 | |
| Clinical data | |
|---|---|
| Pronunciation | ne TAR soo dil |
| Trade names | Rhopressa, Rhokiinsa |
| Other names | AR-11324 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618014 |
| License data |
|
| Routes of administration | Eye drops, topical |
| Drug class | Rho kinase inhibitor[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Esterases in the cornea |
| Metabolites | AR-13503 (active metabolite) |
| Elimination half-life | 16–17 hrs |
| Duration of action | ≥ 24 hrs |
| Identifiers | |
| |
| Chemical and physical data | |
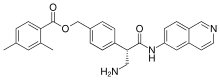
| Formula | C28H27N3O3 |
| Molar mass | 453.542 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Netarsudil, sold under the brand name Rhopressa and Rhokiinsa, is a medication used to treat increased intraocular pressure including open-angle glaucoma.[3][1] It is used as an eye drop.[3]
Common side effects include red eyes, conjunctival bleeding, blurry vision, and tearing.[1] Other side effects may include bacterial keratitis.[1] It is a rho kinase inhibitor.[1]
Netarsudil was approved for medical use in the United States in 2017 and Europe in 2019.[1][3] In the United States a 2.5 ml bottle costs about 305 USD as of 2021.[4] It is also available as the combination netarsudil/latanoprost.[1] It is not commercially available in Europe and the United Kingdom as of 2021.[5]
References
edit- ^ a b c d e f g h i "Netarsudil Mesylate Monograph for Professionals". Drugs.com. Archived from the original on 3 June 2020. Retrieved 12 November 2021.
- ^ "Rhopressa- netarsudil solution/ drops". DailyMed. Archived from the original on 25 March 2021. Retrieved 2 May 2021.
- ^ a b c d e "Rhokiinsa EPAR". European Medicines Agency (EMA). 16 September 2019. Archived from the original on 29 December 2019. Retrieved 27 September 2020.
- ^ "Rhopressa Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 12 November 2021.
- ^ "Netarsudil". SPS - Specialist Pharmacy Service. 25 January 2016. Archived from the original on 13 November 2021. Retrieved 12 November 2021.