indirectly. Fis can modulate supercoiling by repressing the transcription of the genes encoding DNA gyrase.[113] There is genetic evidence to suggest that HU controls supercoiling levels by stimulating DNA gyrase and reducing the activity of Topo I.[114][115] In support of the genetic studies, HU was shown to stimulate DNA gyrase-catalyzed decatenation of DNA in vitro.[116] It is unclear mechanistically how HU modulates the activities of the gyrase and Topo I. HU might physically interact with DNA gyrase and Topo I or DNA organization activities of HU such as DNA bending may facilitate or inhibit the action of DNA gyrase and Topo I respectively.[114][116]
Plectonemic supercoils organize into multiple topological domains edit
One of the striking features of the nucleoid is that plectonemic supercoils are organized into multiple topological domains.[117] In other words, a single cut in one domain will only relax that domain and not the others. A topological domain forms because of a supercoiling-diffusion barrier. Independent studies employing different methods have reported that the topological domains are variable in size ranging from 10 to 400 kb.[95][117][118] A random placement of barriers commonly observed in these studies seems to explain the wide variability in the size of domains.[117]
Although identities of domain barriers remain to be established, possible mechanisms responsible for the formation of the barriers include: (i) A domain barrier could form when a protein with an ability to restrain supercoils simultaneously binds to two distinct sites on the chromosome forming a topologically isolated DNA loop or domain. It has been experimentally demonstrated that protein-mediated looping in supercoiled DNA can create a topological domain.[119][120] NAPs such as H-NS and Fis are potential candidates, based on their DNA looping abilities and the distribution of their binding sites. (ii) Bacterial interspersed mosaic elements (BIMEs) also appear as potential candidates for domain barriers. BIMEs are palindromic repeats sequences that are usually found between genes. A BIME has been shown to impede diffusion of supercoiling in a synthetically designed topological cassette inserted in the E. coli chromosome.[121] There are ~600 BIMEs distributed across the genome, possibly dividing the chromosome into 600 topological domains.[122] (iii) Barriers could also result from the attachment of DNA to the cell membrane through a protein which binds to both DNA and membrane or through nascent transcription and the translation of membrane-anchored proteins. (iv) Transcription activity can generate supercoiling-diffusion barriers. An actively transcribing RNAP has been shown to block dissipation of plectonemic supercoils, thereby forming a supercoiling-diffusion barrier.[123][124][125]
Growth-phase dependent nucleoid dynamics edit
The nucleoid reorganizes in stationary phase cells suggesting that the nucleoid structure is highly dynamic, determined by the physiological state of cells. A comparison of high-resolution contact maps of the nucleoid revealed that the long-range contacts in the Ter macrodomain increased in the stationary phase, compared to the growth phase.[126] Furthermore, CID boundaries in the stationary phase were different from those found in the growth phase. Finally, nucleoid morphology undergoes massive transformation during prolonged stationary phase;[127] the nucleoid exhibits ordered, toroidal structures.[128]
Growth-phase specific changes in nucleoid structure could be brought about by a change in levels of nucleoid-associated DNA architectural proteins (the NAPs and the Muk subunits), supercoiling, and transcription activity. The abundance of NAPs and the Muk subunits changes according to the bacterial growth cycle. Fis and the starvation-induced DNA binding protein Dps, another NAP, are almost exclusively present in the growth phase and stationary phase respectively. Fis levels rise upon entry into exponential phase and then rapidly decline while cells are still in the exponential phase, reaching levels that are undetectable in stationary phase.[129] While Fis levels start to decline, levels of Dps start to rise and reach a maximum in the stationary phase.[21] A dramatic transition in the nucleoid structure observed in the prolonged stationary phase has been mainly attributed to Dps. It forms DNA/crystalline assemblies that act to protect the nucleoid from DNA damaging agents present during starvation.[128]
HU, IHF, and H-NS are present in both growth phase and stationary phase.[21] However, their abundance changes significantly such that HU and Fis are the most abundant NAPs in the growth phase, whereas IHF and Dps become the most abundant NAPs in the stationary phase.[21] HUαα is the predominant form in early exponential phase, whereas the heterodimeric form predominates in the stationary phase, with minor amounts of homodimers.[130] This transition has functional consequences regarding nucleoid structure, because the two forms appear to organize and condense DNA differently; both homo- and heterodimers form filaments, but only the homodimer can bring multiple DNA segments together to form a DNA network.[45] The copy number of MukB increases two-fold in stationary phase.[131][132] An increase in the number of MukB molecules could have influence on the processivity of the MukBEF complex as a DNA loop extruding factor resulting in larger or a greater number of the loops.[131][132]
Supercoiling can act in a concerted manner with DNA architectural proteins to reorganize the nucleoid. The overall supercoiling level decreases in the stationary phase, and supercoiling exhibits a different pattern at the regional level.[133] Changes in supercoiling can alter the topological organization of the nucleoid. Furthermore, because a chromosomal region of high transcription activity forms a CID boundary, changes in transcription activity during different growth phases could alter the formation of CID boundaries, and thus the spatial organization of the nucleoid. It is possible that changes in CID boundaries observed in the stationary phase could be due to the high expression of a different set of genes in the stationary phase compared to the growth phase.[126]
Nucleoid structure and gene expression edit
NAPs and gene expression edit
The E. coli chromosome structure and gene expression appear to influence each other reciprocally. On the one hand, a correlation of a CID boundary with high transcription activity indicates that chromosome organization is driven by transcription. On the other hand, the 3D structure of DNA within nucleoid at every scale may be linked to gene expression. First, it has been shown that reorganization of the 3D architecture of the nucleoid in E. coli can dynamically modulate cellular transcription pattern.[134] A mutant of HUa made the nucleoid very much condensed by increased positive superhelicity of the chromosomal DNA. Consequently, many genes were repressed, and many quiescent genes were expressed. Besides, there are many specific cases in which protein-mediated local architectural changes alter gene transcription. For example, the formation of rigid nucleoprotein filaments by H-NS blocks RNAP access to the promoter thus prevent gene transcription.[135] Through gene silencing, H-NS acts as a global repressor preferentially inhibiting transcription of horizontally transferred genes.[50][27] In another example, specific binding of HU at the gal operon facilitates the formation of a DNA loop that keeps the gal operon repressed in the absence of the inducer.[136] The topologically distinct DNA micro-loop created by coherent bending of DNA by Fis at stable RNA promoters activates transcription.[77] DNA bending by IHF differentially controls transcription from the two tandem promoters of the ilvGMEDA operon in E. coli.[137][138] Specific topological changes by NAPs not only regulate gene transcription, but are also involved in other processes such as DNA replication initiation, recombination, and transposition.[9][10][11] In contrast to specific gene regulation, how higher-order chromosome structure and its dynamics influences gene expression globally at the molecular level remains to be worked out.[139]
DNA supercoiling and gene expression edit
A two-way interconnectedness exists between DNA supercoiling and gene transcription.[139] Negative supercoiling of the promoter region can stimulate transcription by facilitating the promoter melting and by increasing the DNA binding affinity of a protein regulator. Stochastic bursts of transcription appear to be a general characteristic of highly expressed genes, and supercoiling levels of the DNA template contributes to transcriptional bursting.[140] According to the twin supercoiling domain model, transcription of a gene can influence transcription of other nearby genes through a supercoiling relay. One such example is the activation of the leu-500 promoter.[139] Supercoiling not only mediates gene-specific changes, but it also mediates large-scale changes in gene expression. Topological organization of the nucleoid could allow independent expression of supercoiling-sensitive genes in different topological domains. A genome-scale map of unrestrained supercoiling showed that genomic regions have different steady-state supercoiling densities, indicating that the level of supercoiling differs in individual topological domains.[133] As a result, a change in supercoiling can result in domain-specific gene expression, depending on the level of supercoiling in each domain.[133]
The effect of supercoiling on gene expression can be mediated by NAPs that directly or indirectly influence supercoiling. The effect of HU on gene expression appears to involve a change in supercoiling and perhaps a higher-order DNA organization. A positive correlation between DNA gyrase binding and upregulation of the genes caused by the absence of HU suggests that changes in supercoiling are responsible for differential expression. HU was also found to be responsible for a positional effect on gene expression by insulating transcriptional units by constraining transcription-induced supercoiling.[141] Point mutations in HUa dramatically changed the gene expression profile of E. coli, altering its morphology, physiology, and metabolism. As a result, the mutant strain was more invasive of mammalian cells.[134][142] This dramatic effect was concomitant with nucleoid compaction and increased positive supercoiling.[45][143] The mutant protein was an octamer, in contrast to the wild-type dimer. It wraps DNA on its surface in a right-handed manner, restraining positive supercoils as opposed to wild-type HU.[143] These studies show that amino acid substitutions in HU can have a dramatic effect on nucleoid structure, that in turn results in significant phenotypic changes.[143]
Since MukB and HU have emerged as critical players in long-range DNA interactions, it will be worthwhile to compare the effect of each of these two proteins on global gene expression.[144] Although HU appears to control gene expression by modulating supercoiling density, the exact molecular mechanism remains unknown and the impact of MukB on gene expression is yet to be analyzed.[144][145]
Spatial organization edit
Chromosomal interaction domains edit
In recent years, the advent of a molecular method called chromosome conformation capture (3C) has allowed studying a high-resolution spatial organization of chromosomes in both bacteria and eukaryotes.[146] 3C and its version that is coupled with deep sequencing (Hi-C)[147] determine physical proximity, if any, between any two genomic loci in 3D space. A high-resolution contact map of bacterial chromosomes including the E. coli chromosome has revealed that a bacterial chromosome is segmented into many highly self-interacting regions called chromosomal interaction domains (CIDs).[126][148][149] CIDs are equivalent to topologically associating domains (TADs) observed in many eukaryotic chromosomes,[150] suggesting that the formation of CIDs is a general phenomenon of genome organization. Two characteristics define CIDs or TADs. First, genomic regions of a CID physically interact with each other more frequently than with the genomic regions outside that CID or with those of a neighboring CID. Second, the presence of a boundary between CIDs that prevents physical interactions between genomic regions of two neighboring CIDs.[126]
The E. coli chromosome was found to consist of 31 CIDs in the growth phase. The size of the CIDs ranged from 40 to ~300 kb. It appears that a supercoiling-diffusion barrier responsible for segregating plectonemic DNA loops into topological domains functions as a CID boundary in E. coli and many other bacteria. In other words, the presence of a supercoiling-diffusion barrier defines the formation of CIDs. Findings from the Hi-C probing of chromosomes in E. coli, Caulobacter crescentus, and Bacillus subtilis converge on a model that CIDs form because plectonemic looping together with DNA organization activities of NAPs promotes physical interactions among genomic loci, and a CID boundary consists of a plectoneme-free region (PFR) that prevents these interactions. A PFR is created due to high transcription activity because the helical unwinding of DNA by actively transcribing RNAP restrains plectonemic supercoils. As a result, dissipation of supercoils is also blocked, creating a supercoiling-diffusion barrier. Indirect evidence for this model comes from an observation that CIDs of bacterial chromosomes including the E. coli chromosome display highly transcribed genes at their boundaries, indicating a role of transcription in the formation of a CID boundary.[126][148] More direct evidence came from a finding that the placement of a highly transcribed gene at a position where no boundary was present created a new CID boundary in the C. crescentus chromosome.[148] However, not all CID boundaries correlated with highly transcribed genes in the E. coli chromosome suggesting that other unknown factors are also responsible for the formation of CID boundaries and supercoiling diffusion barriers.[148]
Macrodomains edit
Plectonemic DNA loops organized as topological domains or CIDs appear to coalesce further to form large spatially distinct domains called macrodomains (MDs). In E. coli, MDs were initially identified as large segments of the genome whose DNA markers localized together (co-localized) in fluorescence in situ hybridization (FISH) studies.[151][152] A large genomic region (~1-Mb) covering oriC (origin of chromosome replication) locus co-localized and was called Ori macrodomain. Likewise, a large genomic region (~1-Mb) covering the replication terminus region (ter) co-localized and was called Ter macrodomain. MDs were later identified based on how frequently pairs of lambda att sites that were inserted at various distant locations in the chromosome recombined with each other. In this recombination-based method, an MD was defined as a large genomic region whose DNA sites can primarily recombine with each other, but not with those outside of that MD. The recombination-based method confirmed the Ori and Ter MDs that were identified in FISH studies and identified two additional MDs.[12][153]
The two additional MDs were formed by the additional ~1-Mb regions flanking the Ter and were referred to as Left and Right. These four MDs (Ori, Ter, Left, and Right) composed most of the genome, except for two genomic regions flanking the Ori. These two regions (NS-L and NS-R) were more flexible and non-structured compared to an MD as DNA sites in them recombined with DNA sites located in MDs on both sides. The genetic position of oriC appears to dictate the formation of MDs, because repositioning of oriC by genetic manipulation results in the reorganization of MDs. For example, genomic regions closest to the oriC always behave as an NS regardless of DNA sequence and regions further away always behave as MDs.[154]
The Hi-C technique further confirmed a hierarchical spatial organization of CIDs in the form of macrodomains.[126] In other words, CIDs of a macrodomain physically interacted with each other more frequently than with CIDs of a neighboring macrodomain or with genomic loci outside of that macrodomain. The Hi-C data showed that the E. coli chromosome was partitioning into two distinct domains. The region surrounding ter formed an insulated domain that overlapped with the previously identified Ter MD. DNA-DNA contacts in this domain occurred only in the range of up to ~280 kb. The rest of the chromosome formed a single domain whose genomic loci exhibited contacts in the range of >280-kb.[126] While most of the contacts in this domain were restricted to a maximum distance of ~500 kb, there were two loose regions whose genomic loci formed contacts at even greater distances (up to ~1 Mb). These loose regions corresponded to the previously identified flexible and less-structured regions (NS). The boundaries of the insulated domain encompassing ter and the two loose regions identified by the Hi-C method segmented the entire chromosome into six regions that correspond with the four MDs and two NS regions defined by recombination-based assays.[126]
Proteins that drive macrodomain formation edit
MatP edit
A search for protein(s) responsible for macrodomain formation led to identification of Macrodomain Ter protein (MatP). MatP almost exclusively binds in the Ter MD by recognizing a 13-bp motif called the macrodomain ter sequence (matS).[32] There are 23 matS sites present in the Ter domain, on average there is one site every 35-kb. Further evidence of MatP binding in the Ter domain comes from fluorescence imaging of MatP. Discrete MatP foci were observed that co-localized with Ter domain DNA markers.[32] A strong enrichment of ChIP-Seq signal in the Ter MD also corroborates the preferential binding of MatP to this domain.[32]
MatP condenses DNA in the Ter domain because the lack of MatP increased the distance between two fluorescent DNA markers located 100-kb apart in the Ter domain. Furthermore, MatP is a critical player in insulating the Ter domain from the rest of the chromosome.[126] It promotes DNA-DNA contacts within the Ter domain but prevents contacts between the DNA loci of Ter domain and those of flanking regions. How does MatP condense DNA and promote DNA-DNA contacts? The experimental results are conflicting. MatP can form a DNA loop between two matS sites in vitro and its DNA looping activity depends on MatP tetramerization. Tetramerization occurs via coiled-coil interactions between two MatP molecules bound to DNA.[156] One obvious model based on in vitro results is that MatP promotes DNA-DNA contacts in vivo by bridging matS sites. However, although MatP connected distant sites in Hi-C studies, it did not specifically connect the matS sites. Furthermore, a MatP mutant that was unable to form tetramers behaved like wild-type. These results argue against the matS bridging model for Ter organization, leaving the mechanism of MatP action elusive. One possibility is that MatP spreads to nearby DNA segments from its primary matS binding site and bridge distant sites via a mechanism that does not depend on the tetramerization.[156]
MukBEF edit
MukB belongs to a family of ATPases called structural maintenance of chromosome proteins (SMCs), which participate in higher-order chromosome organization in eukaryotes.[145] Two MukB monomers associate via continuous antiparallel coiled-coil interaction forming a 100-nm long rigid rod. A flexible hinge region occurs in the middle of the rod.[162][163] Due to the flexibility of the hinge region, MukB adopts a characteristic V-shape of the SMC family. The non-SMC subunits associating with MukB are MukE and MukF. The association closes the V formation, resulting in large ring-like structures. MukE and MukF are encoded together with MukB in the same operon in E. coli.[164] Deletion of either subunit results in the same phenotype suggesting that the MukBEF complex is the functional unit in vivo.[160] DNA binding activities of the complex reside in the MukB subunit, whereas MukE and MukF modulate MukB activity.[164]
MukBEF complex, together with Topo IV, is required for decatenation and repositioning of newly replicated oriCs.[165][166][167][168][155] The role of MukBEF is not restricted during DNA replication. It organizes and condenses DNA even in non-replicating cells.[131] The recent high-resolution chromosome conformation map of the MukB-depleted E. coli strain reveals that MukB participates in the formation of DNA-DNA interactions on the entire chromosome, except in the Ter domain.[126] How is MukB prevented from acting in the Ter domain? MatP physically interacts with MukB, thus preventing MukB from localizing to the Ter domain.[155] This is evident in the DNA binding of MatP and MukB in the Ter domain. DNA binding of MatP is enriched in the Ter domain, whereas DNA binding of MukB is reduced compared to the rest of the genome. Furthermore, in a strain already lacking MatP, the absence of MukB causes a reduction in DNA contacts throughout the chromosome, including the Ter domain.[126] This result agrees with the view that MatP displaces MukB from the Ter domain.[126]
How does the MukBEF complex function to organize the E. coli chromosome? According to the current view, SMC complexes organize chromosomes by extruding DNA loops.[169] SMC complexes translocate along DNA to extrude loops in a cis-manner (on the same DNA molecule), wherein the size of loops depends on processivity of the complex. SMC complexes from different organisms differ in the mechanism of loop extrusion.[169] Single molecule fluorescence microscopy of MukBEF in E. coli suggests that the minimum functional unit in vivo is a dimer of dimers.[160] This unit is formed by joining of two ATP-bound MukBEF complexes through MukF-mediated dimerization. MukBEF localizes in the cell as 1-3 clusters that are elongated parallel to the long axis of the cell. Each cluster contains an average ~ 8-10 dimers of dimers. According to the current model, the MukBEF extrudes DNA loops in a “rock-climbing” manner.[160][170] A dimer of the dimers releases one segment of DNA and capture a new DNA segment without dissociating from the chromosome. Besides DNA looping, a link between negative supercoiling and in vivo MukBEF function together with the ability of the MukB subunit to constrain negative supercoils in vitro suggests that MukBEF organizes DNA by generating supercoils.[171][172][173]
Role of NAPs and naRNAs edit
In addition to contributing to the chromosome compaction by bending, bridging, and looping DNA at a smaller scale (~1-kb), NAPs participate in DNA condensation and organization by promoting long-rang DNA-DNA contacts. Two NAPs, Fis and HU, emerged as the key players in promoting long-range DNA-DNA contacts that occur throughout the chromosome.[126] It remains to be studied how DNA organization activities of Fis and HU that are well understood at a smaller scale (~1-kb) results in the formation of long-range DNA-DNA interactions. Nonetheless, some of the HU-mediated DNA interactions require the presence of naRNA4.[86] naRNA4 also participates in making long-range DNA contacts. HU catalyzes some of the contacts, not all, suggesting that RNA participates with other NAPs in forming DNA contacts. HU also appears to act together with MukB to promote long-range DNA-DNA interactions. This view is based on observations that the absence of either HU or MukB caused a reduction in the same DNA-DNA contacts. It is unclear how MukB and HU potentially act together in promoting DNA-DNA interactions. It is possible that the two proteins interact physically. Alternatively, while MukBEF extrudes large DNA loops, HU condenses and organizes those loops.[169][48]
edit
There are reports that functionally-related genes of E. coli are physically together in 3-D space within the chromosome even though they are far apart by genetic distance. Spatial proximity of functionally-related genes not only make the biological functions more compartmentalized and efficient but would also contribute to the folding and spatial organization of the nucleoid. A recent study using fluorescent markers for detection of specific DNA loci examined pairwise physical distances between the seven rRNA operons that are genetically separated from each other (by as much as two million bp). It reported that all of the operons, except rrnC, were in physical proximity.[174][175] Surprisingly, 3C-seq studies did not reveal the physical clustering of rrn operons, contradicting the results of the fluorescence-based study.[126] Therefore, further investigation is required to resolve these contradicting observations. In another example, GalR, forms an interaction network of GalR binding sites that are scattered across the chromosome.[176] GalR is a transcriptional regulator of the galactose regulon composed of genes encoding enzymes for transport and metabolism of the sugar D-galactose.[177] GalR exists in only one to two foci in cells[176] and can self-assemble into large ordered structures.[178] Therefore, it appears that DNA-bound GalR multimerizes to form long-distance interactions.[176][178]
Global shape and structure edit
Conventional transmission electron microscopy (TEM) of chemically fixed E. coli cells portrayed the nucleoid as an irregularly shaped organelle. However, wide-field fluorescence imaging of live nucleoids in 3D revealed a discrete, ellipsoid shape.[3][14][15] The overlay of a phase-contrast image of the cell and the fluorescent image of the nucleoid showed a close juxtaposition only in the radial dimension along its entire length of the nucleoid to the cell periphery. This finding indicates radial confinement of the nucleoid.[13] A detailed examination of the 3D fluorescence image after cross-sectioning perpendicular to its long axis further revealed two global features of the nucleoid: curvature and longitudinal, high-density regions. Examining the chirality of the centerline of the nucleoid by connecting the center of intensity of each cross-section showed that the overall nucleoid shape is curved.[15] The fluorescence intensity distribution in the cross-sections revealed a density substructure, consisting of curved, high-density regions or bundles at the central core, and low-density regions at the periphery.[13][14] One implication of the radial confinement is that it determines the curved shape of the nucleoid. According to one model, the nucleoid is forced to bend because it is confined into a cylindrical E. coli cell whose radius is smaller than its bendable length (persistence length).[13] This model was supported by observations that removal of the cell wall or inhibition of cell wall synthesis increased the radius of the cell and resulted in a concomitant increase in the helical radius and a decrease in the helical pitch in the nucleoid.[13]
Nucleoid-membrane connections edit
An expansion force due to DNA-membrane connections appears to function in opposition to condensation forces to maintain an optimal condensation level of the nucleoid. Cell-fractionation and electron microscopy studies first indicated the possibility of DNA-membrane connections.[179][180] There are now several known examples of DNA-membrane connections. Transertion is a mechanism of concurrent transcription, translation, and insertion of nascent membrane proteins that forms transient DNA-membrane contacts.[181] Transertion of two membrane proteins LacY and TetA has been demonstrated to cause the repositioning of chromosomal loci toward the membrane.[182] Another mechanism of nucleoid-membrane connections is through a direct contact between membrane-anchored transcription regulators and their target sites in the chromosome. One example of such as transcription regulator in E. coli is CadC. CadC contains a periplasmic sensory domain and a cytoplasmic DNA binding domain. Sensing of an acidic environment by its periplasmic sensory domain stimulates DNA binding activity of CadC, which then activates transcription of its target genes.[183] The membrane-localization of genes regulated by a membrane-anchored transcription regulator is yet to be demonstrated. Nonetheless, activation of target genes in the chromosome by these regulators is expected to result in a nucleoid-membrane contact albeit it would be a dynamic contact. Besides these examples, the chromosome is also specifically anchored to the cell membrane through protein-protein interaction between DNA-bound proteins, e.g., SlmA and MatP, and the divisome.[184][185] Since membrane-protein encoding genes are distributed throughout the genome, dynamic DNA-membrane contacts through transertion can act as a nucleoid expansion force. This expansion force would function in opposition to condensation forces to maintain an optimal condensation level. The formation of highly condensed nucleoids upon the exposure of E. coli cells to chloramphenicol, which blocks translation, provides support for the expansion force of transient DNA-membrane contacts formed through transertion.[186][187] The round shape of overly-condensed nucleoids after chloramphenicol treatment also suggests a role for transertion-mediated DNA-membrane contacts in defining the ellipsoid shape of the nucleoid.[187]
Visualization edit
The nucleoid can be clearly visualized on an electron micrograph at very high magnification, where, although its appearance may differ, it is clearly visible against the cytosol.[188] Sometimes even strands of what is thought to be DNA are visible. By staining with the Feulgen stain, which specifically stains DNA, the nucleoid can also be seen under a light microscope.[189] The DNA-intercalating stains DAPI and ethidium bromide are widely used for fluorescence microscopy of nucleoids. It has an irregular shape and is found in prokaryotic cells.[13][14]
DNA damage and repair edit
Changes in the structure of the nucleoid of bacteria and archaea are observed after exposure to DNA damaging conditions. The nucleoids of the bacteria Bacillus subtilis and Escherichia coli both become significantly more compact after UV irradiation.[190][191] Formation of the compact structure in E. coli requires RecA activation through specific RecA-DNA interactions.[192] The RecA protein plays a key role in homologous recombinational repair of DNA damage.
Similar to B. subtilis and E. coli above, exposures of the archaeon Haloferax volcanii to stresses that damage DNA cause compaction and reorganization of the nucleoid.[193] Compaction depends on the Mre11-Rad50 protein complex that catalyzes an early step in homologous recombinational repair of double-strand breaks in DNA. It has been proposed that nucleoid compaction is part of a DNA damage response that accelerates cell recovery by helping DNA repair proteins to locate targets, and by facilitating the search for intact DNA sequences during homologous recombination.[193]
See also edit
References edit
This article was adapted from the following source under a CC BY 4.0 license (2019) (reviewer reports):
Subhash Verma; Zhong Qian; Sankar L Adhya (December 2019). "Architecture of the Escherichia coli nucleoid". PLOS Genetics. 15 (12): e1008456. doi:10.1371/JOURNAL.PGEN.1008456. ISSN 1553-7390. PMC 6907758. PMID 31830036. Wikidata Q84825966.{{cite journal}}: CS1 maint: unflagged free DOI (link)
- ^ Thanbichler M, Wang SC, Shapiro L (October 2005). "The bacterial nucleoid: a highly organized and dynamic structure". Journal of Cellular Biochemistry. 96 (3): 506–21. doi:10.1002/jcb.20519. PMID 15988757.
- ^ a b c Dame RT, Tark-Dame M (June 2016). "Bacterial chromatin: converging views at different scales". Current Opinion in Cell Biology. 40: 60–65. doi:10.1016/j.ceb.2016.02.015. PMID 26942688.
- ^ a b c Kleckner N, Fisher JK, Stouf M, White MA, Bates D, Witz G (December 2014). "The bacterial nucleoid: nature, dynamics and sister segregation". Current Opinion in Microbiology. 22: 127–37. doi:10.1016/j.mib.2014.10.001. PMC 4359759. PMID 25460806.
- ^ a b Bloomfield VA (1997). "DNA condensation by multivalent cations". Biopolymers. 44 (3): 269–82. doi:10.1002/(SICI)1097-0282(1997)44:3<269::AID-BIP6>3.0.CO;2-T. PMID 9591479.
- ^ a b c d Trun NJ, Marko JF (1998). "Architecture of a bacterial chromosome" (PDF). American Society of Microbiology News. 64 (5): 276–283.
- ^ Surovtsev, Ivan V.; Jacobs-Wagner, Christine (March 2018). "Subcellular Organization: A Critical Feature of Bacterial Cell Replication". Cell. 172 (6): 1271–1293. doi:10.1016/j.cell.2018.01.014. PMC 5870143. PMID 29522747.
- ^ a b Stonington OG, Pettijohn DE (January 1971). "The folded genome of Escherichia coli isolated in a protein-DNA-RNA complex". Proceedings of the National Academy of Sciences of the United States of America. 68 (1): 6–9. Bibcode:1971PNAS...68....6S. doi:10.1073/pnas.68.1.6. PMC 391088. PMID 4924971.
- ^ Worcel A, Burgi E (November 1972). "On the structure of the folded chromosome of Escherichia coli". Journal of Molecular Biology. 71 (2): 127–47. doi:10.1016/0022-2836(72)90342-7. PMID 4564477.
- ^ a b c d e Kano Y, Goshima N, Wada M, Imamoto F (1989). "Participation of hup gene product in replicative transposition of Mu phage in Escherichia coli". Gene. 76 (2): 353–8. doi:10.1016/0378-1119(89)90175-3. PMID 2666261.
- ^ a b c d e Ogura T, Niki H, Kano Y, Imamoto F, Hiraga S (January 1990). "Maintenance of plasmids in HU and IHF mutants of Escherichia coli". Molecular & General Genetics. 220 (2): 197–203. doi:10.1007/bf00260482. PMID 2183003. S2CID 10701528.
- ^ a b c d e Hwang DS, Kornberg A (November 1992). "Opening of the replication origin of Escherichia coli by DnaA protein with protein HU or IHF". The Journal of Biological Chemistry. 267 (32): 23083–6. doi:10.1016/S0021-9258(18)50059-4. PMID 1429655.
- ^ a b Valens M, Penaud S, Rossignol M, Cornet F, Boccard F (October 2004). "Macrodomain organization of the Escherichia coli chromosome". The EMBO Journal. 23 (21): 4330–41. doi:10.1038/sj.emboj.7600434. PMC 524398. PMID 15470498.
- ^ a b c d e f g Fisher JK, Bourniquel A, Witz G, Weiner B, Prentiss M, Kleckner N (May 2013). "Four-dimensional imaging of E. coli nucleoid organization and dynamics in living cells". Cell. 153 (4): 882–95. doi:10.1016/j.cell.2013.04.006. PMC 3670778. PMID 23623305.
- ^ a b c d e Le Gall A, Cattoni DI, Guilhas B, Mathieu-Demazière C, Oudjedi L, Fiche JB, et al. (July 2016). "Bacterial partition complexes segregate within the volume of the nucleoid". Nature Communications. 7: 12107. Bibcode:2016NatCo...712107L. doi:10.1038/ncomms12107. PMC 4935973. PMID 27377966.
- ^ a b c Hadizadeh Yazdi N, Guet CC, Johnson RC, Marko JF (December 2012). "Variation of the folding and dynamics of the Escherichia coli chromosome with growth conditions". Molecular Microbiology. 86 (6): 1318–33. doi:10.1111/mmi.12071. PMC 3524407. PMID 23078205.
- ^ Olins AL, Olins DE (January 1974). "Spheroid chromatin units (v bodies)". Science. 183 (4122): 330–2. Bibcode:1974Sci...183..330O. doi:10.1126/science.183.4122.330. PMID 4128918. S2CID 83480762.
- ^ a b Luger K, Mäder AW, Richmond RK, Sargent DF, Richmond TJ (September 1997). "Crystal structure of the nucleosome core particle at 2.8 A resolution". Nature. 389 (6648): 251–60. Bibcode:1997Natur.389..251L. doi:10.1038/38444. PMID 9305837. S2CID 4328827.
- ^ a b Khorasanizadeh S (January 2004). "The nucleosome: from genomic organization to genomic regulation". Cell. 116 (2): 259–72. doi:10.1016/s0092-8674(04)00044-3. PMID 14744436. S2CID 15504162.
- ^ Talukder A, Ishihama A (September 2015). "Growth phase dependent changes in the structure and protein composition of nucleoid in Escherichia coli". Science China Life Sciences. 58 (9): 902–11. doi:10.1007/s11427-015-4898-0. PMID 26208826.
- ^ a b c d e f Azam TA, Ishihama A (November 1999). "Twelve species of the nucleoid-associated protein from Escherichia coli. Sequence recognition specificity and DNA binding affinity". The Journal of Biological Chemistry. 274 (46): 33105–13. doi:10.1074/jbc.274.46.33105. PMID 10551881. S2CID 9807664.
- ^ a b c d e f g Ali Azam T, Iwata A, Nishimura A, Ueda S, Ishihama A (October 1999). "Growth phase-dependent variation in protein composition of the Escherichia coli nucleoid". Journal of Bacteriology. 181 (20): 6361–70. doi:10.1128/JB.181.20.6361-6370.1999. PMC 103771. PMID 10515926.
- ^ a b Swinger KK, Lemberg KM, Zhang Y, Rice PA (July 2003). "Flexible DNA bending in HU-DNA cocrystal structures". The EMBO Journal. 22 (14): 3749–60. doi:10.1093/emboj/cdg351. PMC 165621. PMID 12853489.
- ^ a b Guo F, Adhya S (March 2007). "Spiral structure of Escherichia coli HUalphabeta provides foundation for DNA supercoiling". Proceedings of the National Academy of Sciences of the United States of America. 104 (11): 4309–14. Bibcode:2007PNAS..104.4309G. doi:10.1073/pnas.0611686104. PMC 1838598. PMID 17360520.
- ^ a b c Pinson V, Takahashi M, Rouviere-Yaniv J (April 1999). "Differential binding of the Escherichia coli HU, homodimeric forms and heterodimeric form to linear, gapped and cruciform DNA". Journal of Molecular Biology. 287 (3): 485–97. doi:10.1006/jmbi.1999.2631. PMID 10092454.
- ^ Craig NL, Nash HA (December 1984). "E. coli integration host factor binds to specific sites in DNA". Cell. 39 (3 Pt 2): 707–16. doi:10.1016/0092-8674(84)90478-1. PMID 6096022. S2CID 26758055.
- ^ a b Ou HD, Phan S, Deerinck TJ, Thor A, Ellisman MH, O'Shea CC (July 2017). "ChromEMT: Visualizing 3D chromatin structure and compaction in interphase and mitotic cells". Science. 357 (6349): eaag0025. doi:10.1126/science.aag0025. PMC 5646685. PMID 28751582.
- ^ a b c Lang B, Blot N, Bouffartigues E, Buckle M, Geertz M, Gualerzi CO, et al. (September 2007). "High-affinity DNA binding sites for H-NS provide a molecular basis for selective silencing within proteobacterial genomes". Nucleic Acids Research. 35 (18): 6330–7. doi:10.1093/nar/gkm712. PMC 2094087. PMID 17881364.
- ^ a b c Gulvady R, Gao Y, Kenney LJ, Yan J (November 2018). "A single molecule analysis of H-NS uncouples DNA binding affinity from DNA specificity". Nucleic Acids Research. 46 (19): 10216–10224. doi:10.1093/nar/gky826. PMC 6212787. PMID 30239908.
- ^ a b c Shao Y, Feldman-Cohen LS, Osuna R (February 2008). "Functional characterization of the Escherichia coli Fis-DNA binding sequence". Journal of Molecular Biology. 376 (3): 771–85. doi:10.1016/j.jmb.2007.11.101. PMC 2292415. PMID 18178221.
- ^ a b c d Stella S, Cascio D, Johnson RC (April 2010). "The shape of the DNA minor groove directs binding by the DNA-bending protein Fis". Genes & Development. 24 (8): 814–26. doi:10.1101/gad.1900610. PMC 2854395. PMID 20395367.
- ^ Narayan K, Subramaniam S (November 2015). "Focused ion beams in biology". Nature Methods. 12 (11): 1021–31. doi:10.1038/nmeth.3623. PMC 6993138. PMID 26513553.
- ^ a b c d Mercier R, Petit MA, Schbath S, Robin S, El Karoui M, Boccard F, Espéli O (October 2008). "The MatP/matS site-specific system organizes the terminus region of the E. coli chromosome into a macrodomain" (PDF). Cell. 135 (3): 475–85. doi:10.1016/j.cell.2008.08.031. PMID 18984159. S2CID 3582710.
- ^ Rouvière-Yaniv J, Gros F (September 1975). "Characterization of a novel, low-molecular-weight DNA-binding protein from Escherichia coli". Proceedings of the National Academy of Sciences of the United States of America. 72 (9): 3428–32. Bibcode:1975PNAS...72.3428R. doi:10.1073/pnas.72.9.3428. PMC 433007. PMID 1103148.
- ^ Suryanarayana T, Subramanian AR (September 1978). "Specific association of two homologous DNA-binding proteins to the native 30-S ribosomal subunits of Escherichia coli". Biochimica et Biophysica Acta (BBA) - Nucleic Acids and Protein Synthesis. 520 (2): 342–57. doi:10.1016/0005-2787(78)90232-0. PMID 213117.
- ^ Mende L, Timm B, Subramanian R (December 1978). "Primary structures of two homologous ribosome-associated DNA-binding proteins of Escherichia coli". FEBS Letters. 96 (2): 395–8. doi:10.1016/0014-5793(78)80446-3. PMID 215461. S2CID 39245157.
- ^ Megraw TL, Chae CB (June 1993). "Functional complementarity between the HMG1-like yeast mitochondrial histone HM and the bacterial histone-like protein HU". The Journal of Biological Chemistry. 268 (17): 12758–63. doi:10.1016/S0021-9258(18)31453-4. PMID 8509411.
- ^ Paull TT, Johnson RC (April 1995). "DNA looping by Saccharomyces cerevisiae high mobility group proteins NHP6A/B. Consequences for nucleoprotein complex assembly and chromatin condensation". The Journal of Biological Chemistry. 270 (15): 8744–54. doi:10.1074/jbc.270.15.8744. PMID 7721780.
- ^ Kamashev D, Rouviere-Yaniv J (December 2000). "The histone-like protein HU binds specifically to DNA recombination and repair intermediates". The EMBO Journal. 19 (23): 6527–35. doi:10.1093/emboj/19.23.6527. PMC 305869. PMID 11101525.
- ^ Shindo H, Furubayashi A, Shimizu M, Miyake M, Imamoto F (April 1992). "Preferential binding of E.coli histone-like protein HU alpha to negatively supercoiled DNA". Nucleic Acids Research. 20 (7): 1553–8. doi:10.1093/nar/20.7.1553. PMC 312237. PMID 1579448.
- ^ Pontiggia A, Negri A, Beltrame M, Bianchi ME (February 1993). "Protein HU binds specifically to kinked DNA". Molecular Microbiology. 7 (3): 343–50. doi:10.1111/j.1365-2958.1993.tb01126.x. PMID 8459763. S2CID 39362449.
- ^ Bonnefoy E, Takahashi M, Yaniv JR (September 1994). "DNA-binding parameters of the HU protein of Escherichia coli to cruciform DNA". Journal of Molecular Biology. 242 (2): 116–29. doi:10.1006/jmbi.1994.1563. PMID 8089835.
- ^ Castaing B, Zelwer C, Laval J, Boiteux S (April 1995). "HU protein of Escherichia coli binds specifically to DNA that contains single-strand breaks or gaps". The Journal of Biological Chemistry. 270 (17): 10291–6. doi:10.1074/jbc.270.17.10291. PMID 7730334.
- ^ Lyubchenko YL, Shlyakhtenko LS, Aki T, Adhya S (February 1997). "Atomic force microscopic demonstration of DNA looping by GalR and HU". Nucleic Acids Research. 25 (4): 873–6. doi:10.1093/nar/25.4.873. PMC 146491. PMID 9016640.
- ^ Swinger KK, Rice PA (January 2007). "Structure-based analysis of HU-DNA binding". Journal of Molecular Biology. 365 (4): 1005–16. doi:10.1016/j.jmb.2006.10.024. PMC 1945228. PMID 17097674.
- ^ a b c d e f g Hammel M, Amlanjyoti D, Reyes FE, Chen JH, Parpana R, Tang HY, et al. (July 2016). "HU multimerization shift controls nucleoid compaction". Science Advances. 2 (7): e1600650. Bibcode:2016SciA....2E0650H. doi:10.1126/sciadv.1600650. PMC 4966879. PMID 27482541.
- ^ a b c d e Prieto AI, Kahramanoglou C, Ali RM, Fraser GM, Seshasayee AS, Luscombe NM (April 2012). "Genomic analysis of DNA binding and gene regulation by homologous nucleoid-associated proteins IHF and HU in Escherichia coli K12". Nucleic Acids Research. 40 (8): 3524–37. doi:10.1093/nar/gkr1236. PMC 3333857. PMID 22180530.
- ^ a b c d e f Macvanin M, Edgar R, Cui F, Trostel A, Zhurkin V, Adhya S (November 2012). "Noncoding RNAs binding to the nucleoid protein HU in Escherichia coli". Journal of Bacteriology. 194 (22): 6046–55. doi:10.1128/JB.00961-12. PMC 3486375. PMID 22942248.
- ^ a b c d e van Noort J, Verbrugge S, Goosen N, Dekker C, Dame RT (May 2004). "Dual architectural roles of HU: formation of flexible hinges and rigid filaments". Proceedings of the National Academy of Sciences of the United States of America. 101 (18): 6969–74. Bibcode:2004PNAS..101.6969V. doi:10.1073/pnas.0308230101. PMC 406450. PMID 15118104.
- ^ Sarkar R, Rybenkov VV (2016-12-06). "A Guide to Magnetic Tweezers and Their Applications". Frontiers in Physics. 4: 48. Bibcode:2016FrP.....4...48S. doi:10.3389/fphy.2016.00048. S2CID 44183628.
- ^ a b c d Kahramanoglou C, Seshasayee AS, Prieto AI, Ibberson D, Schmidt S, Zimmermann J, et al. (March 2011). "Direct and indirect effects of H-NS and Fis on global gene expression control in Escherichia coli". Nucleic Acids Research. 39 (6): 2073–91. doi:10.1093/nar/gkq934. PMC 3064808. PMID 21097887.
- ^ a b Rice PA, Yang S, Mizuuchi K, Nash HA (December 1996). "Crystal structure of an IHF-DNA complex: a protein-induced DNA U-turn". Cell. 87 (7): 1295–306. doi:10.1016/s0092-8674(00)81824-3. PMID 8980235. S2CID 9291279.
- ^ Murtin C, Engelhorn M, Geiselmann J, Boccard F (December 1998). "A quantitative UV laser footprinting analysis of the interaction of IHF with specific binding sites: re-evaluation of the effective concentration of IHF in the cell". Journal of Molecular Biology. 284 (4): 949–61. doi:10.1006/jmbi.1998.2256. PMID 9837718.
- ^ Ditto MD, Roberts D, Weisberg RA (June 1994). "Growth phase variation of integration host factor level in Escherichia coli". Journal of Bacteriology. 176 (12): 3738–48. doi:10.1128/jb.176.12.3738-3748.1994. PMC 205563. PMID 8206852.
- ^ a b c Lin J, Chen H, Dröge P, Yan J (2012). "Physical organization of DNA by multiple non-specific DNA-binding modes of integration host factor (IHF)". PLOS ONE. 7 (11): e49885. Bibcode:2012PLoSO...749885L. doi:10.1371/journal.pone.0049885. PMC 3498176. PMID 23166787.
- ^ Jacquet M, Cukier-Kahn R, Pla J, Gros F (December 1971). "A thermostable protein factor acting on in vitro DNA transcription". Biochemical and Biophysical Research Communications. 45 (6): 1597–607. doi:10.1016/0006-291x(71)90204-x. PMID 4942735.
- ^ Cukier-Kahn R, Jacquet M, Gros F (December 1972). "Two heat-resistant, low molecular weight proteins from Escherichia coli that stimulate DNA-directed RNA synthesis". Proceedings of the National Academy of Sciences of the United States of America. 69 (12): 3643–7. Bibcode:1972PNAS...69.3643C. doi:10.1073/pnas.69.12.3643. PMC 389839. PMID 4566454.
- ^ Spassky A, Buc HC (November 1977). "Physico-chemical properties of a DNA binding protein: Escherichia coli factor H1". European Journal of Biochemistry. 81 (1): 79–90. doi:10.1111/j.1432-1033.1977.tb11929.x. PMID 338303.
- ^ Varshavsky AJ, Nedospasov SA, Bakayev VV, Bakayeva TG, Georgiev GP (August 1977). "Histone-like proteins in the purified Escherichia coli deoxyribonucleoprotein". Nucleic Acids Research. 4 (8): 2725–45. doi:10.1093/nar/4.8.2725. PMC 342604. PMID 333393.
- ^ Falconi M, Gualtieri MT, La Teana A, Losso MA, Pon CL (May 1988). "Proteins from the prokaryotic nucleoid: primary and quaternary structure of the 15-kD Escherichia coli DNA binding protein H-NS". Molecular Microbiology. 2 (3): 323–9. doi:10.1111/j.1365-2958.1988.tb00035.x. PMID 3135462. S2CID 36215353.
- ^ Ueguchi C, Suzuki T, Yoshida T, Tanaka K, Mizuno T (October 1996). "Systematic mutational analysis revealing the functional domain organization of Escherichia coli nucleoid protein H-NS". Journal of Molecular Biology. 263 (2): 149–62. doi:10.1006/jmbi.1996.0566. PMID 8913298.
- ^ a b Rimsky S, Zuber F, Buckle M, Buc H (December 2001). "A molecular mechanism for the repression of transcription by the H-NS protein". Molecular Microbiology. 42 (5): 1311–23. doi:10.1046/j.1365-2958.2001.02706.x. PMID 11886561. S2CID 32623528.
- ^ Bouffartigues E, Buckle M, Badaut C, Travers A, Rimsky S (May 2007). "H-NS cooperative binding to high-affinity sites in a regulatory element results in transcriptional silencing". Nature Structural & Molecular Biology. 14 (5): 441–8. doi:10.1038/nsmb1233. PMID 17435766. S2CID 43768346.
- ^ Dame RT, Wyman C, Goosen N (September 2000). "H-NS mediated compaction of DNA visualised by atomic force microscopy". Nucleic Acids Research. 28 (18): 3504–10. doi:10.1093/nar/28.18.3504. PMC 110753. PMID 10982869.
- ^ Amit R, Oppenheim AB, Stavans J (April 2003). "Increased bending rigidity of single DNA molecules by H-NS, a temperature and osmolarity sensor". Biophysical Journal. 84 (4): 2467–73. Bibcode:2003BpJ....84.2467A. doi:10.1016/S0006-3495(03)75051-6. PMC 1302812. PMID 12668454.
- ^ Dame RT, Noom MC, Wuite GJ (November 2006). "Bacterial chromatin organization by H-NS protein unravelled using dual DNA manipulation". Nature. 444 (7117): 387–90. Bibcode:2006Natur.444..387D. doi:10.1038/nature05283. PMID 17108966. S2CID 4314858.
- ^ a b c d e f Liu Y, Chen H, Kenney LJ, Yan J (February 2010). "A divalent switch drives H-NS/DNA-binding conformations between stiffening and bridging modes". Genes & Development. 24 (4): 339–44. doi:10.1101/gad.1883510. PMC 2816733. PMID 20159954.
- ^ van der Valk RA, Vreede J, Qin L, Moolenaar GF, Hofmann A, Goosen N, Dame RT (September 2017). "Mechanism of environmentally driven conformational changes that modulate H-NS DNA-bridging activity". eLife. 6. doi:10.7554/eLife.27369. PMC 5647153. PMID 28949292.
- ^ Yamada H, Muramatsu S, Mizuno T (September 1990). "An Escherichia coli protein that preferentially binds to sharply curved DNA". Journal of Biochemistry. 108 (3): 420–5. doi:10.1093/oxfordjournals.jbchem.a123216. PMID 2126011.
- ^ Martin-Orozco N, Touret N, Zaharik ML, Park E, Kopelman R, Miller S, et al. (January 2006). "Visualization of vacuolar acidification-induced transcription of genes of pathogens inside macrophages". Molecular Biology of the Cell. 17 (1): 498–510. doi:10.1091/mbc.e04-12-1096. PMC 1345685. PMID 16251362.
- ^ Winardhi RS, Yan J, Kenney LJ (October 2015). "H-NS Regulates Gene Expression and Compacts the Nucleoid: Insights from Single-Molecule Experiments". Biophysical Journal. 109 (7): 1321–9. Bibcode:2015BpJ...109.1321W. doi:10.1016/j.bpj.2015.08.016. PMC 4601063. PMID 26445432.
- ^ Walthers D, Li Y, Liu Y, Anand G, Yan J, Kenney LJ (January 2011). "Salmonella enterica response regulator SsrB relieves H-NS silencing by displacing H-NS bound in polymerization mode and directly activates transcription". The Journal of Biological Chemistry. 286 (3): 1895–902. doi:10.1074/jbc.M110.164962. PMC 3023485. PMID 21059643.
- ^ Gao Y, Foo YH, Winardhi RS, Tang Q, Yan J, Kenney LJ (November 2017). "Charged residues in the H-NS linker drive DNA binding and gene silencing in single cells". Proceedings of the National Academy of Sciences of the United States of America. 114 (47): 12560–12565. Bibcode:2017PNAS..11412560G. doi:10.1073/pnas.1716721114. PMC 5703333. PMID 29109287.
- ^ Hancock SP, Stella S, Cascio D, Johnson RC (2016). "DNA Sequence Determinants Controlling Affinity, Stability and Shape of DNA Complexes Bound by the Nucleoid Protein Fis". PLOS ONE. 11 (3): e0150189. Bibcode:2016PLoSO..1150189H. doi:10.1371/journal.pone.0150189. PMC 4784862. PMID 26959646.
- ^ Kostrewa D, Granzin J, Koch C, Choe HW, Raghunathan S, Wolf W, et al. (January 1991). "Three-dimensional structure of the E. coli DNA-binding protein FIS". Nature. 349 (6305): 178–80. Bibcode:1991Natur.349..178K. doi:10.1038/349178a0. PMID 1986310. S2CID 4318862.
- ^ Kostrewa D, Granzin J, Stock D, Choe HW, Labahn J, Saenger W (July 1992). "Crystal structure of the factor for inversion stimulation FIS at 2.0 A resolution". Journal of Molecular Biology. 226 (1): 209–26. doi:10.1016/0022-2836(92)90134-6. PMID 1619650.
- ^ Cho BK, Knight EM, Barrett CL, Palsson BØ (June 2008). "Genome-wide analysis of Fis binding in Escherichia coli indicates a causative role for A-/AT-tracts". Genome Research. 18 (6): 900–10. doi:10.1101/gr.070276.107. PMC 2413157. PMID 18340041.
- ^ a b Travers A, Muskhelishvili G (June 1998). "DNA microloops and microdomains: a general mechanism for transcription activation by torsional transmission". Journal of Molecular Biology. 279 (5): 1027–43. doi:10.1006/jmbi.1998.1834. PMID 9642081.
- ^ Skoko D, Yan J, Johnson RC, Marko JF (November 2005). "Low-force DNA condensation and discontinuous high-force decondensation reveal a loop-stabilizing function of the protein Fis". Physical Review Letters. 95 (20): 208101. Bibcode:2005PhRvL..95t8101S. doi:10.1103/PhysRevLett.95.208101. PMID 16384101. S2CID 37405026.
- ^ a b Skoko D, Yoo D, Bai H, Schnurr B, Yan J, McLeod SM, et al. (December 2006). "Mechanism of chromosome compaction and looping by the Escherichia coli nucleoid protein Fis". Journal of Molecular Biology. 364 (4): 777–98. doi:10.1016/j.jmb.2006.09.043. PMC 1988847. PMID 17045294.
- ^ Johnson RC, Simon MI (July 1985). "Hin-mediated site-specific recombination requires two 26 bp recombination sites and a 60 bp recombinational enhancer". Cell. 41 (3): 781–91. doi:10.1016/s0092-8674(85)80059-3. PMID 2988787. S2CID 34572809.
- ^ a b Kahmann R, Rudt F, Koch C, Mertens G (July 1985). "G inversion in bacteriophage Mu DNA is stimulated by a site within the invertase gene and a host factor". Cell. 41 (3): 771–80. doi:10.1016/S0092-8674(85)80058-1. PMID 3159478.
- ^ Pettijohn DE, Hecht R (1974). "RNA molecules bound to the folded bacterial genome stabilize DNA folds and segregate domains of supercoiling". Cold Spring Harbor Symposia on Quantitative Biology. 38: 31–41. doi:10.1101/sqb.1974.038.01.006. PMID 4598638.
- ^ a b c Ohniwa RL, Morikawa K, Takeshita SL, Kim J, Ohta T, Wada C, Takeyasu K (October 2007). "Transcription-coupled nucleoid architecture in bacteria". Genes to Cells. 12 (10): 1141–52. doi:10.1111/j.1365-2443.2007.01125.x. PMID 17903174.
- ^ a b c Balandina A, Kamashev D, Rouviere-Yaniv J (August 2002). "The bacterial histone-like protein HU specifically recognizes similar structures in all nucleic acids. DNA, RNA, and their hybrids". The Journal of Biological Chemistry. 277 (31): 27622–8. doi:10.1074/jbc.M201978200. PMID 12006568.
- ^ Balandina A, Claret L, Hengge-Aronis R, Rouviere-Yaniv J (February 2001). "The Escherichia coli histone-like protein HU regulates rpoS translation". Molecular Microbiology. 39 (4): 1069–79. doi:10.1046/j.1365-2958.2001.02305.x. PMID 11251825.
- ^ a b Qian Z, Macvanin M, Dimitriadis EK, He X, Zhurkin V, Adhya S (August 2015). "A New Noncoding RNA Arranges Bacterial Chromosome Organization". mBio. 6 (4): e00998-15. doi:10.1128/mBio.00998-15. PMC 4550694. PMID 26307168.
- ^ Qian Z, Zhurkin VB, Adhya S (November 2017). "Escherichia coli". Proceedings of the National Academy of Sciences of the United States of America. 114 (46): 12225–12230. doi:10.1073/pnas.1711285114. PMC 5699063. PMID 29087325.
- ^ Bauer WR, Crick FH, White JH (July 1980). "Supercoiled DNA". Scientific American. 243 (1): 100–13. PMID 6256851.
- ^ "Linking coefficient". www.encyclopediaofmath.org. Encyclopedia of Mathematics. Retrieved 2020-02-22.
- ^ a b c Bates AD, Maxwell A (2005). DNA topology (2nd ed.). Oxford: Oxford University Press. ISBN 978-0-19-856709-7. OCLC 56793994.
- ^ a b c Vologodskii, Aleksander Vadimovich. (1992). Topology and physics of circular DNA. CRC Press. ISBN 0-8493-4228-7. OCLC 635611152.
- ^ Sinden RR (2006). DNA structure and function. Elsevier. ISBN 978-0-12-645750-6. OCLC 698461259.
- ^ a b Sinden RR, Carlson JO, Pettijohn DE (October 1980). "Torsional tension in the DNA double helix measured with trimethylpsoralen in living E. coli cells: analogous measurements in insect and human cells". Cell. 21 (3): 773–83. doi:10.1016/0092-8674(80)90440-7. PMID 6254668. S2CID 2503376.
- ^ Griffith JD (February 1976). "Visualization of prokaryotic DNA in a regularly condensed chromatin-like fiber". Proceedings of the National Academy of Sciences of the United States of America. 73 (2): 563–7. Bibcode:1976PNAS...73..563G. doi:10.1073/pnas.73.2.563. PMC 335950. PMID 1108025.
- ^ a b c Postow L, Hardy CD, Arsuaga J, Cozzarelli NR (July 2004). "Topological domain structure of the Escherichia coli chromosome". Genes & Development. 18 (14): 1766–79. doi:10.1101/gad.1207504. PMC 478196. PMID 15256503.
- ^ a b Bliska JB, Cozzarelli NR (March 1987). "Use of site-specific recombination as a probe of DNA structure and metabolism in vivo". Journal of Molecular Biology. 194 (2): 205–18. doi:10.1016/0022-2836(87)90369-x. PMID 3039150.
- ^ Holmes VF, Cozzarelli NR (February 2000). "Closing the ring: links between SMC proteins and chromosome partitioning, condensation, and supercoiling". Proceedings of the National Academy of Sciences of the United States of America. 97 (4): 1322–4. Bibcode:2000PNAS...97.1322H. doi:10.1073/pnas.040576797. PMC 34294. PMID 10677457.
- ^ Champoux JJ (2001). "DNA topoisomerases: structure, function, and mechanism". Annual Review of Biochemistry. 70: 369–413. doi:10.1146/annurev.biochem.70.1.369. PMID 11395412. S2CID 18144189.
- ^ Gellert M, Mizuuchi K, O'Dea MH, Nash HA (November 1976). "DNA gyrase: an enzyme that introduces superhelical turns into DNA". Proceedings of the National Academy of Sciences of the United States of America. 73 (11): 3872–6. Bibcode:1976PNAS...73.3872G. doi:10.1073/pnas.73.11.3872. PMC 431247. PMID 186775.
- ^ a b c Depew RE, Liu LF, Wang JC (January 1978). "Interaction between DNA and Escherichia coli protein omega. Formation of a complex between single-stranded DNA and omega protein". The Journal of Biological Chemistry. 253 (2): 511–8. doi:10.1016/S0021-9258(17)38239-X. PMID 338610.
- ^ Kirkegaard K, Wang JC (October 1978). "Escherichia coli DNA topoisomerase I catalyzed linking of single-stranded rings of complementary base sequences". Nucleic Acids Research. 5 (10): 3811–20. doi:10.1093/nar/5.10.3811. PMC 342711. PMID 214763.
- ^ a b Raji A, Zabel DJ, Laufer CS, Depew RE (June 1985). "Genetic analysis of mutations that compensate for loss of Escherichia coli DNA topoisomerase I". Journal of Bacteriology. 162 (3): 1173–9. doi:10.1128/JB.162.3.1173-1179.1985. PMC 215900. PMID 2987184.
- ^ Dean F, Krasnow MA, Otter R, Matzuk MM, Spengler SJ, Cozzarelli NR (1983). "Escherichia coli type-1 topoisomerases: identification, mechanism, and role in recombination". Cold Spring Harbor Symposia on Quantitative Biology. 47 (2): 769–77. doi:10.1101/sqb.1983.047.01.088. PMID 6305585.
- ^ Zechiedrich EL, Khodursky AB, Bachellier S, Schneider R, Chen D, Lilley DM, Cozzarelli NR (March 2000). "Roles of topoisomerases in maintaining steady-state DNA supercoiling in Escherichia coli". The Journal of Biological Chemistry. 275 (11): 8103–13. doi:10.1074/jbc.275.11.8103. PMID 10713132.
- ^ Kato J, Nishimura Y, Imamura R, Niki H, Hiraga S, Suzuki H (October 1990). "New topoisomerase essential for chromosome segregation in E. coli". Cell. 63 (2): 393–404. doi:10.1016/0092-8674(90)90172-b. PMID 2170028. S2CID 42122316.
- ^ Liu LF, Wang JC (October 1987). "Supercoiling of the DNA template during transcription". Proceedings of the National Academy of Sciences of the United States of America. 84 (20): 7024–7. Bibcode:1987PNAS...84.7024L. doi:10.1073/pnas.84.20.7024. PMC 299221. PMID 2823250.
- ^ a b Kouzine F, Liu J, Sanford S, Chung HJ, Levens D (November 2004). "The dynamic response of upstream DNA to transcription-generated torsional stress". Nature Structural & Molecular Biology. 11 (11): 1092–100. doi:10.1038/nsmb848. PMID 15502847. S2CID 28956216.
- ^ Rouvière-Yaniv J, Yaniv M, Germond JE (June 1979). "E. coli DNA binding protein HU forms nucleosomelike structure with circular double-stranded DNA". Cell. 17 (2): 265–74. doi:10.1016/0092-8674(79)90152-1. PMID 222478. S2CID 28092421.
- ^ Broyles SS, Pettijohn DE (January 1986). "Interaction of the Escherichia coli HU protein with DNA. Evidence for formation of nucleosome-like structures with altered DNA helical pitch". Journal of Molecular Biology. 187 (1): 47–60. doi:10.1016/0022-2836(86)90405-5. PMID 3514923.
- ^ Kundukad B, Cong P, van der Maarel JR, Doyle PS (September 2013). "Time-dependent bending rigidity and helical twist of DNA by rearrangement of bound HU protein". Nucleic Acids Research. 41 (17): 8280–8. doi:10.1093/nar/gkt593. PMC 3783175. PMID 23828037.
- ^ Tupper AE, Owen-Hughes TA, Ussery DW, Santos DS, Ferguson DJ, Sidebotham JM, et al. (January 1994). "The chromatin-associated protein H-NS alters DNA topology in vitro". The EMBO Journal. 13 (1): 258–68. doi:10.1002/j.1460-2075.1994.tb06256.x. PMC 394800. PMID 8306968.
- ^ a b Schneider R, Lurz R, Lüder G, Tolksdorf C, Travers A, Muskhelishvili G (December 2001). "An architectural role of the Escherichia coli chromatin protein FIS in organising DNA". Nucleic Acids Research. 29 (24): 5107–14. doi:10.1093/nar/29.24.5107. PMC 97572. PMID 11812843.
- ^ Schneider R, Travers A, Kutateladze T, Muskhelishvili G (December 1999). "A DNA architectural protein couples cellular physiology and DNA topology in Escherichia coli". Molecular Microbiology. 34 (5): 953–64. doi:10.1046/j.1365-2958.1999.01656.x. PMID 10594821.
- ^ a b Bensaid A, Almeida A, Drlica K, Rouviere-Yaniv J (February 1996). "Cross-talk between topoisomerase I and HU in Escherichia coli". Journal of Molecular Biology. 256 (2): 292–300. doi:10.1006/jmbi.1996.0086. PMID 8594197.
- ^ Malik M, Bensaid A, Rouviere-Yaniv J, Drlica K (February 1996). "Histone-like protein HU and bacterial DNA topology: suppression of an HU deficiency by gyrase mutations". Journal of Molecular Biology. 256 (1): 66–76. doi:10.1006/jmbi.1996.0068. PMID 8609614.
- ^ a b Marians KJ (July 1987). "DNA gyrase-catalyzed decatenation of multiply linked DNA dimers". The Journal of Biological Chemistry. 262 (21): 10362–8. doi:10.1016/S0021-9258(18)61121-4. PMID 3038875.
- ^ a b c Sinden RR, Pettijohn DE (January 1981). "Chromosomes in living Escherichia coli cells are segregated into domains of supercoiling". Proceedings of the National Academy of Sciences of the United States of America. 78 (1): 224–8. Bibcode:1981PNAS...78..224S. doi:10.1073/pnas.78.1.224. PMC 319024. PMID 6165987.
- ^ Higgins NP, Yang X, Fu Q, Roth JR (May 1996). "Surveying a supercoil domain by using the gamma delta resolution system in Salmonella typhimurium". Journal of Bacteriology. 178 (10): 2825–35. doi:10.1128/jb.178.10.2825-2835.1996. PMC 178017. PMID 8631670.
- ^ Yan Y, Ding Y, Leng F, Dunlap D, Finzi L (May 2018). "Protein-mediated loops in supercoiled DNA create large topological domains". Nucleic Acids Research. 46 (9): 4417–4424. doi:10.1093/nar/gky153. PMC 5961096. PMID 29538766.
- ^ Leng F, Chen B, Dunlap DD (December 2011). "Dividing a supercoiled DNA molecule into two independent topological domains". Proceedings of the National Academy of Sciences of the United States of America. 108 (50): 19973–8. Bibcode:2011PNAS..10819973L. doi:10.1073/pnas.1109854108. PMC 3250177. PMID 22123985.
- ^ Moulin L, Rahmouni AR, Boccard F (January 2005). "Topological insulators inhibit diffusion of transcription-induced positive supercoils in the chromosome of Escherichia coli". Molecular Microbiology. 55 (2): 601–10. doi:10.1111/j.1365-2958.2004.04411.x. PMID 15659173.
- ^ Dimri GP, Rudd KE, Morgan MK, Bayat H, Ames GF (July 1992). "Physical mapping of repetitive extragenic palindromic sequences in Escherichia coli and phylogenetic distribution among Escherichia coli strains and other enteric bacteria". Journal of Bacteriology. 174 (14): 4583–93. doi:10.1128/jb.174.14.4583-4593.1992. PMC 206253. PMID 1624447.
- ^ Deng S, Stein RA, Higgins NP (March 2004). "Transcription-induced barriers to supercoil diffusion in the Salmonella typhimurium chromosome". Proceedings of the National Academy of Sciences of the United States of America. 101 (10): 3398–403. Bibcode:2004PNAS..101.3398D. doi:10.1073/pnas.0307550101. PMC 373473. PMID 14993611.
- ^ Deng S, Stein RA, Higgins NP (September 2005). "Organization of supercoil domains and their reorganization by transcription". Molecular Microbiology. 57 (6): 1511–21. doi:10.1111/j.1365-2958.2005.04796.x. PMC 1382059. PMID 16135220.
- ^ Booker BM, Deng S, Higgins NP (December 2010). "DNA topology of highly transcribed operons in Salmonella enterica serovar Typhimurium". Molecular Microbiology. 78 (6): 1348–64. doi:10.1111/j.1365-2958.2010.07394.x. PMID 21143310.
- ^ a b c d e f g h i j k l m n Lioy VS, Cournac A, Marbouty M, Duigou S, Mozziconacci J, Espéli O, et al. (February 2018). "Multiscale Structuring of the E. coli Chromosome by Nucleoid-Associated and Condensin Proteins". Cell. 172 (4): 771–783.e18. doi:10.1016/j.cell.2017.12.027. PMID 29358050.
- ^ Almirón M, Link AJ, Furlong D, Kolter R (December 1992). "A novel DNA-binding protein with regulatory and protective roles in starved Escherichia coli". Genes & Development. 6 (12B): 2646–54. doi:10.1101/gad.6.12b.2646. PMID 1340475. S2CID 10308477.
- ^ a b Frenkiel-Krispin D, Ben-Avraham I, Englander J, Shimoni E, Wolf SG, Minsky A (January 2004). "Nucleoid restructuring in stationary-state bacteria". Molecular Microbiology. 51 (2): 395–405. doi:10.1046/j.1365-2958.2003.03855.x. PMID 14756781.
- ^ Ball CA, Osuna R, Ferguson KC, Johnson RC (December 1992). "Dramatic changes in Fis levels upon nutrient upshift in Escherichia coli". Journal of Bacteriology. 174 (24): 8043–56. doi:10.1128/jb.174.24.8043-8056.1992. PMC 207543. PMID 1459953.
- ^ Claret L, Rouviere-Yaniv J (October 1997). "Variation in HU composition during growth of Escherichia coli: the heterodimer is required for long term survival". Journal of Molecular Biology. 273 (1): 93–104. doi:10.1006/jmbi.1997.1310. PMID 9367749.
- ^ a b c Badrinarayanan A, Lesterlin C, Reyes-Lamothe R, Sherratt D (September 2012). "The Escherichia coli SMC complex, MukBEF, shapes nucleoid organization independently of DNA replication". Journal of Bacteriology. 194 (17): 4669–76. doi:10.1128/JB.00957-12. PMC 3415497. PMID 22753058.
- ^ a b c Petrushenko ZM, Lai CH, Rybenkov VV (November 2006). "Antagonistic interactions of kleisins and DNA with bacterial Condensin MukB". The Journal of Biological Chemistry. 281 (45): 34208–17. doi:10.1074/jbc.M606723200. PMC 1634889. PMID 16982609.
- ^ a b c Lal A, Dhar A, Trostel A, Kouzine F, Seshasayee AS, Adhya S (March 2016). "Genome scale patterns of supercoiling in a bacterial chromosome". Nature Communications. 7: 11055. Bibcode:2016NatCo...711055L. doi:10.1038/ncomms11055. PMC 4820846. PMID 27025941.
- ^ a b Kar S, Edgar R, Adhya S (November 2005). "Nucleoid remodeling by an altered HU protein: reorganization of the transcription program". Proceedings of the National Academy of Sciences of the United States of America. 102 (45): 16397–402. Bibcode:2005PNAS..10216397K. doi:10.1073/pnas.0508032102. PMC 1283455. PMID 16258062.
- ^ Lim CJ, Lee SY, Kenney LJ, Yan J (2012). "Nucleoprotein filament formation is the structural basis for bacterial protein H-NS gene silencing". Scientific Reports. 2: 509. Bibcode:2012NatSR...2E.509L. doi:10.1038/srep00509. PMC 3396134. PMID 22798986.
- ^ Aki T, Choy HE, Adhya S (February 1996). "Histone-like protein HU as a specific transcriptional regulator: co-factor role in repression of gal transcription by GAL repressor". Genes to Cells. 1 (2): 179–88. doi:10.1046/j.1365-2443.1996.d01-236.x. PMID 9140062.
- ^ Pagel JM, Winkelman JW, Adams CW, Hatfield GW (April 1992). "DNA topology-mediated regulation of transcription initiation from the tandem promoters of the ilvGMEDA operon of Escherichia coli". Journal of Molecular Biology. 224 (4): 919–35. doi:10.1016/0022-2836(92)90460-2. PMID 1569580.
- ^ Parekh BS, Hatfield GW (February 1996). "Transcriptional activation by protein-induced DNA bending: evidence for a DNA structural transmission model". Proceedings of the National Academy of Sciences of the United States of America. 93 (3): 1173–7. Bibcode:1996PNAS...93.1173P. doi:10.1073/pnas.93.3.1173. PMC 40051. PMID 8577735.
- ^ a b c Dorman CJ, Dorman MJ (September 2016). "DNA supercoiling is a fundamental regulatory principle in the control of bacterial gene expression". Biophysical Reviews. 8 (3): 209–220. doi:10.1007/s12551-016-0205-y. PMC 5425793. PMID 28510224.
- ^ Chong S, Chen C, Ge H, Xie XS (July 2014). "Mechanism of transcriptional bursting in bacteria". Cell. 158 (2): 314–326. doi:10.1016/j.cell.2014.05.038. PMC 4105854. PMID 25036631.
- ^ Berger M, Gerganova V, Berger P, Rapiteanu R, Lisicovas V, Dobrindt U (August 2016). "Genes on a Wire: The Nucleoid-Associated Protein HU Insulates Transcription Units in Escherichia coli". Scientific Reports. 6: 31512. Bibcode:2016NatSR...631512B. doi:10.1038/srep31512. PMC 4992867. PMID 27545593.
- ^ Koli P, Sudan S, Fitzgerald D, Adhya S, Kar S (2011). "Conversion of commensal Escherichia coli K-12 to an invasive form via expression of a mutant histone-like protein". mBio. 2 (5). doi:10.1128/mBio.00182-11. PMC 3172693. PMID 21896677.
- ^ a b c Kar S, Choi EJ, Guo F, Dimitriadis EK, Kotova SL, Adhya S (December 2006). "Right-handed DNA supercoiling by an octameric form of histone-like protein HU: modulation of cellular transcription". The Journal of Biological Chemistry. 281 (52): 40144–53. doi:10.1074/jbc.M605576200. PMID 17062578.
- ^ a b Kar S, Choi EJ, Guo F, Dimitriadis EK, Kotova SL, Adhya S (December 2006). "Right-handed DNA supercoiling by an octameric form of histone-like protein HU: modulation of cellular transcription". The Journal of Biological Chemistry. 281 (52): 40144–53. doi:10.1074/jbc.M605576200. PMID 17062578.
- ^ a b Uhlmann F (July 2016). "SMC complexes: from DNA to chromosomes". Nature Reviews Molecular Cell Biology. 17 (7): 399–412. doi:10.1038/nrm.2016.30. PMID 27075410. S2CID 20398243.
- ^ Dekker J, Rippe K, Dekker M, Kleckner N (February 2002). "Capturing chromosome conformation". Science. 295 (5558): 1306–11. Bibcode:2002Sci...295.1306D. doi:10.1126/science.1067799. PMID 11847345. S2CID 3561891.
- ^ Lieberman-Aiden E, van Berkum NL, Williams L, Imakaev M, Ragoczy T, Telling A, et al. (October 2009). "Comprehensive mapping of long-range interactions reveals folding principles of the human genome". Science. 326 (5950): 289–93. Bibcode:2009Sci...326..289L. doi:10.1126/science.1181369. PMC 2858594. PMID 19815776.
- ^ a b c d Le TB, Imakaev MV, Mirny LA, Laub MT (November 2013). "High-resolution mapping of the spatial organization of a bacterial chromosome". Science. 342 (6159): 731–4. Bibcode:2013Sci...342..731L. doi:10.1126/science.1242059. PMC 3927313. PMID 24158908.
- ^ Wang X, Le TB, Lajoie BR, Dekker J, Laub MT, Rudner DZ (August 2015). "Condensin promotes the juxtaposition of DNA flanking its loading site in Bacillus subtilis". Genes & Development. 29 (15): 1661–75. doi:10.1101/gad.265876.115. PMC 4536313. PMID 26253537.
- ^ Dekker J, Heard E (October 2015). "Structural and functional diversity of Topologically Associating Domains". FEBS Letters. 589 (20 Pt A): 2877–84. doi:10.1016/j.febslet.2015.08.044. PMC 4598308. PMID 26348399.
- ^ Niki H, Hiraga S (April 1998). "Polar localization of the replication origin and terminus in Escherichia coli nucleoids during chromosome partitioning". Genes & Development. 12 (7): 1036–45. doi:10.1101/gad.12.7.1036. PMC 316681. PMID 9531540.
- ^ Niki H, Yamaichi Y, Hiraga S (January 2000). "Dynamic organization of chromosomal DNA in Escherichia coli". Genes & Development. 14 (2): 212–23. doi:10.1101/gad.14.2.212. PMC 316355. PMID 10652275.
- ^ Boccard F, Esnault E, Valens M (July 2005). "Spatial arrangement and macrodomain organization of bacterial chromosomes". Molecular Microbiology. 57 (1): 9–16. doi:10.1111/j.1365-2958.2005.04651.x. PMID 15948945.
- ^ Duigou S, Boccard F (May 2017). "Long range chromosome organization in Escherichia coli: The position of the replication origin defines the non-structured regions and the Right and Left macrodomains". PLOS Genetics. 13 (5): e1006758. doi:10.1371/journal.pgen.1006758. PMC 5441646. PMID 28486476.
- ^ a b c Nolivos S, Upton AL, Badrinarayanan A, Müller J, Zawadzka K, Wiktor J, et al. (January 2016). "MatP regulates the coordinated action of topoisomerase IV and MukBEF in chromosome segregation". Nature Communications. 7: 10466. Bibcode:2016NatCo...710466N. doi:10.1038/ncomms10466. PMC 4738335. PMID 26818444.
- ^ a b Dupaigne P, Tonthat NK, Espéli O, Whitfill T, Boccard F, Schumacher MA (November 2012). "Molecular basis for a protein-mediated DNA-bridging mechanism that functions in condensation of the E. coli chromosome". Molecular Cell. 48 (4): 560–71. doi:10.1016/j.molcel.2012.09.009. PMC 7505563. PMID 23084832.
- ^ Wang S, Cosstick R, Gardner JF, Gumport RI (October 1995). "The specific binding of Escherichia coli integration host factor involves both major and minor grooves of DNA". Biochemistry. 34 (40): 13082–90. doi:10.1021/bi00040a020. PMID 7548068.
- ^ Karas VO, Westerlaken I, Meyer AS (October 2015). "The DNA-Binding Protein from Starved Cells (Dps) Utilizes Dual Functions To Defend Cells against Multiple Stresses". Journal of Bacteriology. 197 (19): 3206–15. doi:10.1128/JB.00475-15. PMC 4560292. PMID 26216848.
- ^ a b Woo JS, Lim JH, Shin HC, Suh MK, Ku B, Lee KH, et al. (January 2009). "Structural studies of a bacterial condensin complex reveal ATP-dependent disruption of intersubunit interactions" (PDF). Cell. 136 (1): 85–96. doi:10.1016/j.cell.2008.10.050. PMID 19135891. S2CID 4608756.
- ^ a b c d e Badrinarayanan A, Reyes-Lamothe R, Uphoff S, Leake MC, Sherratt DJ (October 2012). "In vivo architecture and action of bacterial structural maintenance of chromosome proteins". Science. 338 (6106): 528–31. Bibcode:2012Sci...338..528B. doi:10.1126/science.1227126. PMC 3807729. PMID 23112333.
- ^ Kumar R, Grosbart M, Nurse P, Bahng S, Wyman CL, Marians KJ (October 2017). "The bacterial condensin MukB compacts DNA by sequestering supercoils and stabilizing topologically isolated loops". The Journal of Biological Chemistry. 292 (41): 16904–16920. doi:10.1074/jbc.M117.803312. PMC 5641887. PMID 28842486.
- ^ Niki H, Imamura R, Kitaoka M, Yamanaka K, Ogura T, Hiraga S (December 1992). "E.coli MukB protein involved in chromosome partition forms a homodimer with a rod-and-hinge structure having DNA binding and ATP/GTP binding activities". The EMBO Journal. 11 (13): 5101–9. doi:10.1002/j.1460-2075.1992.tb05617.x. PMC 556988. PMID 1464330.
- ^ Melby TE, Ciampaglio CN, Briscoe G, Erickson HP (September 1998). "The symmetrical structure of structural maintenance of chromosomes (SMC) and MukB proteins: long, antiparallel coiled coils, folded at a flexible hinge". The Journal of Cell Biology. 142 (6): 1595–604. doi:10.1083/jcb.142.6.1595. PMC 2141774. PMID 9744887.
- ^ a b Yamazoe M, Onogi T, Sunako Y, Niki H, Yamanaka K, Ichimura T, Hiraga S (November 1999). "Complex formation of MukB, MukE and MukF proteins involved in chromosome partitioning in Escherichia coli". The EMBO Journal. 18 (21): 5873–84. doi:10.1093/emboj/18.21.5873. PMC 1171653. PMID 10545099.
- ^ Li Y, Stewart NK, Berger AJ, Vos S, Schoeffler AJ, Berger JM, et al. (November 2010). "Escherichia coli condensin MukB stimulates topoisomerase IV activity by a direct physical interaction". Proceedings of the National Academy of Sciences of the United States of America. 107 (44): 18832–7. doi:10.1073/pnas.1008678107. PMC 2973889. PMID 20921377.
- ^ Vos SM, Stewart NK, Oakley MG, Berger JM (November 2013). "Structural basis for the MukB-topoisomerase IV interaction and its functional implications in vivo". The EMBO Journal. 32 (22): 2950–62. doi:10.1038/emboj.2013.218. PMC 3832749. PMID 24097060.
- ^ Nicolas E, Upton AL, Uphoff S, Henry O, Badrinarayanan A, Sherratt D (February 2014). "The SMC complex MukBEF recruits topoisomerase IV to the origin of replication region in live Escherichia coli". mBio. 5 (1): e01001-13. doi:10.1128/mBio.01001-13. PMC 3950513. PMID 24520061.
- ^ Zawadzki P, Stracy M, Ginda K, Zawadzka K, Lesterlin C, Kapanidis AN, Sherratt DJ (December 2015). "The Localization and Action of Topoisomerase IV in Escherichia coli Chromosome Segregation Is Coordinated by the SMC Complex, MukBEF". Cell Reports. 13 (11): 2587–2596. doi:10.1016/j.celrep.2015.11.034. PMC 5061553. PMID 26686641.
- ^ a b c Baxter J, Oliver AW, Schalbetter SA (January 2019). "Are SMC Complexes Loop Extruding Factors? Linking Theory With Fact". BioEssays. 41 (1): e1800182. doi:10.1002/bies.201800182. PMID 30506702.
- ^ Zawadzka K, Zawadzki P, Baker R, Rajasekar KV, Wagner F, Sherratt DJ, Arciszewska LK (January 2018). "MukB ATPases are regulated independently by the N- and C-terminal domains of MukF kleisin". eLife. 7. doi:10.7554/eLife.31522. PMC 5812716. PMID 29323635.
- ^ Sawitzke JA, Austin S (February 2000). "Suppression of chromosome segregation defects of Escherichia coli muk mutants by mutations in topoisomerase I". Proceedings of the National Academy of Sciences of the United States of America. 97 (4): 1671–6. Bibcode:2000PNAS...97.1671S. doi:10.1073/pnas.030528397. PMC 26494. PMID 10660686.
- ^ Adachi S, Hiraga S (July 2003). "Mutants suppressing novobiocin hypersensitivity of a mukB null mutation". Journal of Bacteriology. 185 (13): 3690–5. doi:10.1128/jb.185.13.3690-3695.2003. PMC 161581. PMID 12813060.
- ^ Petrushenko ZM, Lai CH, Rai R, Rybenkov VV (February 2006). "DNA reshaping by MukB. Right-handed knotting, left-handed supercoiling". The Journal of Biological Chemistry. 281 (8): 4606–15. doi:10.1074/jbc.M504754200. PMC 1633270. PMID 16368697.
- ^ Gaal T, Bratton BP, Sanchez-Vazquez P, Sliwicki A, Sliwicki K, Vegel A, et al. (October 2016). "Colocalization of distant chromosomal loci in space in E. coli: a bacterial nucleolus". Genes & Development. 30 (20): 2272–2285. doi:10.1101/gad.290312.116. PMC 5110994. PMID 27898392.
- ^ Jin DJ, Cabrera JE (November 2006). "Coupling the distribution of RNA polymerase to global gene regulation and the dynamic structure of the bacterial nucleoid in Escherichia coli". Journal of Structural Biology. 156 (2): 284–91. doi:10.1016/j.jsb.2006.07.005. PMID 16934488.
- ^ a b c Qian Z, Dimitriadis EK, Edgar R, Eswaramoorthy P, Adhya S (July 2012). "Galactose repressor mediated intersegmental chromosomal connections in Escherichia coli". Proceedings of the National Academy of Sciences of the United States of America. 109 (28): 11336–41. Bibcode:2012PNAS..10911336Q. doi:10.1073/pnas.1208595109. PMC 3396475. PMID 22733746.
- ^ Weickert MJ, Adhya S (October 1993). "The galactose regulon of Escherichia coli". Molecular Microbiology. 10 (2): 245–51. doi:10.1111/j.1365-2958.1993.tb01950.x. PMID 7934815. S2CID 6872903.
- ^ a b Agerschou ED, Christiansen G, Schafer NP, Madsen DJ, Brodersen DE, Semsey S, Otzen DE (June 2016). "The transcriptional regulator GalR self-assembles to form highly regular tubular structures". Scientific Reports. 6: 27672. Bibcode:2016NatSR...627672A. doi:10.1038/srep27672. PMC 4899725. PMID 27279285.
- ^ Kavenoff R, Ryder OA (March 1976). "Electron microscopy of membrane-associated folded chromosomes of Escherichia coli". Chromosoma. 55 (1): 13–25. doi:10.1007/bf00288323. PMID 767075. S2CID 31879250.
- ^ Worcel A, Burgi E (January 1974). "Properties of a membrane-attached form of the folded chromosome of Escherichia coli". Journal of Molecular Biology. 82 (1): 91–105. doi:10.1016/0022-2836(74)90576-2. PMID 4594427.
- ^ Roggiani M, Goulian M (2015). "Chromosome-Membrane Interactions in Bacteria". Annual Review of Genetics. 49: 115–29. doi:10.1146/annurev-genet-112414-054958. PMID 26436460.
- ^ Libby EA, Roggiani M, Goulian M (May 2012). "Membrane protein expression triggers chromosomal locus repositioning in bacteria". Proceedings of the National Academy of Sciences of the United States of America. 109 (19): 7445–50. Bibcode:2012PNAS..109.7445L. doi:10.1073/pnas.1109479109. PMC 3358875. PMID 22529375.
- ^ Brameyer S, Rösch TC, El Andari J, Hoyer E, Schwarz J, Graumann PL, Jung K (2019). "DNA-binding directs the localization of a membrane-integrated receptor of the ToxR family". Communications Biology. 2: 4. doi:10.1038/s42003-018-0248-7. PMC 6320335. PMID 30740540.
- ^ Espéli O, Borne R, Dupaigne P, Thiel A, Gigant E, Mercier R, Boccard F (May 2012). "A MatP-divisome interaction coordinates chromosome segregation with cell division in E. coli". The EMBO Journal. 31 (14): 3198–211. doi:10.1038/emboj.2012.128. PMC 3400007. PMID 22580828.
- ^ Bernhardt TG, de Boer PA (May 2005). "SlmA, a nucleoid-associated, FtsZ binding protein required for blocking septal ring assembly over Chromosomes in E. coli". Molecular Cell. 18 (5): 555–64. doi:10.1016/j.molcel.2005.04.012. PMC 4428309. PMID 15916962.
- ^ Woldringh CL, Jensen PR, Westerhoff HV (September 1995). "Structure and partitioning of bacterial DNA: determined by a balance of compaction and expansion forces?" (PDF). FEMS Microbiology Letters. 131 (3): 235–42. doi:10.1111/j.1574-6968.1995.tb07782.x. PMID 7557335.
- ^ a b Van Helvoort JM, Huls PG, Vischer NO, Woldringh CL (May 1998). "Fused nucleoids resegregate faster than cell elongation in Escherichia coli pbpB(Ts) filaments after release from chloramphenicol inhibition". Microbiology. 144 (5): 1309–17. doi:10.1099/00221287-144-5-1309. PMID 9611806.
- ^ Eltsov, Mikhail; Zuber, Benoît (2006). "Transmission electron microscopy of the bacterial nucleoid". Journal of Structural Biology. 156 (2): 246–254. doi:10.1016/j.jsb.2006.07.007. ISSN 1047-8477. PMID 16978880.
- ^ Robinow, C; Kellenberger, E (1994). "The bacterial nucleoid revisited". Microbiological Reviews. 58 (2): 211–232. doi:10.1128/mmbr.58.2.211-232.1994. ISSN 0146-0749. PMC 372962. PMID 7521510.
- ^ Smith BT, Grossman AD, Walker GC (January 2002). "Localization of UvrA and effect of DNA damage on the chromosome of Bacillus subtilis". Journal of Bacteriology. 184 (2): 488–93. doi:10.1128/jb.184.2.488-493.2002. PMC 139587. PMID 11751826.
- ^ Odsbu I, Skarstad K (October 2009). "A reduction in ribonucleotide reductase activity slows down the chromosome replication fork but does not change its localization". PLOS ONE. 4 (10): e7617. Bibcode:2009PLoSO...4.7617O. doi:10.1371/journal.pone.0007617. PMC 2773459. PMID 19898675.
- ^ Levin-Zaidman S, Frenkiel-Krispin D, Shimoni E, Sabanay I, Wolf SG, Minsky A (June 2000). "Ordered intracellular RecA-DNA assemblies: a potential site of in vivo RecA-mediated activities". Proceedings of the National Academy of Sciences of the United States of America. 97 (12): 6791–6. Bibcode:2000PNAS...97.6791L. doi:10.1073/pnas.090532397. PMC 18741. PMID 10829063.
- ^ a b Delmas S, Duggin IG, Allers T (January 2013). "DNA damage induces nucleoid compaction via the Mre11-Rad50 complex in the archaeon Haloferax volcanii". Molecular Microbiology. 87 (1): 168–79. doi:10.1111/mmi.12091. PMC 3565448. PMID 23145964.