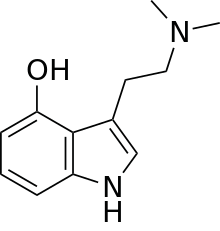
Psilocin (also known as 4-HO-DMT, 4-hydroxy DMT, psilocine, psilocyn, or psilotsin) is a substituted tryptamine alkaloid and a serotonergic psychedelic substance. It is present in most psychedelic mushrooms[3] together with its phosphorylated counterpart psilocybin. Psilocin is a Schedule I drug under the Convention on Psychotropic Substances.[4] Acting on the 5-HT2A serotonin receptors, psilocin’s psychedelic effects are directly correlated with the drug's occupancy at these receptor sites.[5] The subjective mind-altering effects of psilocin are highly variable and are said to resemble those of LSD and DMT.
 | |
 | |
| Clinical data | |
|---|---|
| Other names | 4-hydroxy-N,N-dimethyltryptamine |
| Routes of administration | Oral, IV |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | 1-3 hours[2] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.007.543 |
| Chemical and physical data | |
| Formula | C12H16N2O |
| Molar mass | 204.273 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 173 to 176 °C (343 to 349 °F) |
| |
| |
| | |
Chemistry
editPsilocin and its phosphorylated cousin, psilocybin, were first isolated and named in 1958 by Swiss chemist Albert Hofmann. Hofmann obtained the chemicals from laboratory-grown specimens of the entheogenic mushroom Psilocybe mexicana. Hofmann also succeeded in finding synthetic routes to these chemicals.[6]
Psilocin can be obtained by dephosphorylation of natural psilocybin under strongly acidic or under alkaline conditions (hydrolysis). A synthetic route uses the Speeter–Anthony tryptamine synthesis procedure. First, 4-hydroxyindole is Friedel-Crafts-acylated with oxalyl chloride in position 3. The compound is further reacted with dimethylamine, yielding the indole-3-yl-glyoxamide. Finally, this 4-hydroxyindole-3-N,N-dimethylglyoxamide is reduced by lithium aluminum hydride yielding psilocin.[7]
Psilocin is relatively unstable in solution due to its phenolic hydroxy (-OH) group. In the presence of oxygen, it readily forms bluish and dark black degradation products.[8] Similar products are also formed in the presence of oxygen and Fe3+ ions.
Structural analogs
editSulfur analogs are known with a benzothienyl replacement[9] as well as 4-SH-DMT.[10] 1-Methylpsilocin is a functionally 5-HT2C receptor preferring agonist.[11] 4-Fluoro-N,N-dimethyltryptamine is known.[11] O-Acetylpsilocin (4-AcO-DMT) is the ester of acetic acid with psilocin. Additionally, replacement of a methyl group at the dimethylated nitrogen with an isopropyl or ethyl group yields 4-HO-MIPT (4-hydroxy-N-methyl-N-isopropyltryptamine) and 4-HO-MET (4-hydroxy-N-methyl-N-ethyltryptamine), respectively. 4-Acetoxy-MET (4-Acetoxy-N-methyl-N-ethyltryptamine), also known as 4-AcO-MET, is the acetate ester of 4-HO-MET, and a homologue of 4-AcO-DMT.
Pharmacology
editPsilocin is the pharmacologically active agent in the body after ingestion of psilocybin or some species of psychedelic mushrooms.
Psilocybin is rapidly dephosphorylated in the body to psilocin which acts as a 5-HT2A, 5-HT2C and 5-HT1A agonist or partial agonist. Psilocin exhibits functional selectivity in that it activates phospholipase A2 instead of activating phospholipase C as the endogenous ligand serotonin does. Psilocin is structurally similar to serotonin (5-HT),[12] differing only by the hydroxyl group being on the 4-position rather than the 5 and the dimethyl groups on the nitrogen. Its effects are thought to come from its agonist activity at 5-HT2A serotonin receptors in the prefrontal cortex.
Psilocin has no significant effect on dopamine receptors (unlike LSD) and only affects the noradrenergic system at very high dosages.[13]
Psilocin's half-life ranges from 1 to 3 hours.[2]
Behavioral and non-behavioral effects
editThis section relies largely or entirely upon a single source. (September 2022) |
Its physiological effects are similar to a sympathetic arousal state. Specific effects observed after ingestion can include but are not limited to tachycardia, dilated pupils, restlessness or arousal, euphoria, open and closed eye visuals (common at medium to high doses), synesthesia (e.g. hearing colours and seeing sounds), increased body temperature, headache, sweating and chills, and nausea. Psilocin acts as a 5-HT2A, 5-HT2C, and 5-HT1A receptor agonist or partial agonist. Such receptors are claimed to significantly regulate visuals, decision making, mood, decreased blood pressure, and heart rate.[12]
There has been no direct lethality associated with psilocin.[12][14] There has been no reported withdrawal syndrome when chronic use of this drug is ceased.[12][15] There is cross tolerance among psilocin, mescaline, LSD,[12][16] and other 5-HT2A, 5-HT2C, and 5-HT1A agonists due to down-regulation of these receptors.
Legal status
editThe United Nations Convention on Psychotropic Substances (adopted in 1971) requires its members to prohibit psilocybin, and parties to the treaty are required to restrict the use of the drug to medical and scientific research under strictly controlled conditions.
Australia
editPsilocin is considered a Schedule 9 prohibited substance in Australia under the Poisons Standard (October 2015).[17] A Schedule 9 substance is a substance which may be abused or misused, the manufacture, possession, sale or use of which should be prohibited by law except when required for medical or scientific research, or for analytical, teaching or training purposes with approval of Commonwealth and/or State or Territory Health Authorities.[17]
Russia
editPsilocin and psilocybin are banned in Russia, due to their status as narcotic drugs, with a criminal penalty for possession of more than 50 mg.[18]
See also
editReferences
edit- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ^ a b Tylš F, Páleníček T, Horáček J (March 2014). "Psilocybin--summary of knowledge and new perspectives". European Neuropsychopharmacology. 24 (3): 342–356. doi:10.1016/j.euroneuro.2013.12.006. PMID 24444771. S2CID 10758314.
- ^ Gotvaldová K, Borovička J, Hájková K, Cihlářová P, Rockefeller A, Kuchař M (November 2022). "Extensive Collection of Psychotropic Mushrooms with Determination of Their Tryptamine Alkaloids". International Journal of Molecular Sciences. 23 (22): 14068. doi:10.3390/ijms232214068. PMC 9693126. PMID 36430546.
- ^ "List of psychotropic substances under international control" (PDF) (23rd ed.). Vienna Austria: International Narcotics Control Board. August 2003. Archived from the original (PDF) on 4 February 2012. Retrieved 2012-10-11.
- ^ Madsen MK, Fisher PM, Burmester D, Dyssegaard A, Stenbæk DS, Kristiansen S, et al. (June 2019). "Psychedelic effects of psilocybin correlate with serotonin 2A receptor occupancy and plasma psilocin levels". Neuropsychopharmacology. 44 (7): 1328–1334. doi:10.1038/s41386-019-0324-9. PMC 6785028. PMID 30685771.
- ^ Hofmann A, Heim R, Brack A, Kobel H, Frey A, Ott H, Petrzilka T, Troxler F (1959). "Psilocybin und Psilocin, zwei psychotrope Wirkstoffe aus mexikanischen Rauschpilzen" [Psilocybin and psilocin, two psychotropic substances in Mexican magic mushrooms]. Helvetica Chimica Acta (in German). 42 (5): 1557–72. doi:10.1002/hlca.19590420518.
- ^ Kargbo RB, Sherwood A, Walker A, Cozzi NV, Dagger RE, Sable J, et al. (July 2020). "Direct Phosphorylation of Psilocin Enables Optimized cGMP Kilogram-Scale Manufacture of Psilocybin". ACS Omega. 5 (27): 16959–16966. doi:10.1021/acsomega.0c02387. PMC 7364850. PMID 32685866. S2CID 220599227.
- ^ Lenz C, Wick J, Braga D, García-Altares M, Lackner G, Hertweck C, et al. (January 2020). "Injury-Triggered Blueing Reactions of Psilocybe "Magic" Mushrooms". Angewandte Chemie. 59 (4): 1450–1454. doi:10.1002/anie.201910175. PMC 7004109. PMID 31725937.
- ^ Chapman NB, Scrowston RM, Sutton TM (1972). "Synthesis of the sulphur analogue of psilocin and some related compounds". Journal of the Chemical Society, Perkin Transactions 1: 3011–15. doi:10.1039/P19720003011.
- ^ CH 421960, Hofmann A, Troxler F, issued 1967; CA 68:95680n
- ^ a b Sard H, Kumaran G, Morency C, Roth BL, Toth BA, He P, Shuster L (October 2005). "SAR of psilocybin analogs: discovery of a selective 5-HT 2C agonist". Bioorganic & Medicinal Chemistry Letters. 15 (20): 4555–4559. doi:10.1016/j.bmcl.2005.06.104. PMID 16061378.
- ^ a b c d e Diaz J (1996). How Drugs Influence Behavior: A Neurobehavioral Approach. Englewood Cliffs: Prentice Hall. ISBN 978-0-02-328764-0.
- ^ Jerome L (March–April 2007). "Psilocybin Investigator's Brochure" (PDF). Retrieved 2012-10-11.
- ^ Garcia-Romeu A, Kersgaard B, Addy PH (August 2016). "Clinical applications of hallucinogens: A review". Experimental and Clinical Psychopharmacology. 24 (4): 229–68. doi:10.1037/pha0000084. PMC 5001686. PMID 27454674.
- ^ Assessing Drug Risks: A Scientific Framework. European Monitoring Centre for Drugs and Drug Addiction. Luxembourg: EMCDDA. 2016.
- ^ Nichols DE (April 2016). "Psychedelics". Pharmacological Reviews. 68 (2): 264–355. doi:10.1124/pr.115.011478. PMC 4813425. PMID 26841800.
- ^ a b "Poisons Standard". Therapeutics Goods Administration, Department of Health. Australian Government. October 2015.
- ^ "On approval of significant, large and particularly large amounts of narcotic drugs and psychotropic substances, as well as significant, large and particularly large sizes for plants containing narcotic drugs or psychotropic substances, or parts thereof, containing narcotic drugs or psychotropic substances for the purposes of articles 228, 228.1, 229 and 229.1 of the Criminal Code of the Russian Federation (as amended) (translated)". Resolution of the Government of the Russian Federation. Criminal Code of the Russian Federation. 1 October 2012. 1002. Retrieved 1 April 2018.