 | |
| Clinical data | |
|---|---|
| Trade names | Baxdela, Quofenix |
| Other names | Delafloxacin meglumine; ABT-492; RX-3341; WQ-3034 |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | By mouth, intravenous injection |
| Drug class | Fluoroquinolone[1] |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| Chemical and physical data | |
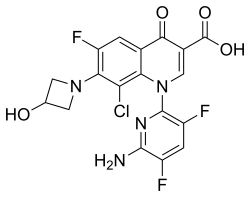
| Formula | C18H12ClF3N4O4 |
| Molar mass | 440.76 g·mol−1 |
| 3D model (JSmol) | |
| |
Delafloxacin sold under the brand name Baxdela among others, is an antibiotic used to treat bacterial skin and skin structure infections.[2] It is generally effective for MRSA and Pseudomonas.[1] It may be given by mouth or by injection into a vein.[2]
Common side effects include diarrhea and nausea.[2] Other side effects may include tendinitis, peripheral neuropathy, psychosis, Clostridioides difficile infection, and anaphylaxis.[1] Safety in pregnancy is unclear.[1] It is a fluoroquinolone.[1]
Delafloxacin was approved for medical use in the United States in 2017 and Europe in 2019.[1][2] In the United Kingdom five days of treatment costs the NHS about £615 as of 2021.[3] This amount in the United States is about 760 USD.[4]
References
edit- ^ a b c d e f g "Delafloxacin Monograph for Professionals". Drugs.com. Archived from the original on 21 January 2021. Retrieved 22 December 2021.
- ^ a b c d e "Quofenix EPAR". European Medicines Agency (EMA). 16 October 2019. Archived from the original on 5 August 2020. Retrieved 12 May 2020. This article incorporates text from this source, which is in the public domain.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 595. ISBN 978-0857114105.
- ^ "Baxdela Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 22 December 2021.