 | |
| Clinical data | |
|---|---|
| Trade names | Nozinan, Levoprome, Detenler, Hirnamin, Levotomin, Neurocil, others |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | By mouth, IV, SC, IM[1] |
| Drug class | Typical antipsychotic |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~50–60% |
| Metabolism | Liver |
| Onset of action | 0.5 to 3 hr[2] |
| Elimination half-life | ~20 hours |
| Duration of action | 8 hr[2] |
| Excretion | In feces and urine (metabolites), unchanged drug only 1% |
| Identifiers | |
| |
| Chemical and physical data | |
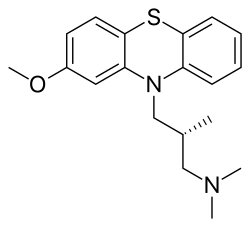
| Formula | C19H24N2OS |
| Molar mass | 328.47 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Levomepromazine, also known as methotrimeprazine, is a medication used for schizophrenia and palliative care.[1] In palliative care it is used for restlessness, pain, and vomiting.[1] It may be used by mouth or by injection.[1] Effects generally begin around 0.5 to 3 hours and last for 8 hours.[2]
Side effects may include low blood pressure with standing, sleepiness, dry mouth, liver problems, and dystonia.[2][1] Serious side effect may include priapism, QT prolongation, and neuroleptic malignant syndrome.[2][1][3] It is an antipsychotic of the phenothiazine type.[2] It works by blocking a variety of receptors, including adrenergic, dopamine, histamine, muscarinic acetylcholine, and serotonin.[2]
Levomepromazine was patented in 1954 and come into medical use in the United State in 1957.[4] It is available as a generic medication.[1] In the United Kingdom 84 tablets of 25 mg costs the NHS about £20 as of 2020.[1] It has been widely used;[2] though is no longer commercially available in the United States.[5]
References
edit- ^ a b c d e f g h i j BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 460. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ^ a b c d e f g h i Dietz, I; Schmitz, A; Lampey, I; Schulz, C (19 January 2013). "Evidence for the use of Levomepromazine for symptom control in the palliative care setting: a systematic review". BMC palliative care. 12: 2. doi:10.1186/1472-684X-12-2. PMID 23331515.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ "Levomepromazine Hydrochloride 25mg/ml Solution for Injection - Summary of Product Characteristics (SmPC) - (emc)". www.medicines.org.uk. Archived from the original on 28 August 2021. Retrieved 13 August 2021.
- ^ Buschmann, Helmut; Holenz, Jörg; Párraga, Antonio; Torrens, Antoni; Vela, José Miguel; Díaz, José Luis (16 April 2007). Antidepressants, Antipsychotics, Anxiolytics, 2 Volume Set: From Chemistry and Pharmacology to Clinical Application. John Wiley & Sons. p. 502. ISBN 978-3-527-31058-6. Archived from the original on 28 August 2021. Retrieved 13 August 2021.
- ^ "Drugs@FDA: FDA-Approved Drugs". www.accessdata.fda.gov. Archived from the original on 24 March 2021. Retrieved 13 August 2021.