 | |
| Clinical data | |
|---|---|
| Pronunciation | ay" za sye' ti deen |
| Trade names | Vidaza, Azadine, Onureg |
| Other names | 5-Azacytidine, azacytidine, ladakamycin, 4-Amino-1-β-D-ribofuranosyl-s-triazin-2(1H)-one, U-18496, CC-486 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607068 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Subcutaneous injection, intravenous, by mouth |
| Drug class | Nucleoside analog[2] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 4 hr.[5] |
| Identifiers | |
| |
| Chemical and physical data | |
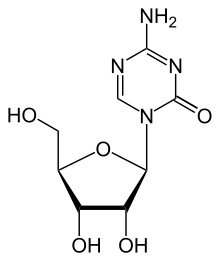
| Formula | C8H12N4O5 |
| Molar mass | 244.207 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Azacitidine, sold under the brand name Vidaza among others, is a medication used to treat myelodysplastic syndrome, acute myeloid leukaemia (AML), chronic myelomonocytic leukaemia.[6][7] It is given by injection into a vein or under the skin.[2]
Common side effects include nausea, low red blood cells, low platelets, fever, low white blood cells, diarrhea, low potassium, and bruises.[8] Other side effects may include liver problems, kidney problems, and tumor lysis syndrome.[8] Use in pregnancy may harm the baby.[8] It is a cytosine analogue which works by blocking DNA methylation.[6]
Azacitidine was approved for medical use in the United States in 2004 and Europe in 2008.[8][9] It is available as a generic medication.[7] In the United Kingdom 100 mg costs the NHS about £220 as of 2021.[7] In the United States this amount costs about 83 USD.[10]
References edit
- ^ "Azacitidine (Vidaza) Use During Pregnancy". Drugs.com. 5 May 2020. Archived from the original on 5 December 2020. Retrieved 12 August 2020.
- ^ a b "Azacitidine Monograph for Professionals". Drugs.com. Archived from the original on 5 March 2021. Retrieved 17 January 2022.
- ^ "Vidaza- azacitidine injection, powder, lyophilized, for solution". DailyMed. 2 March 2020. Archived from the original on 5 November 2021. Retrieved 27 September 2020.
- ^ "Onureg EPAR". European Medicines Agency. 20 April 2021. Archived from the original on 6 September 2021. Retrieved 6 September 2021.
- ^ Vallerand AH, Deglin JH (2009). Davis's drug guide for nurses. Philadelphia: F.A. Davis Company. pp. 204–206. ISBN 978-0-8036-1912-8.
- ^ a b c "Azacitidine". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 11 December 2021. Retrieved 17 January 2022.
- ^ a b c d BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 950. ISBN 978-0857114105.
- ^ a b c d e "DailyMed - VIDAZA- azacitidine injection, powder, lyophilized, for solution". dailymed.nlm.nih.gov. Archived from the original on 5 November 2021. Retrieved 17 January 2022.
- ^ "Vidaza". Archived from the original on 29 December 2021. Retrieved 17 January 2022.
- ^ "Azacitidine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 17 January 2022.