Hydrocortisone is the name for the hormone cortisol when supplied as a medication.[14] Uses include conditions such as adrenocortical insufficiency, adrenogenital syndrome, high blood calcium, thyroiditis, rheumatoid arthritis, dermatitis, asthma, and COPD.[1] It is the treatment of choice for adrenocortical insufficiency.[15] It can be given by mouth, topically, or by injection.[1] Stopping treatment after long-term use should be done slowly.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | A-hydrocort, Cortef, Solu-cortef, others[1] |
| Other names | Cortisol; 11β,17α,21-Trihydroxypregn-4-ene-3,20-dione; 11β,17α,21-Trihydroxyprogesterone |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682206 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth (tablets), intravenous, topical, rectal |
| Drug class | Corticosteroid; Glucocorticoid; Mineralocorticoid |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: 96 ± 20%[11][12] |
| Protein binding | 92 ± 2% (92–93%)[11][12] |
| Metabolism | 11β-HSDs, others[12] |
| Metabolites | Cortisone, others[12] |
| Onset of action | Oral: 1.2 ± 0.4 hours (Tmax)[11] |
| Elimination half-life | 1.2–2.0 hours[11][12] |
| Duration of action | 8–12 hours[13] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
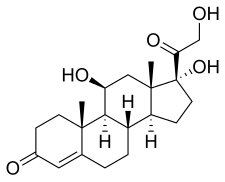
| Formula | C21H30O5 |
| Molar mass | 362.466 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Side effects may include mood changes, increased risk of infection, and edema (swelling).[1] With long-term use common side effects include osteoporosis, upset stomach, physical weakness, easy bruising, and candidiasis (yeast infections).[1] It is unclear if it is safe for use during pregnancy.[16] Hydrocortisone is a glucocorticoid and works as an anti-inflammatory and by immune suppression.[1]
Hydrocortisone was patented in 1936 and approved for medical use in 1941.[17][18] It is on the World Health Organization's List of Essential Medicines.[19] It is available as a generic medication.[1] In 2021, it was the 192nd most commonly prescribed medication in the United States, with more than 2 million prescriptions.[20][21]
Medical uses
editHydrocortisone is the pharmaceutical term for cortisol used in oral administration, intravenous injection, or topical application. It is used as an immunosuppressive drug, given by injection in the treatment of severe allergic reactions such as anaphylaxis and angioedema, in place of prednisolone in patients needing steroid treatment but unable to take oral medication, and perioperatively in patients on long-term steroid treatment to prevent an adrenal crisis. It may also be injected into inflamed joints resulting from diseases such as gout.
It may be used topically for allergic rashes, eczema, psoriasis, itching and other inflammatory skin conditions. Topical hydrocortisone creams and ointments are available in most countries without prescription in strengths ranging from 0.05% to 2.5% (depending on local regulations) with stronger forms available by prescription only.[citation needed]
It may also be used rectally in suppositories to relieve the swelling, itch, and irritation in haemorrhoids.[7]
It may be used as an acetate form (hydrocortisone acetate), which has slightly different pharmacokinetics and pharmacodynamics.[7][22]
-
Cortisol for injection
-
A tube of hydrocortisone cream, purchased over the counter
-
Hydrocortisone 10 mg oral tablets (depicted a package for Russian market)
Pharmacology
editPharmacodynamics
editHydrocortisone is a corticosteroid, acting specifically as both a glucocorticoid and as a mineralocorticoid. That is, it is an agonist of the glucocorticoid and mineralocorticoid receptors.
Hydrocortisone has low potency relative to synthetic corticosteroids.[13] Compared to hydrocortisone, prednisolone is about 4 times as potent and dexamethasone about 40 times as potent in terms of anti-inflammatory effect.[23] Prednisolone can also be used as cortisol replacement, and at replacement dose levels (rather than anti-inflammatory levels), prednisolone is about 8 times more potent than cortisol.[24] The equivalent doses and relative potencies of hydrocortisone compared to various other synthetic corticosteroids have also been reviewed and summarized.[13]
The endogenous production rate of cortisol is approximately 5.7 to 9.9 mg/m2 per day, which corresponds to an oral hydrocortisone dose of approximately 15 to 20 mg/day (for a 70-kg person).[25][26] One review described daily cortisol production of 10 mg in healthy volunteers and reported that daily cortisol production could increase up to 400 mg in conditions of severe stress (e.g., surgery).[11]
The total and/or free concentrations of cortisol/hydrocortisone required for various glucocorticoid effects have been determined.[11]
Pharmacokinetics
editAbsorption
editThe bioavailability of oral hydrocortisone is about 96% ± 20% (SD).[11][12] The pharmacokinetics of hydrocortisone are non-linear.[11] The peak level of oral hydrocortisone is 15.3 ± 2.9 (SD) μg/L per 1 mg dose.[11] The time to peak concentrations of oral hydrocortisone is 1.2 ± 0.4 (SD) hours.[11]
The topical percutaneous absorption of hydrocortisone varies widely depending on experimental circumstances and has been reported to range from 0.5 to 14.9% in different studies.[27] Some skin application sites, like the scrotum and vulva, absorb hydrocortisone much more efficiently than other application sites, like the forearm.[27][28][29] In one study, the amount of hydrocortisone absorbed ranged from 0.2% to 36.2% depending on the application site, with the ball of the foot having the lowest absorption and the scrotum having the highest absorption.[29] The absorption of hydrocortisone by the vulva has ranged from 4.4 to 8.1%, relative to 1.3 to 2.8% for the arm, in different studies and subjects.[29][30][31]
Distribution
editMost cortisol in the blood (all but about 4%) is bound to proteins, including corticosteroid binding globulin (CBG) and serum albumin. A pharmacokinetic review stated that 92% ± 2% (SD) (92–93%) of hydrocortisone is plasma protein-bound.[11] Free cortisol passes easily through cellular membranes.[32] Inside cells it interacts with corticosteroid receptors.[33]
Metabolism
editHydrocortisone is metabolized by 11β-hydroxysteroid dehydrogenases (11β-HSDs) into cortisone, an inactive metabolite.[12][11] It is additionally 5α-, 5β-, and 3α-reduced into dihydrocortisols, dihydrocortisones, tetrahydrocortisols, and tetrahydrocortisones.[34][11][12]
Elimination
editThe elimination half-life of hydrocortisone ranges from about 1.2 to 2.0 (SD) hours, with an average of around 1.5 hours, regardless of oral versus parenteral administration.[11][12] The duration of action of systemic hydrocortisone has been listed as 8 to 12 hours.[13]
Chemistry
editHydrocortisone, also known as 11β,17α,21-trihydroxypregn-4-ene-3,20-dione, is a naturally occurring pregnane steroid.[35][36] A variety of hydrocortisone esters exist and have been marketed for medical use.[35][36]
Society and culture
editLegal status
editIn March 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Efmody, intended for the treatment of congenital adrenal hyperplasia (CAH) in people aged twelve years and older.[37] The applicant for this medicinal product is Diurnal Europe BV.[37] Hydrocortisone (Efmody) was approved for medical use in the European Union, in May 2021, for the treatment of congenital adrenal hyperplasia (CAH) in people aged twelve years and older.[9]
Anti-competitive practices
editIn the UK, the Competition and Markets Authority (CMA) concluded an investigation into the supply of hydrocortisone tablets, finding that from October 2008 onwards, drug suppliers Auden McKenzie and Actavis plc had charged "excessive and unfair prices" for 10mg and 20mg tablets and entered into agreements with potential competitors, paying companies who agreed not to enter the hydrocortisone market and enabling Auden McKenzie and Actavis to supply the drugs as "generic" rather than branded products and thereby escape price controls until eventually other companies entered the market. Auden and Actavis overcharged the UK's National Health Service for over ten years. Fines totalling over £255m were levied against the companies involved in this breach of competition law.[38]
Research
editCOVID-19
editHydrocortisone was found to be effective in reducing mortality rate of critically ill COVID-19 patients when compared to other usual care or a placebo.[39]
References
edit- ^ a b c d e f g h "Hydrocortisone". Drugs.com. American Society of Health-System Pharmacists. 9 February 2015. Archived from the original on 20 September 2016. Retrieved 30 August 2016.
- ^ "Prescribing medicines in pregnancy database". Therapeutic Goods Administration (TGA). Archived from the original on 20 December 2016. Retrieved 21 February 2021.
- ^ "Hydrocortisone Notice of enforcement policy" (PDF). FDA. Archived (PDF) from the original on 12 March 2023. Retrieved 31 December 2022.
- ^ "Ala-cort- hydrocortisone cream". DailyMed. Archived from the original on 27 October 2020. Retrieved 21 February 2021.
- ^ "Ala-scalp- hydrocortisone lotion". DailyMed. Archived from the original on 21 April 2021. Retrieved 21 February 2021.
- ^ "Alkindi Sprinkle- hydrocortisone granule". DailyMed. Archived from the original on 10 February 2022. Retrieved 21 February 2021.
- ^ a b c "Anusol HC- hydrocortisone acetate suppository". DailyMed. Archived from the original on 10 February 2022. Retrieved 21 February 2021.
- ^ "Cortef- hydrocortisone tablet". DailyMed. Archived from the original on 17 April 2021. Retrieved 21 February 2021.
- ^ a b "Efmody EPAR". European Medicines Agency (EMA). 24 March 2021. Archived from the original on 14 June 2021. Retrieved 14 June 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "Efmody Product information". Union Register of medicinal products. Archived from the original on 5 March 2023. Retrieved 3 March 2023.
- ^ a b c d e f g h i j k l m n Czock D, Keller F, Rasche FM, Häussler U (2005). "Pharmacokinetics and pharmacodynamics of systemically administered glucocorticoids". Clinical Pharmacokinetics. 44 (1): 61–98. doi:10.2165/00003088-200544010-00003. PMID 15634032. S2CID 24458998.
- ^ a b c d e f g h i Lennernäs H, Skrtic S, Johannsson G (June 2008). "Replacement therapy of oral hydrocortisone in adrenal insufficiency: the influence of gastrointestinal factors". Expert Opinion on Drug Metabolism & Toxicology. 4 (6): 749–758. doi:10.1517/17425255.4.6.749. PMID 18611115. S2CID 73248541.
- ^ a b c d Liu D, Ahmet A, Ward L, Krishnamoorthy P, Mandelcorn ED, Leigh R, et al. (August 2013). "A practical guide to the monitoring and management of the complications of systemic corticosteroid therapy". Allergy Asthma Clin Immunol. 9 (1): 30. doi:10.1186/1710-1492-9-30. PMC 3765115. PMID 23947590.
- ^ Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. p. 762. ISBN 978-0-7817-1750-2. Archived from the original on 14 September 2016.
- ^ Hamilton R (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 202. ISBN 978-1-284-05756-0.
- ^ "Hydrocortisone Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 20 September 2016. Retrieved 1 September 2016.
- ^ U.S. patent 2,183,589
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 484. ISBN 978-3-527-60749-5. Archived from the original on 10 January 2023. Retrieved 7 September 2020.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Hydrocortisone - Drug Usage Statistics". ClinCalc. Archived from the original on 12 April 2020. Retrieved 14 January 2024.
- ^ Leite FM, Longui CA, Kochi C, Faria CD, Borghi M, Calliari LE, et al. (February 2008). "[Comparative study of prednisolone versus hydrocortisone acetate for treatment of patients with the classic congenital adrenal hyperplasia due to 21-hydroxylase deficiency]". Arq Bras Endocrinol Metabol (in Portuguese). 52 (1): 101–8. doi:10.1590/s0004-27302008000100014. PMID 18345402.
- ^ "Dexamethasone". drugs.com. Archived from the original on 21 June 2013. Retrieved 14 June 2013.
- ^ Caldato MC, Fernandes VT, Kater CE (October 2004). "One-year clinical evaluation of single morning dose prednisolone therapy for 21-hydroxylase deficiency". Arquivos Brasileiros de Endocrinologia e Metabologia. 48 (5): 705–712. doi:10.1590/S0004-27302004000500017. PMID 15761542. S2CID 13986916.
- ^ Arvat E, Falorni A (2016). Cortisol Excess and Insufficiency. Frontiers of Hormone Research. S. Karger AG. p. 1-PA61. ISBN 978-3-318-05840-6. Archived from the original on 27 April 2023. Retrieved 10 April 2023.
- ^ Ghizzoni L, Cappa M, Chrousos GP, Loche S, Maghnie M, eds. (2011). Pediatric Adrenal Diseases: Workshop, May 16-18, 2010, Turin (Italy). Karger Medical and Scientific Publishers. pp. 174–. ISBN 978-3-8055-9643-5. OCLC 1020003143. Archived from the original on 10 April 2023. Retrieved 10 April 2023.
- ^ a b Wester RC, Maibach HI (1993). "Percutaneous absorption of topical corticosteroids". Current Problems in Dermatology. 21: 45–60. doi:10.1159/000422362. ISBN 978-3-8055-5712-2. PMID 8299376.
- ^ Bormann JL, Maibach HI (September 2020). "Effects of anatomical location on in vivo percutaneous penetration in man". Cutaneous and Ocular Toxicology. 39 (3): 213–222. doi:10.1080/15569527.2020.1787434. PMID 32643443. S2CID 220439810.
- ^ a b c Wester RC, Maibach HI (8 June 2021). "Regional Variation in Percutaneous Absorption". Percutaneous Absorption. CRC Press. pp. 165–174. doi:10.1201/9780429202971-11. ISBN 978-0-429-20297-1. S2CID 132864025.
- ^ Britz MB, Maibach HI, Anjo DM (1980). "Human percutaneous penetration of hydrocortisone: the vulva". Archives of Dermatological Research. 267 (3): 313–316. doi:10.1007/BF00403852. PMID 7406539. S2CID 33367289.
- ^ Oriba HA, Bucks DA, Maibach HI (February 1996). "Percutaneous absorption of hydrocortisone and testosterone on the vulva and forearm: effect of the menopause and site". The British Journal of Dermatology. 134 (2): 229–233. doi:10.1111/j.1365-2133.1996.tb07606.x. PMID 8746334. S2CID 30076779.
- ^ Charmandari E, Johnston A, Brook CG, Hindmarsh PC (April 2001). "Bioavailability of oral hydrocortisone in patients with congenital adrenal hyperplasia due to 21-hydroxylase deficiency". The Journal of Endocrinology. 169 (1): 65–70. doi:10.1677/joe.0.1690065. PMID 11250647.
- ^ Boron WF, Boulpaep EL (2011). Medical Physiology (2nd ed.). Philadelphia: Saunders. ISBN 978-1-4377-1753-2.
- ^ Nikolaou N, Hodson L, Tomlinson JW (March 2021). "The role of 5-reduction in physiology and metabolic disease: evidence from cellular, pre-clinical and human studies". The Journal of Steroid Biochemistry and Molecular Biology. 207: 105808. doi:10.1016/j.jsbmb.2021.105808. PMID 33418075. S2CID 230716310.
- ^ a b Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 316. ISBN 978-1-4757-2085-3. Archived from the original on 8 September 2017.
- ^ a b Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 524–. ISBN 978-3-88763-075-1. Archived from the original on 10 January 2023. Retrieved 19 June 2020.
- ^ a b "Efmody: Pending EC decision". European Medicines Agency (EMA). 25 March 2021. Archived from the original on 4 May 2021. Retrieved 27 March 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ This article incorporates text published under the British Open Government Licence: Competition and Markets Authority, Decision: Hydrocortisone tablets. Excessive and unfair pricing and Anti-competitive agreements, published 31 March 2022, accessed 1 June 2023
- ^ Sterne JA, Murthy S, Diaz JV, Slutsky AS, Villar J, Angus DC, et al. (October 2020). "Association Between Administration of Systemic Corticosteroids and Mortality Among Critically Ill Patients With COVID-19: A Meta-analysis". JAMA. 324 (13): 1330–1341. doi:10.1001/jama.2020.17023. PMC 7489434. PMID 32876694.