 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɪˈfævɪrɛnz/ i-FAV-i-renz |
| Trade names | Sustiva, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699004 |
| License data | |
| Routes of administration | By mouth (capsules, tablets) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 40–45% (under fasting conditions) |
| Protein binding | 99.5–99.75% |
| Metabolism | Liver (CYP2A6 and CYP2B6-mediated) |
| Onset of action | 3–5 hours |
| Elimination half-life | 40–55 hours |
| Excretion | Urine (14–34%) and feces (16–61%) |
| Identifiers | |
| |
| Chemical and physical data | |
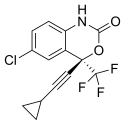
| Formula | C14H9ClF3NO2 |
| Molar mass | 315.68 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Efavirenz (EFV), sold under the brand names Sustiva among others, is an antiretroviral medication used to treat and prevent HIV/AIDS.[1] It is generally recommended for use with other antiretrovirals.[1] It may be used for prevention after a needlestick injury or other potential exposure.[1] It is sold both by itself and in combination as efavirenz/emtricitabine/tenofovir.[1] It is taken by mouth once a day.[1]
Common side effects include rash, nausea, headache, feeling tired, and trouble sleeping.[1] Some of the rashes may be serious such as Stevens–Johnson syndrome.[1] Other serious side effects include depression, thoughts of suicide, liver problems, and seizures.[1] It is not safe for use during pregnancy.[1] It is a non-nucleoside reverse transcriptase inhibitor (NNRTI) and works by blocking the function of reverse transcriptase.[1]
Efavirenz was approved for medical use in the United States in 1998.[1] It is on the World Health Organization's List of Essential Medicines.[4] As of 2016, it is available as a generic medication.[5][6] The wholesale cost in the developing world is about US$3.27 to 9.15 per month.[7] As of 2015 the cost for a typical month of medication in the United States is more than US$200.[8]
References
edit- ^ a b c d e f g h i j k l "Efavirenz". The American Society of Health-System Pharmacists. Archived from the original on 17 November 2016. Retrieved 28 November 2016.
- ^ Cite error: The named reference
MSF2020was invoked but never defined (see the help page). - ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 30 July 2020. Retrieved 21 September 2020.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Efavirenz Drug Profile". DrugPatentWatch. Archived from the original on 2016-11-10. Retrieved 2016-11-09.
- ^ "Efavirenz: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 31 July 2020. Retrieved 25 April 2020.
- ^ "Efavirenz". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 28 November 2016.
- ^ Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 62. ISBN 9781284057560.