 | |
| Clinical data | |
|---|---|
| Trade names | Sporanox, Sporaz, Orungal, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692049 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (capsules, solution), local (vaginal suppository), intravenous (IV) |
| Drug class | Antifungal (triazole)[2] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~55%, maximal if taken with full meal |
| Protein binding | 99.8% |
| Metabolism | Extensive in liver (CYP3A4) |
| Metabolites | Hydroxy-itraconazole, keto-itraconazole, N-desalkyl-itraconazole[4] |
| Elimination half-life | 21 hours |
| Excretion | Urine (35%), faeces (54%)[5] |
| Identifiers | |
| |
| Chemical and physical data | |
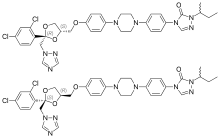
| Formula | C35H38Cl2N8O4 |
| Molar mass | 705.64 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Itraconazole is an antifungal medication used to treat a number of fungal infections.[2] This includes aspergillosis, blastomycosis, coccidioidomycosis, histoplasmosis, and paracoccidioidomycosis.[2] It may be given by mouth or intravenously.[2]
Common side effects include nausea, diarrhea, abdominal pain, rash, and headache.[2] Severe side effects may include liver problems, heart failure, Stevens–Johnson syndrome and allergic reactions including anaphylaxis.[2] It is unclear if use during pregnancy or breastfeeding is safe.[1] It is in the triazole family of medications.[2] It stops fungal growth by affecting the cell membrane or affecting their metabolism.[2]
Itraconazole was patented in 1978 and approved for medical use in the United States in 1992.[2][8] It is on the World Health Organization's List of Essential Medicines.[9] The wholesale cost in the developing world is about US$0.29 per day of treatment as of 2015[update].[10] In the United States, as of 2021[update], the wholesale cost of this dose is $2.[11] In the UK, as of 2020, 15 capsules of 100mg itraconazole costs the NHS £3.55.[12]
References
edit- ^ a b c "Itraconazole Use During Pregnancy". Drugs.com. 20 March 2019. Archived from the original on 10 April 2020. Retrieved 15 May 2020.
- ^ a b c d e f g h i j "Itraconazole". The American Society of Health-System Pharmacists. Archived from the original on 10 December 2017. Retrieved 8 December 2017.
- ^ "Sporanox 10 mg/ml Oral Solution - Summary of Product Characteristics (SmPC)". (emc). 1 February 2018. Archived from the original on 13 April 2021. Retrieved 15 May 2020.
- ^ Isoherranen, N; Kunze, KL; Allen, KE; Nelson, WL; Thummel, KE (October 2004). "Role of Itraconazole Metabolites in CYP3A4 Inhibition". Drug Metabolism and Disposition. 32 (10): 1121–31. doi:10.1124/dmd.104.000315. PMID 15242978.
- ^ "Sporanox (itraconazole) Capsules. Full Prescribing Information" (PDF). Janssen Pharmaceuticals, Inc. Archived from the original (PDF) on 17 May 2018. Retrieved 28 August 2016.
- ^ Cite error: The named reference
MSF2020was invoked but never defined (see the help page). - ^ Cite error: The named reference
WHO2020DDDwas invoked but never defined (see the help page). - ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 503. ISBN 9783527607495. Archived from the original on 2020-08-02. Retrieved 2021-01-22.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Single Drug Information". International Medical Products Price Guide. Archived from the original on 10 December 2017. Retrieved 9 December 2017.
- ^ "NADAC (National Average Drug Acquisition Cost) | Data.Medicaid.gov". Centers for Medicare and Medicaid Services. Archived from the original on 20 March 2021. Retrieved 29 January 2021.
- ^ BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 632-633. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link)