 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Demerol, others |
| Other names | Meperidine (USAN US) |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Dependence liability | High |
| Routes of administration | By mouth, IV, IM, IT,[1] SC, epidural[2] |
| Drug class | Opioid[3] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 50–60% (Oral), 80–90% (Oral, in cases of hepatic impairment) |
| Protein binding | 65–75% |
| Metabolism | Liver |
| Onset of action | < 1 hr[3] |
| Elimination half-life | 2.5–4 hours, 7–11 hours (liver disease) |
| Duration of action | 2 to 4 hrs[3] |
| Excretion | Kidney |
| Identifiers | |
| |
| Chemical and physical data | |
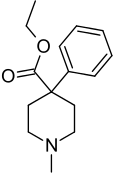
| Formula | C15H21NO2 |
| Molar mass | 247.338 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Pethidine, also known as meperidine and sold under the brand name Demerol among others, is a pain medication of the opioid type.[3] Specifically it may be used for moderate to severe pain for a few days.[3] It is not a first line opioid and long term use is not recommended.[3] It can be used mouth, or by injection into a muscle, or vein.[3] It; however, is not very effective by mouth and such use is associated with greater toxicity.[3] Onset of effects is within an hour and last for up to 4 hours.[3]
Common side effects include sleepiness, seizures, agitation, vomiting, constipation, and decreased effort to breath.[3] Other serious complications include heart arrhythmias, psychosis, addiction, low blood pressure, and serotonin syndrome.[3] Prolonged use during pregnancy may result in neonatal abstinence syndrome in the baby.[4] Pethidine works by binding and activating the opiate receptor.[3]
Pethidine was patented in 1937 and approved for medical use in 1943.[5] In the 20th century it was a commonly used opioid; in 1975, 60% of doctors prescribed it for acute pain and 22% for chronic severe pain.[6] In the 2000s its use has become less common.[7] It is avaliable as a generic medication.[8] In the United Kingdom the injectable formulation costs the NHS about 0.5 pounds per 50 mg in 2020.[8]
References edit
- ^ Ngan Kee, WD (April 1998). "Intrathecal pethidine: pharmacology and clinical applications". Anaesthesia and Intensive Care. 26 (2): 137–46. doi:10.1177/0310057X9802600202. PMID 9564390.
- ^ Ngan Kee, WD (June 1998). "Epidural pethidine: pharmacology and clinical experience". Anaesthesia and Intensive Care. 26 (3): 247–55. doi:10.1177/0310057X9802600303. PMID 9619217.
- ^ a b c d e f g h i j k l m n "Meperidine Hydrochloride Monograph for Professionals". Drugs.com. Archived from the original on 28 August 2021. Retrieved 10 November 2020.
- ^ "Meperidine Use During Pregnancy". Drugs.com. Archived from the original on 28 October 2020. Retrieved 10 November 2020.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 526. ISBN 9783527607495. Archived from the original on 2021-04-15. Retrieved 2020-11-10.
- ^ Latta, KS; Ginsberg, B; Barkin, RL (January–February 2002). "Meperidine: a critical review". American Journal of Therapeutics. 9 (1): 53–68. doi:10.1097/00045391-200201000-00010. PMID 11782820. S2CID 23410891.
- ^ Murray, Michael J.; Rose, Steven H.; Wedel, Denise J.; Wass, C. Thomas; Harrison, Barry A.; Mueller, Jeff T.; Trentman, Terence L. (2014). Faust's Anesthesiology Review E-Book. Elsevier Health Sciences. p. 169. ISBN 978-1-4377-0367-2. Archived from the original on 2021-08-28. Retrieved 2020-11-10.
- ^ a b BNF 79 : March 2020. London: Royal Pharmaceutical Society. 2020. p. 481. ISBN 9780857113658.