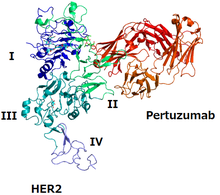
 The structure of HER2 and pertuzumab | |
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Humanized (from mouse) |
| Target | HER2 |
| Clinical data | |
| Trade names | Perjeta |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | Intravenous |
| Drug class | Monoclonal antibody[1] |
| Legal status | |
| Legal status |
|
| | |
Pertuzumab, sold under the brand name Perjeta, is a medication used to treat breast cancer.[1] Specifically it is used for HER2-positive cases in combination with trastuzumab and possibly docetaxel.[1] It is given by gradual injection into a vein.[1]
Common side effects include diarrhea, hair loss, low neutrophils, nausea, tiredness, rash, and peripheral nerve problems.[1] Other side effects may include heart failure, infusion reaction, and anaphylaxis.[2] Use during pregnancy may harm the baby.[2] It is a monoclonal antibody which attaches to HER2 on cells and blocks signals that cause the cancer to grow.[3]
Pertuzumab was approved for medical use in the United States in 2012 and Europe in 2013.[2][3] In the United Kingdom it costs the NHS about £2,400 per 420 mg as of 2021.[4] This amount in the United States costs about 5,800 USD.[5]
References
edit- ^ a b c d e f g "DailyMed - PERJETA- pertuzumab injection, solution, concentrate". dailymed.nlm.nih.gov. Archived from the original on 25 March 2021. Retrieved 27 October 2021.
- ^ a b c "Pertuzumab Monograph for Professionals". Drugs.com. Archived from the original on 27 February 2021. Retrieved 27 October 2021.
- ^ a b "Perjeta". Archived from the original on 16 September 2021. Retrieved 27 October 2021.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 926. ISBN 978-0857114105.
- ^ "Perjeta Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 27 October 2021.