 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Velcade, Chemobort, Bortecad, others |
| Other names | PS-341 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607007 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Subcutaneous, IV |
| Drug class | Anti-cancer medication[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 83% |
| Metabolism | Liver, CYP extensively involved |
| Elimination half-life | 9 to 15 hours |
| Identifiers | |
| |
| Chemical and physical data | |
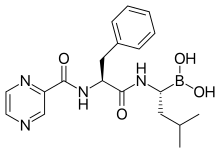
| Formula | C19H25BN4O4 |
| Molar mass | 384.24 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Bortezomib, sold under the brand name Velcade among others, is an anti-cancer medication used to treat multiple myeloma and mantle cell lymphoma.[1] This includes multiple myeloma in those who have and have not previously received treatment.[3] It is generally used together with other medications.[3] It is given by injection.[1]
Common side effects include nausea, diarrhea, tiredness, low platelets, fever, numbness, low white blood cells, shortness of breath, rash, and abdominal pain.[1] Other severe side effects include low blood pressure, tumour lysis syndrome, heart failure, and reversible posterior leukoencephalopathy syndrome.[1][3] It is in the class of medications known as proteasome inhibitor.[1] It works by inhibiting proteasomes, cellular complexes that break down proteins.[3]
Bortezomib was approved for medical use in the United States in 2003 and in Europe in 2004.[1][3] It is on the World Health Organization's List of Essential Medicines.[4] In the United States it costs US$1,360 per 3.5 mg vial.[5] In the United Kingdom this amount costs the NHS £762 such that a course of treatment is about £12,261 as of 2014.[6]
References edit
- ^ a b c d e f g h i "Bortezomib Monograph for Professionals". Drugs.com. Retrieved 13 October 2019.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Retrieved 11 September 2020.
- ^ a b c d e "Velcade". European Medicines Agency. 17 September 2018. Retrieved 13 October 2019.
- ^ World Health Organization (2019). "World Health Organization model list of essential medicines: 21st list 2019". World Health Organization (WHO). hdl:10665/325771.
{{cite journal}}: Cite journal requires|journal=(help) - ^ "Bortezomib Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 13 October 2019.
- ^ "Bortezomib for induction therapy in multiple myeloma before high-dose chemotherapy and autologous stem cell transplantation". NICE. February 2014. p. 2. Retrieved 13 October 2019.