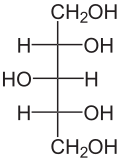
Xylitol is a chemical compound with the formula C
5H
12O
5, or HO(CH2)(CHOH)3(CH2)OH; specifically, one particular stereoisomer with that structural formula. It is a colorless or white crystalline solid that is freely soluble in water. It is classified as a polyalcohol and a sugar alcohol, specifically an alditol. The name derives from Ancient Greek: ξύλον, xyl[on] 'wood', with the suffix -itol used to denote it being a sugar alcohol.
| |
 Xylitol crystals
| |
| Names | |
|---|---|
| Pronunciation | /ˈzaɪlɪtɒl/ |
| IUPAC name
meso-Xylitol
| |
| Systematic IUPAC name
(2R,3R,4S)-Pentane-1,2,3,4,5-pentol | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.626 |
| E number | E967 (glazing agents, ...) |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H12O5 | |
| Molar mass | 152.146 g·mol−1 |
| Density | 1.52 g/cm3 |
| Melting point | 92 to 96 °C (198 to 205 °F; 365 to 369 K) |
| Boiling point | 345.39 °C (653.70 °F; 618.54 K) Predicted value using Adapted Stein & Brown method[2] |
| ~100 g/L | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related alkanes
|
Pentane |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Xylitol is used as a food additive and sugar substitute. Its European Union code number is E967.[3] Replacing sugar with xylitol in food products may promote better dental health, but evidence is lacking on whether xylitol itself prevents dental cavities.[4]
History
editEmil Fischer, a German chemistry professor, and his assistant Rudolf Stahel isolated a new compound from beech wood chips in September 1890 and named it Xylit, the German word for xylitol. The following year, the French chemist M. G. Bertrand isolated xylitol syrup by processing wheat and oat straw.[5] Sugar rationing during World War II led to an interest in sugar substitutes. Interest in xylitol and other polyols became intense, leading to their characterization and manufacturing methods.[6][7]
Structure, production, commerce
editXylitol is one of three 5-carbon sugar alcohols. The others are arabitol and ribitol. These three compounds differ in the stereochemistry of the three secondary alcohol groups.
Xylitol occurs naturally in small amounts in plums, strawberries, cauliflower, and pumpkin; humans and many other animals make trace amounts during metabolism of carbohydrates.[6] Unlike most sugar alcohols, xylitol is achiral.[8] Most other isomers of pentane-1,2,3,4,5-pentol are chiral, but xylitol has a plane of symmetry.
Industrial production starts with lignocellulosic biomass from which xylan is extracted; raw biomass materials include hardwoods, softwoods, and agricultural waste from processing maize, wheat, or rice. The mixture is hydrolyzed with acid to give xylose. The xylose is purified by chromatography. Purified xylose is catalytically hydrogenated into xylitol using a Raney nickel catalyst.[9] The conversion changes the sugar (xylose, an aldehyde) into the primary alcohol, xylitol.[6]
Xylitol can also be obtained by industrial fermentation, but this methodology are not as economical as the acid hydrolysis/chromatography route described above. Fermentation is effected by bacteria, fungi, or yeast, especially Candida tropicalis.[6][10] According to the US Department of Energy, xylitol production by fermentation from discarded biomass is one of the most valuable renewable chemicals for commerce, forecast to be a US $1.4 billion industry by 2025.[11]
Uses
editXylitol is used as a sugar substitute in such manufactured products as drugs, dietary supplements, confections, toothpaste, and chewing gum, but is not a common household sweetener.[4][12] Xylitol has negligible effects on blood sugar because its assimilation and metabolism are independent of insulin.[12] It is approved as a food additive in the United States[13] and elsewhere. Xylitol is often marketed as "birch sugar".
Xylitol is also found as an additive to saline solution for nasal irrigation and has been reported to be effective in improving symptoms of chronic sinusitis.[14]
Xylitol can also be incorporated into fabrics to produce a cooling fabric. When moisture, such as sweat, comes into contact with the xylitol embedded in the fabric, it produces a cooling sensation.[15]
Food properties
editNutrition, taste, and cooking
editHumans absorb xylitol more slowly than sucrose, and xylitol supplies 40% fewer calories than an equal mass of sucrose.[12]
Xylitol has about the same sweetness as sucrose,[12] but is sweeter than similar compounds like sorbitol and mannitol.[6]
Xylitol is stable enough to be used in baking,[16] but because xylitol and other polyols are more heat-stable, they do not caramelise as sugars do. When used in foods, they lower the freezing point of the mixture.[17]
Food risks
editNo serious health risk exists in most humans for normal levels of consumption. The European Food Safety Authority has not set a limit on daily intake of xylitol. Due to the adverse laxative effect that all polyols have on the digestive system in high doses, xylitol is banned from soft drinks in the European Union. Similarly due to a 1985 report, by the E.U. Scientific Committee on Food, stating that "ingesting 50 g a day of xylitol can cause diarrhea", tabletop sweeteners, as well as other products containing xylitol are required to display the warning: "Excessive consumption may induce laxative effects".[18]
Metabolism
editXylitol has 2.4 kilocalories of food energy per gram of xylitol (10 kilojoules per gram) according to U.S. and E.U. food-labeling regulations.[19][3] The real value can vary, depending on metabolic factors.[20]
Primarily, the liver metabolizes absorbed xylitol. The main metabolic route in humans occurs in cytoplasm, via nonspecific NAD-dependent dehydrogenase (polyol dehydrogenase), which transforms xylitol to D-xylulose. Specific xylulokinase phosphorylates it to D-xylulose-5-phosphate. This then goes to pentose phosphate pathway for further processing.[20]
About 50% of eaten xylitol is absorbed via the intestines. Of the remaining 50% that is not absorbed by the intestines, in humans, 50–75% of the xylitol remaining in the gut is fermented by gut bacteria into short-chain organic acids and gases, which may produce flatulence. The remnant unabsorbed xylitol that escapes fermentation is excreted unchanged, mostly in feces; less than 2 g of xylitol out of every 100 g ingested is excreted via urine.[20]
Xylitol ingestion also increases motilin secretion, which may be related to xylitol's ability to cause diarrhea.[21] The less-digestible but fermentable nature of xylitol also contributes to constipation relieving effects.[22]
Health effects
editDental care
editA 2015 Cochrane review of ten studies between 1991 and 2014 suggested a positive effect in reducing tooth decay of xylitol-containing fluoride toothpastes when compared to fluoride-only toothpaste, but there was insufficient evidence to determine whether other xylitol-containing products can prevent tooth decay in infants, children or adults.[23] Subsequent reviews support the belief that xylitol can suppress the growth of pathogenic Streptococcus in the mouth, thereby reducing dental caries and gingivitis, although there is concern that swallowed xylitol may cause intestinal dysbiosis.[24][25][26] A 2022 review suggested that xylitol-containing chewing gum decreases plaque, but not xylitol-containing candy.[27]
Earache
editIn 2011 EFSA "concluded that there was not enough evidence to support" the claim that xylitol-sweetened gum could prevent middle-ear infections, also known as acute otitis media (AOM).[18][28] A 2016 review indicated that xylitol in chewing gum or a syrup may have a moderate effect in preventing AOM in healthy children.[29] It may be an alternative to conventional therapies (such as antibiotics) to lower risk of earache in healthy children – reducing risk of occurrence by 25%[30] – although there is no definitive proof that it could be used as a therapy for earache.[29]
Diabetes
editIn 2011, EFSA approved a marketing claim that foods or beverages containing xylitol or similar sugar replacers cause lower blood glucose and lower insulin responses compared to sugar-containing foods or drinks.[16][31] Xylitol products are used as sucrose substitutes for weight control,[16][22] as xylitol has 40% fewer calories than sucrose (2.4 kcal/g compared to 4.0 kcal/g for sucrose).[16][32] The glycemic index (GI) of xylitol is only 7% of the GI for glucose.[33]
Adverse effects
editHumans
editWhen ingested at high doses, xylitol and other polyols may cause gastrointestinal discomfort, including flatulence, diarrhea, and irritable bowel syndrome (see Metabolism above); some people experience the adverse effects at lower doses.[18][34] Xylitol has a lower laxation threshold than some sugar alcohols but is more easily tolerated than mannitol and sorbitol.[35]
Increased xylitol consumption can increase oxalate, calcium, and phosphate excretion to urine (termed oxaluria, calciuria, and phosphaturia, respectively). These are known risk factors for kidney stone disease, but despite that, xylitol has not been linked to kidney disease in humans.[36]
Dogs and other animals
editXylitol is poisonous to dogs.[37] Ingesting 100 milligrams of xylitol per kilogram of body weight (mg/kg bw) causes dogs to experience a dose-dependent insulin release; depending on the dose it can result in life-threatening hypoglycemia. Hypoglycemic symptoms of xylitol toxicity may arise as quickly as 30 to 60 minutes after ingestion. Vomiting is a common first symptom, which can be followed by tiredness and ataxia. At doses above 500 mg/kg bw, liver failure is likely and may result in coagulopathies like disseminated intravascular coagulation.[38]
Xylitol is safe for rhesus macaques, horses, and rats.[38]
A 2018 study suggests that xylitol is safe for cats in doses of up to 1000 mg/kg; however, this study was performed on only 6 cats and should not be considered definitive.[39]
See also
editReferences
edit- ^ Safety data sheet for xylitol Archived 3 March 2016 at the Wayback Machine from Fisher Scientific. Retrieved 2014-11-02.
- ^ "Xylitol". Chemspider.com. Chemical Structure. Retrieved 13 May 2015.
- ^ a b "Food legislation". polyols-eu.org. European Association of Polyol Producers. 22 March 2017. Retrieved 7 February 2019.
- ^ a b
Riley, P.; Moore, D.; Ahmed, F.; Sharif, M.O.; Worthington, H.V. (26 March 2015). "Xylitol-containing products for preventing dental caries in children and adults". The Cochrane Database of Systematic Reviews. 2015 (3): CD010743. doi:10.1002/14651858.CD010743.pub2. PMC 9345289. PMID 25809586.
Riley, P.; Moore, D.; Ahmed, F.; Sharif, M. O.; Worthington, H. V. (2015). "Can xylitol – used in products like sweets, candy, chewing gum, and toothpaste – help prevent tooth decay in children and adults?". The Cochrane Database of Systematic Reviews. Lay summary. 2015 (3): CD010743. doi:10.1002/14651858.CD010743.pub2. PMC 9345289. PMID 25809586. - ^ Mäkinen KK (June 2000). "The rocky road of xylitol to its clinical application". Journal of Dental Research. 79 (6): 1352–5. doi:10.1177/00220345000790060101. PMID 10890712. S2CID 31432699.
- ^ a b c d e Ur-Rehman, S.; Mushtaq, Z.; Zahoor, T.; Jamil, A.; Murtaza, M.A. (2015). "Xylitol: A review on bio-production, application, health benefits, and related safety issues". Critical Reviews in Food Science and Nutrition. 55 (11): 1514–1528. doi:10.1080/10408398.2012.702288. PMID 24915309. S2CID 20359589.
- ^ Hicks, Jesse (Spring 2010). "The pursuit of sweet". Science History. Science History Institute.
- ^ Wrolstad, Ronald E. (2012). Food Carbohydrate Chemistry. John Wiley & Sons. p. 176. ISBN 9780813826653. Retrieved 20 October 2012 – via Google Books.
- ^ Schiweck, Hubert; Bär, Albert; Vogel, Roland; Schwarz, Eugen; Kunz, Markwart; Dusautois, Cécile; Clement, Alexandre; Lefranc, Caterine; Lüssem, Bernd; Moser, Matthias; Peters, Siegfried (2012). "Sugar Alcohols". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a25_413.pub3. ISBN 9783527303854.
- ^ Jain, H.; Mulay, S. (March 2014). "A review on different modes and methods for yielding a pentose sugar: Xylitol". International Journal of Food Sciences and Nutrition. 65 (2): 135–143. doi:10.3109/09637486.2013.845651. PMID 24160912. S2CID 39929588.
- ^ Felipe Hernández-Pérez, Andrés; de Arruda, Priscila Vaz; Sene, Luciane; da Silva, Silvio Silvério; Kumar Chandel, Anuj; de Almeida Felipe, Maria das Graças (16 July 2019). "Xylitol bioproduction: State-of-the-art, industrial paradigm shift, and opportunities for integrated biorefineries". Critical Reviews in Biotechnology. 39 (7): 924–943. doi:10.1080/07388551.2019.1640658. ISSN 0738-8551. PMID 31311338. S2CID 197421362.
- ^ a b c d "Xylitol". Drugs.com. 2018. Retrieved 12 October 2018.
- ^ "Xylitol". United States Code of Federal Regulations. Food Additives Permitted for Direct Addition to Food for Human Consumption, Special Dietary and Nutritional Additives. U.S. Food and Drug Administration. 1 April 2012. CFR Title 21, Part 172, Section 172.395.
- ^ Weissman, Joshua D.; Fernandez, Francisca; Hwang, Peter H. (November 2011). "Xylitol nasal irrigation in the management of chronic rhinosinusitis: A pilot study". The Laryngoscope. 121 (11): 2468–2472. doi:10.1002/lary.22176. ISSN 1531-4995. PMID 21994147. S2CID 36572019.
- ^ Peng, Yucan; Cui, Yi (15 April 2020). "Advanced Textiles for Personal Thermal Management and Energy". Joule. 4 (4): 724–742. Bibcode:2020Joule...4..724P. doi:10.1016/j.joule.2020.02.011. ISSN 2542-4351.
- ^ a b c d "Xylitol". Diabetes.co.uk. Retrieved 28 October 2018.
- ^ Burgos, Karen; Subramaniam, Persis; Arthur, Jennifer (21 November 2016). "Reformulation guide for small to medium sized companies" (PDF). Leatherhead Food Research. Archived from the original (PDF) on 27 September 2020. Retrieved 28 October 2018 – via The Food and Drink Federation.
- ^ a b c "Is xylitol good for your teeth?". Live well: Eat well. U.K. National Health Service. 13 April 2016. Retrieved 28 October 2018.
- ^ "Chapter 3: Energy Conversion Factors". Calculation of the Energy Content of Foods. Food and Agriculture Organization (Report). The United Nations. Retrieved 30 March 2017.
- ^ a b c Livesey, G. (2003). "Health potential of polyols as sugar replacers, with emphasis on low glycaemic properties". Nutrition Research Reviews. 16 (2): 163–191. doi:10.1079/NRR200371. ISSN 1475-2700. PMID 19087388.
- ^ Wölnerhanssen, B. K.; Meyer-Gerspach, A. C.; Beglinger, C.; Islam, M. S. (June 2019). "Metabolic effects of the natural sweeteners xylitol and erythritol: A comprehensive review". Critical Reviews in Food Science and Nutrition. 60 (12): 1986–1998. doi:10.1080/10408398.2019.1623757. PMID 31204494. S2CID 189944738.
- ^ a b Salli, Krista; Lehtinen, Markus J.; Tiihonen, Kirsti; Ouwehand, Arthur C. (6 August 2019). "Xylitol's health benefits beyond dental health: A comprehensive review". Nutrients. 11 (8): 1813. doi:10.3390/nu11081813. ISSN 2072-6643. PMC 6723878. PMID 31390800.
- ^ Riley P, Moore D, Ahmed F, Worthington HV (2015). "Xylitol-containing products for preventing dental caries in children and adults". Cochrane Library#The_Cochrane Database of Systematic Reviews. 2015 (3): CD010743. doi:10.1002/14651858.CD010743.pub2. PMC 9345289. PMID 25809586.
- ^ Benahmed AG, Gasmi A, Bjørklund G (2020). "Health benefits of xylitol". Applied Microbiology and Biotechnology. 104 (17): 225–7237. doi:10.1007/s00253-020-10708-7. PMID 32638045.
- ^ ALHumaid J, Bamashmous M (2022). "Meta-analysis on the Effectiveness of Xylitol in Caries Prevention". Journal of International Society of Preventive & Community Dentistry. 12 (2): 133–138. doi:10.4103/jispcd.JISPCD_164_21. PMC 9022379. PMID 35462747.
- ^ Janket S, Benwait J, Meurman JH (2019). "Oral and Systemic Effects of Xylitol Consumption". Caries Research. 53 (5): 491–501. doi:10.1159/000499194. PMID 31060040.
- ^ Söderling, Eva; Pienihäkkinen, Kaisu (2022). "Effects of xylitol chewing gum and candies on the accumulation of dental plaque: a systematic review". Clinical Oral Investigations. 26 (1): 119–129. doi:10.1007/s00784-021-04225-8. ISSN 1432-6981. PMC 8791908. PMID 34677696.
- ^ EFSA pannel (June 2011). "Scientific opinion on the substantiation of health claims related to sugar-free chewing gum sweetened with xylitol and plaque acid neutralisation (ID 485), maintenance of tooth mineralisation (ID 486, 562, 1181), reduction of dental plaque (ID 485, 3085)". EFSA Journal. 9 (6): 2266. doi:10.2903/j.efsa.2011.2266.
- ^ a b Azarpazhooh, A.; Lawrence, H.P.; Shah, P.S. (3 August 2016). "Xylitol for preventing acute otitis media in children up to 12 years of age". The Cochrane Database of Systematic Reviews. 2016 (8): CD007095. doi:10.1002/14651858.CD007095.pub3. PMC 8485974. PMID 27486835.
- ^ Marom, Tal; Marchisio, Paola; Tamir, Sharon Ovnat; Torretta, Sara; Gavriel, Haim; Esposito, Susanna (12 February 2016). "Complementary and alternative medicine treatment options for otitis media". Medicine. 95 (6): e2695. doi:10.1097/MD.0000000000002695. ISSN 0025-7974. PMC 4753897. PMID 26871802.
- ^ EFSA panel (April 2011). "Scientific opinion on the substantiation of health claims related to the sugar replacers xylitol, sorbitol, mannitol, maltitol, lactitol, isomalt, erythritol, D-tagatose, isomaltulose, sucralose and polydextrose and maintenance of tooth mineralisation by decreasing tooth demineralisation, and reduction of post-prandial glycaemic responses". EFSA Journal. 9 (4): 2076. doi:10.2903/j.efsa.2011.2076.
- ^ Tiefenbacher, Karl F. (2017). "Technology of Main Ingredients – Sweeteners and Lipids". Wafer and Waffle. Elsevier. pp. 123–225. doi:10.1016/b978-0-12-809438-9.00003-x. ISBN 978-0-12-809438-9.
- ^ Foster-Powell, K.; Holt, S.H.; Brand-Miller, J.C. (July 2002). "International table of glycemic index and glycemic load values: 2002". The American Journal of Clinical Nutrition. 76 (1): 5–56. doi:10.1093/ajcn/76.1.5. PMID 12081815.
- ^ Mäkinen, Kauko (20 October 2016). "Gastrointestinal disturbances associated with the consumption of sugar alcohols with special consideration of xylitol: Scientific review and instructions for dentists and other health-care professionals". International Journal of Dentistry. 2016: 5967907. doi:10.1155/2016/5967907. PMC 5093271. PMID 27840639.
- ^ Sugar Alcohols (PDF) (Report). Canadian Diabetes Association. 1 May 2005. Archived from the original (PDF) on 25 April 2012. Retrieved 14 March 2012.
- ^ Janket, S.; Benwait, J.; Isaac, P.; Ackerson, L.K.; Meurman, J.H. (2019). "Oral and systemic effects of xylitol consumption". Caries Research. 53 (5): 491–501. doi:10.1159/000499194. hdl:10138/305074. PMID 31060040. S2CID 146811298.
- ^ "Paws off xylitol; It's dangerous for dogs". US Food and Drug Administration. 7 July 2021. Retrieved 9 September 2021.
- ^ a b Schmid, R. D.; Hovda, L. R. (2016). "Acute hepatic failure in a dog after xylitol ingestion". Journal of Medical Toxicology. 12 (2): 201–205. doi:10.1007/s13181-015-0531-7. PMC 4880608. PMID 26691320.
- ^ Jerzsele, A.; et al. (2018). "Effects of p.o. administered xylitol in cats". Journal of Veterinary Pharmacology and Therapeutics. 41 (3): 409–414. doi:10.1111/jvp.12479. PMID 29430681.
