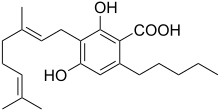
Cannabigerolic acid (CBGA) is the acidic form of cannabigerol (CBG). It is a dihydroxybenzoic acid and olivetolic acid in which the hydrogen at position 3 is substituted by a geranyl group. It is a biosynthetic precursor to Delta-9-tetrahydrocannabinol, which is the principal psychoactive constituent of the Cannabis plant. It is also a meroterpenoid (i.e. a polyketide and a terpenoid), a member of resorcinols and a phytocannabinoid. It derives from an olivetolic acid. It is a conjugate acid of a cannabigerolate.[1]
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
3-[(2E)-3,7-Dimethylocta-2,6-dien-1-yl]-2,4-dihydroxy-6-pentylbenzoic acid | |
| Identifiers | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| Properties | |
| C22H32O4 | |
| Molar mass | 360.494 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
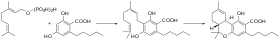
In the Cannabis plant, olivetolic acid and geranyl diphosphate are synthesized into CBGA.[2]: 6 [3]: 17 CBGA is converted in the plant by CBCA synthase, CBDA synthase and THCA synthase into CBCA, CBDA and THCA respectively.[2]: 6–7 Afterwards, THCA and CBDA can be decarboxylated into THC and CBD by drying and heating plant material. CBGA has emerging pharmacological properties; for example, it had anticonvulsant effects in a mouse model of Dravet syndrome, a form of epilepsy.[4]
COVID-19 edit
In an analysis by the University of Rhode Island on phytocannabinoids it was found that CBGA had the 2nd highest 3C-like protease inhibitor activity against COVID-19 out of all the phytocannabinoids tested within that study but not as high as the antiviral drug GC376 (72% CBGA vs 24% CBG vs 100% GC376). [5]
A 2022 pre-clinical study by researchers from the Oregon State University and Oregon Health & Science University found that CBGA (along with CBDA and THCA) could prevent infection by SARS-CoV-2. They found that CBGA was able to block infection by the reference strain (WA-1/2020), alpha variant (B.1.1.7) and beta variant (B.1.351) at micromolar concentrations (IC50 values 37 μg/mL, 26 μg/mL, and 35 μg/mL, respectively). The results, however, are yet to be replicated in animal models or clinical trials. Out of the cannabinoids studied, CBGA was the only one capable of allosteric binding, in addition to orthosteric binding.[6]
References edit
- ^ "Cannabigerolic acid". PubChem. United States National Library of Medicine. Retrieved April 7, 2020.
- ^ a b Thomas BF, ElSohly MA (2015). The Analytical Chemistry of Cannabis: Quality Assessment, Assurance, and Regulation of Medicinal Marijuana and Cannabinoid Preparations. Emerging Issues in Analytical Chemistry. Elsevier Science. ISBN 978-0-12-804670-8.
- ^ Degenhardt VF, Stehle F, Kayser O (2016). "The biosynthesis of cannabinoids". In Preedy VR (ed.). Handbook of Cannabis and Related Pathologies: Biology, Pharmacology, Diagnosis, and Treatment. Academic Press. pp. 13–23. ISBN 978-0128008270.
- ^ Anderson LL, Heblinski M, Absalom NL, Hawkins NA, Bowen MT, Benson MJ, et al. (December 2021). "Cannabigerolic acid, a major biosynthetic precursor molecule in cannabis, exhibits divergent effects on seizures in mouse models of epilepsy". British Journal of Pharmacology. 178 (24): 4826–4841. doi:10.1111/bph.15661. PMC 9292928. PMID 34384142. S2CID 236997090.
- ^ Liu C, Puopolo T, Li H, Cai A, Seeram NP, Ma H (September 2022). "Identification of SARS-CoV-2 Main Protease Inhibitors from a Library of Minor Cannabinoids by Biochemical Inhibition Assay and Surface Plasmon Resonance Characterized Binding Affinity". Molecules. 27 (18): 6127. doi:10.3390/molecules27186127. PMC 9502466. PMID 36144858.
- ^ van Breemen, Richard B.; Muchiri, Ruth N.; Bates, Timothy A.; Weinstein, Jules B.; Leier, Hans C.; Farley, Scotland; Tafesse, Fikadu G. (2022-01-28). "Cannabinoids Block Cellular Entry of SARS-CoV-2 and the Emerging Variants". Journal of Natural Products. 85 (1): 176–184. doi:10.1021/acs.jnatprod.1c00946. ISSN 0163-3864. PMC 8768006. PMID 35007072.
Further reading edit
- Jacqueline H (9 September 2019). "What is CBGA (Cannabigerolic Acid) & what does this cannabinoid do?". Leafly.