Levonorgestrel is a hormonal medication which is used in a number of birth control methods.[3][7] It is combined with an estrogen to make combination birth control pills.[8] As an emergency birth control, sold under the brand names Plan B One-Step and Julie, among others, it is useful within 72 hours of unprotected sex.[3][7][9] The more time that has passed since sex, the less effective the medication becomes, and it does not work after pregnancy (implantation) has occurred.[7] Levonorgestrel works by preventing ovulation or fertilization from occurring.[10] It decreases the chances of pregnancy by 57–93%.[11] In an intrauterine device (IUD), such as Mirena among others, it is effective for the long-term prevention of pregnancy.[7] A levonorgestrel-releasing implant is also available in some countries.[12]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Plan B, Julie, Mirena, Plan B One-Step, Liletta, others |
| Other names | LNG; LNG-EC; d-Norgestrel; d(–)-Norgestrel; D-Norgestrel; WY-5104; SH-90999; NSC-744007; 18-Methylnorethisterone; 17α-Ethynyl-18-methyl-19-nortestosterone; 17α-Ethynyl-18-methylestr-4-en-17β-ol-3-one; 13β-Ethyl-17α-hydroxy-18,19-dinorpregn-4-en-20-yn-3-one |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a610021 |
| Pregnancy category | |
| Routes of administration | By mouth, transdermal patch, intrauterine device, subcutaneous implant |
| Drug class | Progestogen (medication); Progestin |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 95% (range 85–100%)[4][5] |
| Protein binding | 98% (50% to albumin, 48% to SHBG)[4] |
| Metabolism | Liver (reduction, hydroxylation, conjugation)[4][6] |
| Metabolites | • 5α-Dihydro-LNG[4] |
| Elimination half-life | 24–32 hours[4] |
| Excretion | Urine: 20–67% Feces: 21–34%[6] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.011.227 |
| Chemical and physical data | |
| Formula | C21H28O2 |
| Molar mass | 312.453 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 235 to 237 °C (455 to 459 °F) |
| |
| |
| (verify) | |
Common side effects include nausea, breast tenderness, headaches, and increased, decreased, or irregular menstrual bleeding.[7] When used as an emergency contraceptive, if pregnancy occurs, there is no evidence that its use harms the fetus.[7] It is safe to use during breastfeeding.[7] Birth control that contains levonorgestrel will not change the risk of sexually transmitted infections.[7] It is a progestin and has effects similar to those of the hormone progesterone.[7] It works primarily by preventing ovulation and closing off the cervix to prevent the passage of sperm.[7]
Levonorgestrel was patented in 1960 and introduced for medical use together with ethinylestradiol in 1970.[13][14] It is on the World Health Organization's List of Essential Medicines.[15] It is available as a generic medication.[16] In the United States, levonorgestrel-containing emergency contraceptives are available over the counter (OTC) for all ages.[17] In 2020, it was the 323rd most commonly prescribed medication in the United States, with more than 800 thousand prescriptions.[18]
Medical uses
editBirth control
editAt low doses, levonorgestrel is used in monophasic and triphasic formulations of combined oral contraceptive pills, with available monophasic doses ranging from 100 to 250 μg, and triphasic doses of 50 μg, 75 μg, and 125 μg.[19] It is combined with the estrogen ethinylestradiol in these formulations.[19]
At very low daily dose of 30 μg, levonorgestrel is used in some progestogen-only pill formulations.[19]
Levonorgestrel is the active ingredient in a number of intrauterine devices including Mirena and Skyla.[19][20] It is also the active ingredient in the birth control implants Norplant and Jadelle.[19][20]
One of the more common forms of contraception that contains only levonorgestrel is an IUD. One IUD, the Mirena, is a small hollow cylinder containing levonorgestrel and polydimethylsiloxane and covered with a release rate controlling membrane.[21]
Emergency birth control
editLevonorgestrel is used in emergency contraceptive pills (ECPs), both in a combined Yuzpe regimen which includes estrogen, and as a levonorgestrel-only method. The levonorgestrel-only method uses levonorgestrel 1.5 mg (as a single dose or as two 0.75 mg doses 12 hours apart) taken within three days of unprotected sex. With one study indicating that beginning as late as 120 hours (5 days) after intercourse could be effective.[medical citation needed] However, taking more than one dose of emergency contraception does not increase the chance of pregnancy not happening. Planned Parenthood asserts "Taking the morning-after pill (also known as emergency contraception) multiple times doesn't change its effectiveness, and won't cause any long-term side effects."[22] There is no age, I.D., or prescription required to purchase emergency contraception. Plan B hit the market in 1999 where it could be bought by anyone older than 18. However, in 2013, the rules were changed so that Plan B could be purchased at any age without ID or prescription.
The primary mechanism of action of levonorgestrel as a progestogen-only emergency contraceptive pill is, according to International Federation of Gynecology and Obstetrics (FIGO), to prevent fertilization by inhibition of ovulation and thickening of cervical mucus.[23][24][25][26] FIGO has stated that: "review of the evidence suggests that LNG [levonorgestreol] ECPs cannot prevent implantation of a fertilized egg. Language on implantation should not be included in LNG ECP product labeling."[27][28] In November 2013, the European Medicines Agency (EMA) approved a change to the label saying it cannot prevent implantation of a fertilized egg.[29]
Other studies still find the evidence to be unclear.[30] While it is unlikely that emergency contraception affects implantation it is impossible to completely exclude the possibility of post-fertilization effect.[31]
In November 2013, the EMA also approved a change to the label for HRA Pharma's NorLevo saying: "In clinical trials, contraceptive efficacy was reduced in women weighing 75 kg [165 pounds] or more, and levonorgestrel was not effective in women who weighed more than 80 kg [176 pounds]."[29][32][33] In November 2013 and January 2014, the FDA and the EMA said they were reviewing whether increased weight and body mass index (BMI) reduce the efficacy of emergency contraceptives.[29]
An analysis of four WHO randomised clinical trials, published in January 2017, showed pregnancy rates of 1.25% (68/5428) in women with BMI under 25, 0.61% (7/1140) in women with BMI between 25 and 30, and 2.03% (6/295) in women with BMI over 30.[34] These values yield an eight-fold reduction in efficacy for women with BMI over 30 compared to women with BMI under 25. However, emergency contraceptives remain effective regardless of BMI.
Hormone therapy
editLevonorgestrel is used in combination with an estrogen in menopausal hormone therapy.[19][35] It is used under the brand name Klimonorm as a combined oral tablet with estradiol valerate and under the brand name Climara Pro as a combined transdermal patch with estradiol.[19][35]
Available forms
editAs a type of emergency contraception, levonorgestrel is used after unprotected intercourse to reduce the risk of pregnancy.[36] However, it can serve different hormonal purposes in its different methods of delivery. It is available for use in a variety of forms:
By mouth
editLevonorgestrel can be taken by mouth as a form of emergency birth control. The typical dosage is either 1.5 mg taken once or 0.75 mg taken 12–24 hours apart.[37] The effectiveness in both methods is similar.[37] The most widely used form of oral emergency contraception is the progestin-only pill, which contains a 1.5 mg dosage of levonorgestrel.[36] Levonorgestrel-only emergency contraceptive pills are reported to have an 89% effectiveness rate if taken within the recommended 72 hours after sex.[38] The efficacy of the drug decreases by 50% for each 12-hour delay in taking the dose after the emergency contraceptive regimen has been started.[38]
Skin patch
editEstradiol with levonorgestrel in the form of a skin patch is used under the brand name Climara Pro for hormone replacement therapy in postmenstrual women, treating symptoms such as hot flashes or osteoporosis.[39] The simultaneous delivery of a progestogen such as levonorgestrel is necessary for the protection of the endometrium.[40][41]
Intrauterine device
editThe levonorgestrel intrauterine system (LNG-IUS) is a type of long-term birth control that releases the progestin into the uterine cavity.[42][21] Levonorgestrel is released at a constant, gradual rate of 0.02 mg per day by the polydimethylsiloxane membrane of the device, which renders it effective for up to five years.[42] Because it is inserted directly into the uterus, levonorgestrel is present in the endometrium in much higher concentrations that would result from a LNG-containing oral pill; the LNG-IUS delivers 391 ng of levonorgestrel to the inner uterine region while a comparable oral contraceptive delivers only 1.35 ng.[42] This high level of levonorgestrel impedes the function of the endometrium, making it hostile for sperm transport, fertilization, and implantation of an ovum.[42]
Implant
editSubcutaneous implants of levonorgestrel have been marketed as birth control implants under the brand names Norplant and Jadelle and are available for use in some countries.[43][19]
Contraindications
editKnown or suspected pregnancy is a contraindication of levonorgestrel as an emergency contraceptive.[44]
Side effects
editAfter an intake of 1.5 mg levonorgestrel in clinical trials, very common side effects (reported by 10% or more) included: hives, dizziness, hair loss, headache, nausea, abdominal pain, uterine pain, delayed menstruation, heavy menstruation, uterine bleeding, and fatigue; common side effects (reported by 1% to 10%) included diarrhea, vomiting, and painful menstruation; these side effects usually disappeared within 48 hours.[45][46] However, the long term side effects common with oral contraceptives such as arterial disease are lower with levonorgestrel than in combination pills.[medical citation needed]
Levonorgestrel as a contraceptive intrauterine device is associated with a higher risk of breast cancer than with non-use.[47]
Overdose
editOverdose of levonorgestrel as an emergency contraceptive has not been described.[44] Nausea and vomiting might be expected.[44]
Interactions
editIf taken together with drugs that induce the CYP3A4 cytochrome P450 liver enzyme, levonorgestrel may be metabolized faster and may have lower effectiveness.[48] These include, but are not limited to barbiturates, bosentan, carbamazepine, felbamate, griseofulvin, oxcarbazepine, phenytoin, rifampin, St. John's wort and topiramate.[medical citation needed]
Pharmacology
editPharmacodynamics
editLevonorgestrel is a progestogen with weak androgenic activity.[4] It has no other important hormonal activity, including no estrogenic, glucocorticoid, or antimineralocorticoid activity.[4] The lack of significant mineralocorticoid or antimineralocorticoid activity with levonorgestrel is in spite of it having relatively high affinity for the mineralocorticoid receptor, which is as much as 75% of that of aldosterone.[4]
| Compound | PR | AR | ER | GR | MR | SHBG | CBG |
|---|---|---|---|---|---|---|---|
| Levonorgestrel | 150–162 | 34a, 45 | 0 | 1–8 | 17–75 | 50 | 0 |
| 5α-Dihydrolevonorgestrel | 50 | 38a | 0 | ? | ? | ? | ? |
| 3α,5α-Tetrahydrolevonorgestrel | ? | ? | 0.4 | ? | ? | ? | ? |
| 3β,5α-Tetrahydrolevonorgestrel | ? | ? | 2.4 | ? | ? | ? | ? |
| Notes: Values are percentages (%). Reference ligands (100%) were promegestone for the PR, metribolone (a = mibolerone) for the AR, E2 for the ER, DEXA for the GR, aldosterone for the MR, DHT for SHBG, and cortisol for CBG. Sources: See template. | |||||||
Progestogenic activity
editLevonorgestrel is a progestogen; that is, an agonist of the progesterone receptor (PR), the main biological target of the progestogen sex hormone progesterone.[4] It has effects similar to those of the hormone progesterone.[7] As a contraceptive, it works primarily by preventing ovulation and closing off the cervix to prevent the passage of sperm.[7] The endometrial transformation dose of levonorgestrel is 150 to 250 μg/day or 2.5 to 6 mg per cycle.[4][49][50][51]
Antigonadotropic effects
editDue to its progestogenic activity, levonorgestrel has antigonadotropic effects and is able to suppress the secretion of the gonadotropins, luteinizing hormone and follicle-stimulating hormone, from the pituitary gland.[4] This in turn, results in suppression of gonadal activity, including reduction of fertility and gonadal sex hormone production in both women and men.[4][52] The ovulation-inhibiting dose of levonorgestrel in premenopausal women is 50 to 60 μg/day.[4][49][53]
In men, levonorgestrel causes marked suppression of circulating testosterone levels secondary to its antigonadotropic effects.[54] In healthy young men, levonorgestrel alone at a dose of 120 to 240 μg/day orally for 2 weeks suppressed testosterone levels from ~450 ng/dL to ~248 ng/dL (–45%).[55] Because of its effects on testosterone levels, and due to its androgenic activity being only weak and hence insufficient for purposes of androgen replacement in males, levonorgestrel has potent functional antiandrogenic effects in men.[54] Consequently, it is able to produce adverse effects like decreased libido and erectile dysfunction, among others.[54] In relation to this, levonorgestrel has been combined with an androgen like testosterone or dihydrotestosterone when it has been studied as a hormonal contraceptive in men.[52][54]
Androgenic activity
editLevonorgestrel is a weak agonist of the androgen receptor (AR), the main biological target of the androgen sex hormone testosterone.[4] It is a weakly androgenic progestin and in women may cause androgenic biochemical changes and side effects such as decreased sex hormone-binding globulin (SHBG) levels, decreased HDL cholesterol levels, weight gain, and acne.[4][56]
In combination with a potent estrogen like ethinylestradiol however, all contraceptives containing androgenic progestins are negligibly androgenic in practice and in fact can be used to treat androgen-dependent conditions like acne and hirsutism in women.[56] This is because ethinylestradiol causes a marked increase in SHBG levels and thereby decreases levels of free and hence bioactive testosterone, acting as a functional antiandrogen.[56] Nonetheless, contraceptives containing progestins that are less androgenic increase SHBG levels to a greater extent and may be more effective for such indications.[56] Levonorgestrel is currently the most androgenic progestin that is used in contraceptives, and contraceptives containing levonorgestrel may be less effective for androgen-dependent conditions relative to those containing other progestins that are less androgenic.[57][58][59]
Other activity
editLevonorgestrel stimulates the proliferation of MCF-7 breast cancer cells in vitro, an action that is independent of the classical PRs and is instead mediated via the progesterone receptor membrane component-1 (PGRMC1).[60][61] Certain other progestins act similarly in this assay, whereas progesterone acts neutrally.[60][61] It is unclear if these findings may explain the different risks of breast cancer observed with progesterone and progestins in clinical studies.[62]
Pharmacokinetics
editThe bioavailability of levonorgestrel is approximately 95% (range 85 to 100%).[4][5] The plasma protein binding of levonorgestrel is about 98%.[4] It is bound 50% to albumin and 48% to SHBG.[4] Levonorgestrel is metabolized in the liver, via reduction, hydroxylation, and conjugation (specifically glucuronidation and sulfation).[4][6] Oxidation occurs primarily at the C2α and C16β positions, while reduction occurs in the A ring.[6] 5α-Dihydrolevonorgestrel is produced as an active metabolite of levonorgestrel by 5α-reductase.[4] The elimination half-life of levonorgestrel is 24 to 32 hours, although values as short as 8 hours and as great as 45 hours have been reported.[4][6] About 20 to 67% of a single oral dose of levonorgestrel is eliminated in urine and 21 to 34% in feces.[6]
Chemistry
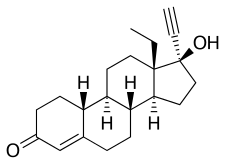
editLevonorgestrel, also known as 17α-ethynyl-18-methyl-19-nortestosterone or as 17α-ethynyl-18-methylestr-4-en-17β-ol-3-one, is a synthetic estrane steroid and a derivative of testosterone.[63][64] It is the C13β or levorotatory stereoisomer and enantiopure form of norgestrel, the C13α or dextrorotatory isomer being inactive.[65][66] Levonorgestrel is more specifically a derivative of norethisterone (17α-ethynyl-19-nortestosterone) and is the parent compound of the gonane (18-methylestrane or 13β-ethylgonane) subgroup of the 19-nortestosterone family of progestins.[67] Besides levonorgestrel itself, this group includes desogestrel, dienogest, etonogestrel, gestodene, norelgestromin, norgestimate, and norgestrel.[68] Levonorgestrel acetate and levonorgestrel butanoate are C17β esters of levonorgestrel.[69][70] Levonorgestrel has a molecular weight of 312.45 g/mol and a partition coefficient (log P) of 3.8.[71][72]
History
editNorgestrel (rac-13-ethyl-17α-ethynyl-19-nortestosterone), the racemic mixture containing levonorgestrel and dextronorgestrel, was discovered by Hughes and colleagues at Wyeth in 1963 via structural modification of norethisterone (17α-ethynyl-19-nortestosterone).[73][74][75][76] It was the first progestogen to be manufactured via total chemical synthesis.[75][76] Norgestrel was introduced for medical use as a combined birth control pill with ethinylestradiol under the brand name Eugynon in Germany in 1966 and under the brand name Ovral in the United States 1968, and as a progestogen-only pill under the brand name Ovrette in the United States in 1973.[76][77][78][79] Following its discovery, norgestrel had been licensed by Wyeth to Schering AG, which separated the racemic mixture into its two optical isomers and identified levonorgestrel (13β-ethyl-17α-ethynyl-19-nortestosterone) as the active component of the mixture.[14][75][76] Levonorgestrel was first studied in humans by 1970, and was introduced for medical use in Germany as a combined birth control pill with ethinylestradiol under the brand name Neogynon in August 1970.[14][77][78][80][81][82] A more widely used formulation, containing lower doses of ethinylestradiol and levonorgestrel, was introduced under the brand name Microgynon by 1973.[19][83][84] In addition to combined formulations, levonorgestrel was introduced as a progestogen-only pill under the brand names Microlut by 1972 and Microval by 1974.[85][86] Many other formulations and brand names of levonorgestrel-containing birth control pills have also been marketed.[19]
Levonorgestrel, taken alone in a single high dose, was first evaluated as a form of emergency contraception in 1973.[87] It was the second progestin to be evaluated for such purposes, following a study of quingestanol acetate in 1970.[87][88] In 1974, the Yuzpe regimen, which consisted of high doses of a combined birth control pill containing ethinylestradiol and norgestrel, was described as a method of emergency contraception by A. Albert Yuzpe and colleagues, and saw widespread interest.[89][90] Levonorgestrel-only emergency contraception was introduced under the brand name Postinor by 1978.[91] Ho and Kwan published the first study comparing levonorgestrel only and the Yuzpe regimen as methods of emergency contraception in 1993 and found that they had similar effectiveness but that levonorgestrel alone was better-tolerated.[92][93] In relation to this, the Yuzpe regimen has largely been replaced as a method of emergency contraception by levonorgrestrel-only preparations.[94] Levonorgestrel-only emergency contraception was approved in the United States under the brand name Plan B in 1999, and has also been marketed widely elsewhere throughout the world under other brand names such as Levonelle and NorLevo in addition to Postinor.[19][95] In 2013, the Food and Drug Administration approved Plan B One-Step for sale over-the-counter in the United States without a prescription or age restriction.[96]
Levonorgestrel has also been introduced for use as a progestogen-only intrauterine device under the brand names Mirena and Skyla among others, as a progestogen-only birth control implant under the brand names Norplant and Jadelle, as a combined oral tablet with estradiol valerate for menopausal hormone therapy under the brand name Klimonorm, and as a combined transdermal patch with estradiol for menopausal hormone therapy under the brand name Climara Pro.[19][20][35] Ester prodrugs of levonorgestrel such as levonorgestrel acetate and levonorgestrel butanoate have been developed and studied as other forms of birth control such as long-acting progestogen-only injectable contraceptives and contraceptive vaginal rings, but have not been marketed for medical use.[69][70]
Society and culture
editGeneric names
editLevonorgestrel is the generic name of the drug and its INN, USAN, USP, BAN, DCIT, and JAN, while lévonorgestrel is its DCF.[19][63][64] It is also known as d-norgestrel, d(–)-norgestrel, or D-norgestrel, as well as by its developmental code names WY-5104 (Wyeth) and SH-90999 (Schering AG).[19][63][64][85]
Brand names
editLevonorgestrel is marketed alone or in combination with an estrogen (specifically ethinylestradiol, estradiol, or estradiol valerate) under a multitude of brand names throughout the world, including Alesse, Altavera, Alysena, Amethia, Amethyst, Ashlyna, Aviane, Camrese, Chateal, Climara Pro, Cycle 21, Daysee, Emerres, Enpresse, Erlibelle, Escapelle, Falmina, Introvale, Isteranda, Jadelle, Jaydess, Jolessa, Klimonorm, Kurvelo, Kyleena, Lessina, Levlen, Levodonna, Levonelle, Levonest, Levosert, Levora, Liletta, Loette, Logynon, LoSeasonique, Lutera, Lybrel, Marlissa, Microgynon, Microlut, Microvlar, Min-Ovral, Miranova, Mirena, My Way, Myzilra, Next Choice, Nordette, Norgeston, NorLevo, Norplant, One Pill, Option 2, Orsythia, Ovima, Ovranette, Plan B, Plan B One-Step, Portia, Postinor, Postinor-2, Preventeza, Ramonna, Rigevidon, Quartette, Quasense, Seasonale, Seasonique, Skyla, Sronyx, Tri-Levlen, Trinordiol, Triphasil, Triquilar, Tri-Regol, Trivora, and Upostelle, among many others.[19][64][97] These formulations are used as emergency contraceptives, normal contraceptives, or in menopausal hormone therapy for the treatment of menopausal symptoms.[medical citation needed]
As an emergency contraceptive, levonorgestrel is often referred to colloquially as the "morning-after pill".[98][99]
Availability
editLevonorgestrel is very widely marketed throughout the world and is available in almost every country.[19][64]
Accessibility
editLevonorgestrel-containing emergency contraception is available over-the-counter in some countries, such as the United States.[96] On some college campuses, Plan B is available from vending machines.[100]
A policy update in 2015, required all pharmacies, clinics, and emergency departments run by Indian Health Services (for Native Americans) to have Plan B One-Step in stock, to distribute it to any woman (or her representative) who asked for it without a prescription, age verification, registration or any other requirement, to provide orientation training to all staff regarding the medication, to provide unbiased and medically accurate information about emergency contraception, and to make someone available at all times to distribute the pill in case the primary staffer objected to providing it on religious or moral grounds.[101]
Research
editLevonorgestrel has been studied in combination with androgens such as testosterone and dihydrotestosterone as a hormonal contraceptive for men.[52][54]
References
edit- ^ a b "Levonorgestrel Use During Pregnancy". Drugs.com. 23 March 2020. Archived from the original on 2 July 2020. Retrieved 29 June 2020.
- ^ "Jaydess 13.5 mg intrauterine delivery system - Summary of Product Characteristics (SmPC)". (emc). 1 July 2022. Archived from the original on 13 April 2021. Retrieved 1 July 2022.
- ^ a b c "Plan B One-Step- levonorgestrel tablet". DailyMed. 21 December 2022. Retrieved 26 December 2022.
- ^ a b c d e f g h i j k l m n o p q r s t u Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 (Suppl 1): 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ a b Fotherby K (August 1996). "Bioavailability of orally administered sex steroids used in oral contraception and hormone replacement therapy". Contraception. 54 (2): 59–69. doi:10.1016/0010-7824(96)00136-9. PMID 8842581.
- ^ a b c d e f Shoupe D, Haseltine FP (6 December 2012). Contraception. Springer Science & Business Media. pp. 22–. ISBN 978-1-4612-2730-4. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
- ^ a b c d e f g h i j k l "Progestins (Etonogestrel, Levonorgestrel, Norethindrone)". The American Society of Health-System Pharmacists. Archived from the original on 2015-09-07. Retrieved Aug 21, 2015.
- ^ Postgraduate Gynecology. Jaypee Brothers Medical Pub. 2011. p. 159. ISBN 9789350250822. Archived from the original on 2015-09-26.
- ^ "Levonorgestrel 1.5 mg Tablet Emergency Contraceptive: New Drug Application 21998, Supplement 5" (PDF). U.S. Food and Drug Administration.
- ^ "Now Is the Time to Change Label on Emergency Contraceptives". Relias Media | Online Continuing Medical Education | Relias Media - Continuing Medical Education Publishing. Retrieved 2022-08-16.
- ^ Gemzell-Danielsson K (November 2010). "Mechanism of action of emergency contraception". Contraception. 82 (5): 404–409. doi:10.1016/j.contraception.2010.05.004. PMID 20933113.
- ^ "Chapter 1". Research on reproductive health at WHO : biennial report 2000-2001. Geneva: World health organization. 2002. ISBN 9789241562089. Archived from the original on 2015-09-26.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 479. ISBN 9783527607495. Archived from the original on 2021-08-28. Retrieved 2020-08-05.
- ^ a b c Roth K (2014). Chemische Leckerbissen. John Wiley & Sons. pp. 77–. ISBN 978-3-527-33739-2. Archived from the original on 2021-08-28. Retrieved 2018-04-18.
[Levonorgestrel (24): The product generated by Smith's norgestrel total synthesis was a racemate, so half of each consisted of the left- and the right-handed enantiomer. Chemists at Schering discovered that only the levorotatory enantiomer was effective [49] and developed a biotechnological process for the preparation of the pure levorotatory enantiomer. This was the active ingredient levonorgestrel born. With the single-acting enantiomer, the dose and thus the liver burden could be halved again. The resulting Neogynon® contained 0.25 mg levonorgestrel and 0.05 mg ethinylestradiol and was introduced in 1970.]
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Hamilton RJ (2014). Tarascon pocket pharmacopoeia : 2014 deluxe lab-pocket edition (15th ed.). Sudbury: Jones & Bartlett Learning. pp. 310–312. ISBN 9781284053999. Archived from the original on 2015-09-26.
- ^ "FDA approves Plan B One-Step emergency contraceptive for use without a prescription for all women of child-bearing potential" (Press release). June 20, 2013. Archived from the original on 14 January 2016. Retrieved 2 February 2016.
- ^ "Levonorgestrel - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.
- ^ a b c d e f g h i j k l m n o p "Levonorgestrel". Archived from the original on 2017-08-03. Retrieved 2017-08-03.
- ^ a b c Friend DR (October 2016). "Development of controlled release systems over the past 50 years in the area of contraception". Journal of Controlled Release. 240: 235–241. doi:10.1016/j.jconrel.2015.12.043. PMID 26732558.
- ^ a b Bao Q, Gu B, Price CF, Zou Y, Wang Y, Kozak D, Choi S, Burgess DJ (October 2018). "Manufacturing and characterization of long-acting levonorgestrel intrauterine systems". International Journal of Pharmaceutics. 550 (1–2): 447–454. doi:10.1016/j.ijpharm.2018.09.004. PMC 6622171. PMID 30195080.
- ^ "How often can you take the morning-after pill?". www.plannedparenthood.org. Archived from the original on 2022-06-24. Retrieved 2022-06-18.
- ^ Trussell J, Schwarz EB (2011). "Emergency contraception". In Hatcher RA, Trussell J, Nelson AL, Cates Jr W, Kowal D, Policar MS (eds.). Contraceptive technology (20th revised ed.). New York: Ardent Media. pp. 113–145. ISBN 978-1-59708-004-0. ISSN 0091-9721. OCLC 781956734. p. 121:
Mechanism of action
Copper-releasing IUCs
When used as a regular or emergency method of contraception, copper-releasing IUCs act primarily to prevent fertilization. Emergency insertion of a copper IUC is significantly more effective than the use of ECPs, reducing the risk of pregnancy following unprotected intercourse by more than 99%.2,3 This very high level of effectiveness implies that emergency insertion of a copper IUC must prevent some pregnancies after fertilization.
Emergency contraceptive pills
To make an informed choice, women must know that ECPs—like the birth control pill, patch, ring, shot, and implant,76 and even like breastfeeding77—prevent pregnancy primarily by delaying or inhibiting ovulation and inhibiting fertilization, but may at times inhibit implantation of a fertilized egg in the endometrium. However, women should also be informed that the best available evidence indicates that ECPs prevent pregnancy by mechanisms that do not involve interference with post-fertilization events.
ECPs do not cause abortion78 or harm an established pregnancy. Pregnancy begins with implantation according to medical authorities such as the US FDA, the National Institutes of Health79 and the American College of Obstetricians and Gynecologists (ACOG).80
Ulipristal acetate (UPA). One study has demonstrated that UP can delay ovulation.81... Another study found that UPA altered the endometrium, but whether this change would inhibit implantation is unknown.82
p. 122:
Progestin-only emergency contraceptive pills. Early treatment with ECPs containing only the progestin levonorgestrel has been shown to impair the ovulatory process and luteal function.83–87
p. 123:
Combined emergency contraceptive pills. Several clinical studies have shown that combined ECPs containing ethinyl estradiol and levonorgestrel can inhibit or delay ovulation.107–110 - ^ RCOG Faculty of Sexual; Reproductive Healthcare; Clinical Effectiveness Unit (January 2012). "Clinical guidance: emergency contraception" (PDF). Clinical Guidance. London: Royal College of Obstetricians and Gynaecologists. ISSN 1755-103X. Archived from the original (PDF) on 2012-05-26. Retrieved 2012-04-30. p.3:
How does EC work?
In 2002, a judicial review ruled that pregnancy begins at implantation, not fertilisation.8 The possible mechanisms of action should be explained to the patient as some methods may not be acceptable, depending on individual beliefs about the onset of pregnancy and abortion.
Copper-bearing intrauterine device (Cu-IUD). Copper is toxic to the ovum and sperm and thus the copper-bearing intrauterine device (Cu-IUD) is effective immediately after insertion and works primarily by inhibiting fertilisation.9–11 A systematic review on mechanisms of action of IUDs showed that both pre- and postfertilisation effects contribute to efficacy.11 If fertilisation has already occurred, it is accepted that there is an anti-implantation effect,12,13
Levonorgestrel (LNG). The precise mode of action of levonorgestrel (LNG) is incompletely understood but it is thought to work primarily by inhibition of ovulation.16,17
Ulipristal acetate (UPA). UPA's primary mechanism of action is thought to be inhibition or delay of ovulation.2 - ^ UNDP/UNFPA/WHO/World Bank Special Programme of Research, Development and Research Training in Human Reproduction (HRP) (March 25, 2010). "Fact sheet on the safety of levonorgestrel-alone emergency contraceptive pills (LNG ECPs)" (PDF). Geneva: World Health Organization. Archived (PDF) from the original on March 16, 2012.
Can LNG ECPs cause an abortion?
LNG ECPs do not interrupt an established pregnancy or harm a developing embryo.15 The evidence available to date shows that LNG ECP use does not prevent a fertilized egg from attaching to the uterine lining. The primary mechanism of action is to stop or disrupt ovulation; LNG ECP use may also prevent the sperm and egg from meeting.16 - ^ Speroff L, Darney PD (2011). "Special uses of oral contraception: emergency contraception, the progestin-only minipill". A clinical guide for contraception (5th ed.). Philadelphia: Lippincott Williams & Wilkins. pp. 153–166. ISBN 978-1-60831-610-6. p. 155:
Emergency postcoital contraception
Levonorgestrel
Mechanism and efficacy - ^ Belluck P (June 6, 2012). "No abortion role seen for morning-after pill". The New York Times. p. A1. Archived from the original on February 27, 2017.
Belluck P (June 6, 2012). "Drug's nickname may have aided politicization". The New York Times. p. A14. - ^ International Federation of Gynecology and Obstetrics (FIGO) and International Consortium for Emergency Contraception (ICEC) (April 4, 2011). "Mechanism of action: How do levonorgestrel-only emergency contraceptive pills (LNG ECPs) prevent pregnancy?" (PDF). London: International Federation of Gynecology and Obstetrics. Archived (PDF) from the original on December 29, 2014.
Levonorgestrel-only emergency contraceptive pills:
• Interfere with the process of ovulation;
• May possibly prevent the sperm and the egg from meeting.
Implications of the research:
• Inhibition or delay of ovulation is LNG ECPs principal and possibly only mechanism of action.
• Review of the evidence suggests that LNG-ECs cannot prevent implantation of a fertilized egg. Language on implantation should not be included in LNG ECP product labeling.
• The fact that LNG-ECs have no demonstrated effect on implantation explains why they are not 100% effective in preventing pregnancy, and are less effective the later they are taken. Women should be given a clear message that LNG-ECs are more effective the sooner they are taken.
• LNG ECPs do not interrupt a pregnancy (by any definition of the beginning of pregnancy). However, LNG ECPs can prevent abortions by reducing unwanted pregnancies. - ^ a b c Belluck P (November 26, 2013). "New birth control label counters lawsuit claim; European authorities found that a drug like Plan B One-Step cannot prevent fertilized eggs from implanting in the womb". The New York Times. Archived from the original on March 4, 2014. Retrieved March 5, 2014.
HRA Pharma (November 2013). "NorLevo 1.5 mg tablet Patient Information Leaflet (PIL)" (PDF). Dublin: Irish Medicines Board. Archived (PDF) from the original on March 5, 2014. Retrieved March 5, 2014.NorLevo works by stopping your ovaries from releasing an egg. It cannot stop a fertilized egg from attaching to the womb.
HRA Pharma (November 2013). "NorLevo 1.5 mg tablet Summary of Product Characteristics (SPC)". Dublin: Irish Pharmaceutical Healthcare Association. Archived from the original on June 15, 2012. Retrieved March 5, 2014.
European Medicines Agency (January 24, 2014). "Review of emergency contraceptives started". London: European Medicines Agency. Archived from the original on March 27, 2014. Retrieved March 5, 2014. - ^ Mozzanega B, Cosmi E (June 2011). "How do levonorgestrel-only emergency contraceptive pills prevent pregnancy? Some considerations". Gynecological Endocrinology. 27 (6): 439–42. doi:10.3109/09513590.2010.501885. hdl:11577/2476296. PMID 20670097. S2CID 6431709.
- ^ Leung VW, Levine M, Soon JA (February 2010). "Mechanisms of action of hormonal emergency contraceptives". Pharmacotherapy. 30 (2): 158–68. doi:10.1592/phco.30.2.158. PMID 20099990. S2CID 41337748.
- ^ Glasier A, Cameron ST, Blithe D, Scherrer B, Mathe H, Levy D, Gainer E, Ulmann A (October 2011). "Can we identify women at risk of pregnancy despite using emergency contraception? Data from randomized trials of ulipristal acetate and levonorgestrel". Contraception. 84 (4): 363–7. doi:10.1016/j.contraception.2011.02.009. PMID 21920190.
- ^ Trussell J, Raymond EG, Cleland K (February 2014). "Emergency contraception: a last chance to prevent unintended pregnancy" (PDF). Princeton: Office of Population Research at Princeton University, Association of Reproductive Health Professionals. Archived from the original (PDF) on September 23, 2010. Retrieved April 9, 2014.
- ^ Festin MP, Peregoudov A, Seuc A, Kiarie J, Temmerman M (January 2017). "Effect of BMI and body weight on pregnancy rates with LNG as emergency contraception: analysis of four WHO HRP studies". Contraception. 95 (1): 50–54. doi:10.1016/j.contraception.2016.08.001. PMC 5357708. PMID 27527670.
- ^ a b c Kubíková D (2014). "[Menopausal symptoms and hormone replacement therapy]" (PDF). Praktické Lékárenství. 10 (2): 68–73.
- ^ a b "Emergency Contraception - ACOG". www.acog.org. Archived from the original on 2019-10-09. Retrieved 2019-06-01.
- ^ a b Hansen LB, Saseen JJ, Teal SB (February 2007). "Levonorgestrel-only dosing strategies for emergency contraception". Pharmacotherapy. 27 (2): 278–84. doi:10.1592/phco.27.2.278. PMID 17253917. S2CID 24229915.
- ^ a b Shohel M, Rahman MM, Zaman A, Uddin MM, Al-Amin MM, Reza HM (April 2014). "A systematic review of effectiveness and safety of different regimens of levonorgestrel oral tablets for emergency contraception". BMC Women's Health. 14: 54. doi:10.1186/1472-6874-14-54. PMC 3977662. PMID 24708837.
- ^ "Estradiol And Levonorgestrel (Transdermal Route) Description and Brand Names - Mayo Clinic". www.mayoclinic.org. Archived from the original on 2019-05-31. Retrieved 2019-06-01.
- ^ "Climara Pro® (Estradiol/Levonorgestrel Transdermal System)" (PDF). Food and Drug Administration. Archived (PDF) from the original on 2021-02-28. Retrieved 2019-06-01.
- ^ Mueck AO, Römer T (July 2018). "Choice of progestogen for endometrial protection in combination with transdermal estradiol in menopausal women". Hormone Molecular Biology and Clinical Investigation. 37 (2). doi:10.1515/hmbci-2018-0033. PMID 30063464. S2CID 51886877.
- ^ a b c d Jensen JT (September 2005). "Contraceptive and therapeutic effects of the levonorgestrel intrauterine system: an overview". Obstetrical & Gynecological Survey. 60 (9): 604–12. doi:10.1097/01.ogx.0000175805.90122.af. PMID 16121115. S2CID 43177026.
- ^ Benagiano G, Gabelnick H, Farris M (September 2008). "Contraceptive devices: subcutaneous delivery systems". Expert Review of Medical Devices. 5 (5): 623–637. doi:10.1586/17434440.5.5.623. PMID 18803473. S2CID 207201811.
- ^ a b c "Plan B One-Step (levonorgestrel) tablet, 1.5 mg, for oral use" (PDF). Barr Pharmaceuticals, Inc. U.S. Food and Drug Administration. July 2009. Archived from the original (PDF) on 2019-04-17.
- ^ HRA Pharma (November 2013). "NorLevo 1.5 mg tablet Summary of Product Characteristics (SPC)". Dublin: Irish Pharmaceutical Healthcare Association. Archived from the original on June 15, 2012. Retrieved April 9, 2014.
- ^ Chen X, Wu X, Zhu H (April 2014). "Acute urticaria as a side effect of the Mirena® (levonorgestrel-releasing intrauterine system): a case report". BMC Research Notes. 7: 209. doi:10.1186/1756-0500-7-209. PMC 3992142. PMID 24708811.
- ^ Conz L, Mota BS, Bahamondes L, Teixeira Dória M, Françoise Mauricette Derchain S, Rieira R, Sarian LO (January 2020). "Levonorgestrel-releasing intrauterine system and breast cancer risk: A systematic review and meta-analysis". Acta Obstet Gynecol Scand. 99 (8): 970–982. doi:10.1111/aogs.13817. PMID 31990981. S2CID 210946832.
- ^ Medicines and Healthcare products Regulatory Agency (15 September 2016). "Levonorgestrel-containing emergency hormonal contraception: advice on interactions with hepatic enzyme inducers and contraceptive efficacy". GOV.UK. Archived from the original on 21 January 2017. Retrieved 6 June 2017.
- ^ a b Schindler AE, Campagnoli C, Druckmann R, Huber J, Pasqualini JR, Schweppe KW, Thijssen JH (December 2003). "Classification and pharmacology of progestins". Maturitas. 46 (Suppl 1): S7–S16. doi:10.1016/j.maturitas.2003.09.014. PMID 14670641.
- ^ Knörr K, Knörr-Gärtner H, Beller FK, Lauritzen C (8 March 2013). Geburtshilfe und Gynäkologie: Physiologie und Pathologie der Reproduktion. Springer-Verlag. pp. 583–. ISBN 978-3-642-95583-9.
- ^ Lauritzen C (September 1990). "Clinical use of oestrogens and progestogens". Maturitas. 12 (3): 199–214. doi:10.1016/0378-5122(90)90004-p. PMID 2215269.
- ^ a b c Nieschlag E (November 2010). "Clinical trials in male hormonal contraception" (PDF). Contraception. 82 (5): 457–470. doi:10.1016/j.contraception.2010.03.020. PMID 20933120. Archived (PDF) from the original on 2020-12-05. Retrieved 2019-07-11.
- ^ Endrikat J, Gerlinger C, Richard S, Rosenbaum P, Düsterberg B (December 2011). "Ovulation inhibition doses of progestins: a systematic review of the available literature and of marketed preparations worldwide". Contraception. 84 (6): 549–57. doi:10.1016/j.contraception.2011.04.009. PMID 22078182.
- ^ a b c d e Meriggiola MC, Farley TM, Mbizvo MT (2003). "A review of androgen-progestin regimens for male contraception". Journal of Andrology. 24 (4): 466–83. doi:10.1002/j.1939-4640.2003.tb02695.x. PMID 12826683.
Based on animal studies and clinical studies in women, 19‐norderived progestins are known to be potent in terms of gonadotropin suppression (Couzinet et al, 1996). Among this class of steroidal compounds are norethisterone (NET), norethynodrel, and its dextrorotatory isomer LNG (ie, the biologically active form of this progestin). The progestins of this class are known to be potent suppressors of gonadotropin secretion, and when administered to men these compounds induced a profound suppression of sperm production (Frick, 1973). However, a decrease of libido and sexual potency was also reported, presumably due to the suppression of T production secondary to gonadotropin suppression (Kamischke et al, 2000b). Therefore, like other progestins available thus far, nor‐progestins should not be administered alone for male contraception because their residual androgenic activity is not sufficient to maintain androgen‐dependent physiological functions like libido or sexual potency (Kamischke et al, 2000a).
- ^ Zitzmann M, Rohayem J, Raidt J, Kliesch S, Kumar N, Sitruk-Ware R, Nieschlag E (May 2017). "Impact of various progestins with or without transdermal testosterone on gonadotropin levels for non-invasive hormonal male contraception: a randomized clinical trial". Andrology. 5 (3): 516–526. doi:10.1111/andr.12328. PMID 28189123. S2CID 41502711.
- ^ a b c d Darney PD (January 1995). "The androgenicity of progestins". The American Journal of Medicine. 98 (1A): 104S–110S. doi:10.1016/s0002-9343(99)80067-9. PMID 7825629.
- ^ Knaus JV, Isaacs JH (6 December 2012). Office Gynecology: Advanced Management Concepts. Springer Science & Business Media. pp. 151–. ISBN 978-1-4612-4340-3. Archived from the original on 8 September 2017.
- ^ Golan DE, Tashjian AH, Armstrong EJ (15 December 2011). Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. Lippincott Williams & Wilkins. pp. 516–. ISBN 978-1-60831-270-2. Archived from the original on 8 September 2017.
- ^ Committee on the Relationship Between Oral Contraceptives and BreastCancer (1 January 1991). Oral Contraceptives and Breast Cancer. National Academies. pp. 147–. ISBN 9780309044936. NAP:13774. Archived from the original on 8 September 2017.
- ^ a b Neubauer H, Ma Q, Zhou J, Yu Q, Ruan X, Seeger H, Fehm T, Mueck AO (October 2013). "Possible role of PGRMC1 in breast cancer development". Climacteric. 16 (5): 509–13. doi:10.3109/13697137.2013.800038. PMID 23758160. S2CID 29808177.
- ^ a b Ruan X, Neubauer H, Yang Y, Schneck H, Schultz S, Fehm T, Cahill MA, Seeger H, Mueck AO (October 2012). "Progestogens and membrane-initiated effects on the proliferation of human breast cancer cells". Climacteric. 15 (5): 467–72. doi:10.3109/13697137.2011.648232. PMID 22335423. S2CID 11302554.
- ^ Trabert B, Sherman ME, Kannan N, Stanczyk FZ (September 2019). "Progesterone and breast cancer". Endocr. Rev. 41 (2): 320–344. doi:10.1210/endrev/bnz001. PMC 7156851. PMID 31512725.
- ^ a b c Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 887–. ISBN 978-1-4757-2085-3. Archived from the original on 8 September 2017.
- ^ a b c d e Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 605–. ISBN 978-3-88763-075-1. Archived from the original on 2017-09-08.
- ^ Alldredge BK, Corelli RL, Ernst ME (1 February 2012). Koda-Kimble and Young's Applied Therapeutics: The Clinical Use of Drugs. Lippincott Williams & Wilkins. pp. 1072–. ISBN 978-1-60913-713-7. Archived from the original on 8 September 2017.
- ^ Lavery JP, Sanfilippo JS (6 December 2012). Pediatric and Adolescent Obstetrics and Gynecology. Springer Science & Business Media. pp. 248–. ISBN 978-1-4612-5064-7. Archived from the original on 8 September 2017.
- ^ Offermanns S, Rosenthal W (14 August 2008). Encyclopedia of Molecular Pharmacology. Springer Science & Business Media. pp. 390–. ISBN 978-3-540-38916-3. Archived from the original on 8 September 2017.
- ^ Schindler AE (July 2014). "The "newer" progestogens and postmenopausal hormone therapy (HRT)". J Steroid Biochem Mol Biol. 142: 48–51. doi:10.1016/j.jsbmb.2013.12.003. PMID 24333799. S2CID 32126275.
- ^ a b Population Reports: Injectables and implants. Department of Medical and Public Affairs, George Washington University. 1987. Archived from the original on 2021-04-20. Retrieved 2018-04-15.
The Population Council also plans to test vaginal rings with two other progestins, ST-1435 and levonorgestrel acetate, alone and combined with ethinyl estradiol (168).
- ^ a b Crabbé P, Archer S, Benagiano G, Diczfalusy E, Djerassi C, Fried J, Higuchi T (March 1983). "Long-acting contraceptive agents: design of the WHO Chemical Synthesis Programme". Steroids. 41 (3): 243–53. doi:10.1016/0039-128X(83)90095-8. PMID 6658872. S2CID 12896179.
- ^ "Levonorgestrel". PubChem. Retrieved 18 August 2022.
- ^ "Levonorgestrel: Uses, Interactions, Mechanism of Action". DrugBank Online. 30 October 2013. Retrieved 18 August 2022.
- ^ Meikle AW (1 June 1999). Hormone Replacement Therapy. Springer Science & Business Media. pp. 383–. ISBN 978-1-59259-700-0. Archived from the original on 1 August 2020. Retrieved 15 April 2018.
The gonanes share the structural modifications found in the estranes and also possess [an ethyl] group at the position 13 and a keto group at position 3. Norgestrel was synthesized in 1963 and is a racemic mixture of dextro and levorotatory forms. The levorotatory form, levonorgestrel, provides the biological activity.
- ^ Szejtli J, Szente L (6 December 2012). Proceedings of the Eighth International Symposium on Cyclodextrins: Budapest, Hungary, March 31–April 2, 1996. Springer Science & Business Media. pp. 317–. ISBN 978-94-011-5448-2. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
[Norgestrel] was discovered by Hughes et al. (1963).
- ^ a b c Filshie M, Guillebaud J (22 October 2013). Contraception: Science and Practice. Elsevier Science. pp. 12–. ISBN 978-1-4831-6366-6.
Norgestrel, developed by Wyeth and patented in 1964, was the first progestogen to be manufactured by total chemical synthesis. It was subsequently licensed to Schering AG, who separated the racemic mixture into an inactive structural isomer l-norgestrel and the active d-norgestrel -- more usually known as dextronorgestrel and levonorgestrel respectively, because of the optical isomerism that each displays.
- ^ a b c d Marks L (2010). Sexual Chemistry: A History of the Contraceptive Pill. Yale University Press. pp. 73, 76. ISBN 978-0-300-16791-7.
In 1964 the pharmaceutical company Wyeth developed norgestrel, the first progestogen to be made from a total chemical synthesis. Subsequently licensed to Schering AG, norgestrel was used to develop levonorgestrel, another active progestogen later used for oral contraception.
- ^ a b Pohl WG (2004). Die wissenschaftliche Welt von gestern: die Preisträger des Ignaz L. Lieben-Preises 1865-1937 und des Richard Lieben-Preises 1912-1928 : ein Kapitel österreichischer Wissenschaftsgeschichte in Kurzbiografien. Böhlau Verlag Wien. pp. 150–. ISBN 978-3-205-77303-0. Archived from the original on 2021-08-28. Retrieved 2018-04-18.
[The contraceptive EUGYNON is launched in 1966. NEOGYNON follows in 1970.]
- ^ a b Ortiz-Gómez T, Santesmases MJ (22 April 2016). Gendered Drugs and Medicine: Historical and Socio-Cultural Perspectives. Taylor & Francis. pp. 175–. ISBN 978-1-317-12981-3. Archived from the original on 6 October 2021. Retrieved 18 April 2018.
The 1966 marketing campaign for Schering's second contraceptive, Eugynon, [...] (Schering AG Berline 1966, 11). [...] In 1970 [Schering] had already conducted an opinion poll among doctors in the run up to the marketing campaign for the newly introduced Neogynon. [...]
- ^ Tone A, Watkins ES (8 January 2007). Medicating Modern America: Prescription Drugs in History. NYU Press. pp. 118–119. ISBN 978-0-8147-8300-9.
- ^ Albach H (1993). Culture and Technical Innovation: A Cross-Cultural Analysis and Policy Recommendations. Walter de Gruyter. p. 952. ISBN 978-3-11-013947-1. Archived from the original on August 28, 2021. Retrieved August 5, 2020.
[Since the safety of ovulation inhibition by levonorgestrel was also proven in the clinical studies, the cycle was extremely stable and the side effects were low, the drug was on August 1, 1970 introduced as Neogynon 21 and Neogynon 28 in Germany on the market.] [...] After the OC market had risen sharply in 1968 and 1969, the launch of Neogynon / Schering and Stediril-d / Wyeth in August 1970 gave the market a fresh boost.]
- ^ Apelo R, Veloso I (1970). "Results of a controlled study employing d-norgestrel and ethinyl estradiol". Contraception. 2 (6): 391–400. doi:10.1016/S0010-7824(70)80002-6. ISSN 0010-7824.
The results obtained in these series clinically confirmed the findings in animal work on the potency of d-norgestrel, i.e., that the biological activity of norgestrel resides largely in the d-enantlomer (5,6).
- ^ Brosens I, Van Assche A, Wijnants P (1971). "Comparative clinical and morphological studies on 2 oral contraceptives which contain DL-norgestrel and D-norgestrel respectively". Geburtshilfe und Frauenheilkunde. 31 (3): 251–257. Archived from the original on 2018-04-15. Retrieved 2018-04-15.
Comparison of the effects of Eugynon and Neogynon (.05 mg ethinyl estradiol with .5 mg norgestrel or with .25 mg d-norgestrel, respectively) in 272 women is reported. The 2 preparations were comparable as regards effectiveness (100%), cycle control, and endometrial and cervical morphology. No clinical or biological complications occurred, and the incidence of minor side effects was very small. The d-norgestrel preparation (Neogynon) may be preferable for metabolic reasons because of its lower steroid dose.
- ^ Schneider W, Spona J, Matt K (1974). "Inhibition of ovulation by means of a combined preparation with reduced amounts of active substance". Contraception. 9 (1): 81–92. doi:10.1016/0010-7824(74)90096-1. ISSN 0010-7824.
- ^ Brat T (1974). "Clinical trial with a new low oestrogen combined oral contraceptive". Current Medical Research and Opinion. 2 (8): 465–470. doi:10.1185/03007997409115244. PMID 4614952.
- ^ a b Scharff HJ (1972). "Clinical experience with Microlut". Medical News Schering (1): 2–9. Archived from the original on 2018-04-15.
- ^ Lambotte R, Werbrouck-Navette J (March 1974). "[Minipill as the new contraceptive method]". Revue Médicale de Liège (in French). 29 (6): 157–159. PMID 4817042. Archived from the original on 2018-04-15. Retrieved 2018-04-15.
- ^ a b Kesserü E, Larrañaga A, Parada J (1973). "Postcoital contraception with D-norgestrel". Contraception. 7 (5): 367–379. doi:10.1016/0010-7824(73)90139-X. ISSN 0010-7824.
- ^ Rubio B, Berman E, Larranaga A, Guiloff E (1970). "A new postcoital oral contraceptive". Contraception. 1 (5): 303–314. doi:10.1016/0010-7824(70)90016-8. ISSN 0010-7824.
- ^ Balaji (19 November 2009). Textbook of Oral and Maxillofacial Surgery. Elsevier India. p. 569. ISBN 978-81-312-0300-2. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
There are two main methods involving oral emergency pills, commonly misleadingly described as the 'morning-after pill'. The first older method, developed in the mid-1970s, involves two high-dose combined pills containing oestrogen (50 ug ethinyloestradiol) and progesterone (0.25 mg levonorgestrel): the Yuzpe regime (Schering PC4 or Ovran). The second involves progesterone only (0.75 mg levonorgestrel), and therefore, has a lower incidence of side effects, in particular vomiting (6%).
- ^ Yuzpe AA, Thurlow HJ, Ramzy I, Leyshon JI (August 1974). "Post coital contraception--A pilot study". The Journal of Reproductive Medicine. 13 (2): 53–8. PMID 4844513.
- ^ Farkas M (1978). "Post-coital contraception with Postinor, a preparation containing 0.75 mg d-norgestrel". Magyar Nöorvosok Lapja. 41: 474.
- ^ Artini PG, Genazzani AR, Petraglia F (11 December 2001). Advances in Gynecological Endocrinology. CRC Press. pp. 96–. ISBN 978-1-84214-071-0. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
- ^ Ho PC, Kwan MS (March 1993). "A prospective randomized comparison of levonorgestrel with the Yuzpe regimen in post-coital contraception". Human Reproduction. 8 (3): 389–92. doi:10.1093/oxfordjournals.humrep.a138057. PMID 8473453.
- ^ King (21 October 2013). Varney's Midwifery. Jones & Bartlett Publishers. pp. 493–. ISBN 978-1-284-02542-2. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
- ^ Harrison-Woolrych M (28 January 2015). Medicines For Women. Springer. pp. 205–. ISBN 978-3-319-12406-3. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
- ^ a b "FDA approves over-the-counter sales of Plan B One-Step for all ages". CBS News. 20 June 2013. Archived from the original on 2017-08-03. Retrieved 2017-08-03.
- ^ Trussell J, Cleland K (2007-04-10). "Emergency Contraceptive Pills Worldwide". Princeton University. Archived from the original on 2007-05-22. Retrieved 2007-05-28.
- ^ Oats JJ, Abraham S (17 November 2011). Llewellyn-Jones Fundamentals of Obstetrics and Gynaecology E-Book. Elsevier Health Sciences. pp. 247–. ISBN 978-0-7234-3719-2.
- ^ Carlson KJ, Eisenstat SA, Ziporyn TD (2004). The New Harvard Guide to Women's Health. Harvard University Press. pp. 285–. ISBN 978-0-674-01282-0.
- ^ Dugan Arnett (March 27, 2022). "As some states seek to limit reproductive freedoms, BU opens 'Plan B' vending machine". The Boston Globe. Archived from the original on March 28, 2022. Retrieved March 28, 2022.
- ^ Rankin K (2015-10-22). "This Policy Gives Native Women Equal Access to Emergency Contraception". Colorlines. Archived from the original on 2015-10-26. Retrieved 2015-10-24.
External links
editMedia related to Levonorgestrel at Wikimedia Commons