 | |
| Combination of | |
|---|---|
| Sacubitril | Neprilysin inhibitor |
| Valsartan | Angiotensin II receptor antagonist |
| Clinical data | |
| Trade names | Entresto, Azmarda, Neparvis, others |
| Other names | LCZ696 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a615039 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Angiotensin receptor-neprilysin inhibitor (ARNi)[2] |
| Legal status | |
| Legal status | |
| Chemical and physical data | |
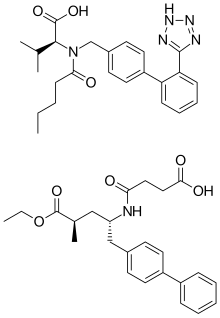
| Formula | C96H120N12Na6O21 |
| Molar mass | 1916.018 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Sacubitril/valsartan, sold under the brand name Entresto among others, is a combination medication used for heart failure with reduced ejection fraction.[7] It may be used to replace an ACE inhibitor or an angiotensin receptor blocker.[7] It is taken by mouth.[7]
Common side effects include low blood pressure, cough, kidney problems, and high potassium.[2] Other side effects may include angioedema.[2] Use in pregnancy may harm the baby.[2] It contains sacubitril, a neprilysin inhibitor, and valsartan, a angiotensin receptor blocker.[2] The combination is sometimes known as an "angiotensin receptor-neprilysin inhibitor" (ARNi).[2]
Sacubitril/valsartan was approved for medical use in the United States and Europe in 2015.[5][2] It was approved in Australia in 2016.[1] In the United States it costs about 575 USD per month as of 2021.[9] This amount in the United Kingdom costs the NHS about £92.[7]
References
edit- ^ a b c "Entresto 24/26 tablets, Entresto 49/51 tablets, Entresto 97/103 tablets (sacubitril/valsartan) Product Information". Therapeutic Goods Administration (TGA). Novartis. Archived from the original on 11 November 2020. Retrieved 21 September 2020.
- ^ a b c d e f g h "Sacubitril and Valsartan Monograph for Professionals". Drugs.com. Archived from the original on 4 January 2020. Retrieved 9 October 2021.
- ^ "Entresto 24 mg/26 mg film-coated tablets - Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 24 January 2021. Retrieved 21 September 2020.
- ^ "Entresto- sacubitril and valsartan tablet, film coated". DailyMed. 14 June 2020. Archived from the original on 19 April 2020. Retrieved 21 September 2020.
- ^ a b "Entresto EPAR". European Medicines Agency (EMA). Archived from the original on 27 February 2021. Retrieved 21 September 2020.
- ^ "Neparvis EPAR". European Medicines Agency (EMA). Archived from the original on 28 December 2020. Retrieved 23 September 2020.
- ^ a b c d e BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 207. ISBN 978-0-85711-369-6.
- ^ "Sacubitril / valsartan (Entresto) Use During Pregnancy". Drugs.com. 20 November 2019. Archived from the original on 17 April 2021. Retrieved 21 September 2020.
- ^ "Entresto Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 9 October 2021.