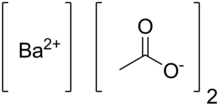
Barium acetate (Ba(C2H3O2)2) is the salt of barium(II) and acetic acid. Barium acetate is toxic to humans, but it has use in chemistry and manufacturing.
| |
| Names | |
|---|---|
| IUPAC name
Barium acetate
| |
| Other names
Barium diacetate
| |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | Ba(OAc)2 |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.008.045 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H6BaO4 | |
| Molar mass | 255.415 g·mol−1 |
| Appearance | White solid |
| Odor | odorless |
| Density | 2.468 g/cm3 (anhydrous) 2.19 g/cm3 (monohydrate) |
| Melting point | 450 °C (842 °F; 723 K) decomposes |
| 55.8 g/100 mL (0 °C) 72 g/100mL (20 °C) | |
| Solubility | slightly soluble in ethanol, methanol |
| -100.1·10−6 cm3/mol (2H2O) | |
| Structure | |
| tetragonal | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Toxic, hazardous on ingestion |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
108 mg/kg (oral, rat) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Preparation
editBarium acetate is generally produced by the reaction of acetic acid with barium carbonate:[2]
The reaction is performed in solution and the barium acetate crystalizes out at temperatures above 41 °C. Between 25 and 40 °C, the monohydrate version crystalizes. Alternatively, barium sulfide can be used:[2]
Again, the solvent is evaporated off and the barium acetate crystallized.
Properties
editBarium acetate is a white powder, which is highly soluble: at 0 °C, 55.8 g of barium acetate can be dissolved in 100 g of water. It decomposes upon heating into barium carbonate.[citation needed]
Reactions
editWhen heated in air, barium acetate decomposes to the carbonate. It reacts with acids: reaction with sulfuric acid, hydrochloric acid and nitric acid give the sulfate, chloride and nitrate respectively.[citation needed]
Uses
editBarium acetate is used as a mordant for printing textile fabrics, for drying paints and varnishes and in lubricating oil. In chemistry, it is used in the preparation of other acetates; and as a catalyst in organic synthesis.[citation needed]
In pop culture
editBarium acetate was featured in a 2001 episode of the television series Forensic Files, recounting the 1993 murder of a man by his teenage daughter (Marie Robards), though the episode and other crime documentary shows examining the Robards case excluded mention of the name of the chemical.
Barium acetate was featured in a 2014 episode of the crime documentary series Redrum.
Barium acetate was named as the choice poison of a teen's murder of her father in an episode of Deadly Women.[citation needed]
References
edit- ^ [1], JT Baker
- ^ a b Barium acetate Archived June 28, 2009, at the Wayback Machine, hillakomem.com, retrieved 30 June 2009
Further reading
edit- I. Gautier-Luneau; A. Mosset (1988). "Crystal structure of anhydrous barium acetate". Journal of Solid State Chemistry. 73 (2): 473–479. Bibcode:1988JSSCh..73..473G. doi:10.1016/0022-4596(88)90133-8.
- After husband's body was found burned, woman is suspected of poisoning another man

