 | |
| Clinical data | |
|---|---|
| Trade names | Relistor |
| Other names | Naltrexone-methyl-bromide, methylnaltrexone bromide |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608052 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous, subcutaneous |
| Drug class | Peripherally acting μ-opioid receptor antagonist[2] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 11–15.3% |
| Metabolism | Liver |
| Elimination half-life | 8 hours |
| Excretion | Urine (50%), faeces (50%) |
| Identifiers | |
| |
| Chemical and physical data | |
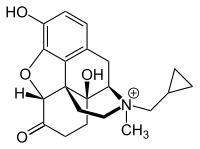
| Formula | C21H26NO4 |
| Molar mass | 356.442 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Methylnaltrexone (MNTX), sold under the brand name Relistor, is a medication used to treat constipation due to opioids.[3] It may be used when other laxatives are not effective.[2] It is taken by mouth or by injection under the skin.[3]
Common side effects include abdominal pain, diarrhea, dizziness, nausea, and opioid withdrawal.[2] Other side effects may include gastrointestinal perforation.[3] It a peripherally acting μ-opioid receptor antagonist, which means it does not generally affect the central pain reducing effects of other opioids.[2]
Methylnaltrexone was approved for medical use in the United States in 2008.[3] In the United Kingdom the injectable formulation costs the NHS about £21 per dose as of 2021.[2] In the United States this amount costs about 150 USD.[4]
References
edit- ^ a b "Methylnaltrexone (Relistor) Use During Pregnancy". Drugs.com. 9 July 2020. Archived from the original on 21 October 2020. Retrieved 21 September 2020.
- ^ a b c d e f BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 70. ISBN 978-0857114105.
- ^ a b c d e "Methylnaltrexone Monograph for Professionals". Drugs.com. Archived from the original on 21 January 2021. Retrieved 17 November 2021.
- ^ "Relistor Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 4 March 2016. Retrieved 17 November 2021.