 | |
| Clinical data | |
|---|---|
| Trade names | Cibinqo |
| Other names | PF-04965842 |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Janus kinase inhibitor[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 2.8–5.2 h |
| Excretion | 1.0–4.4% unchanged in urine |
| Chemical and physical data | |
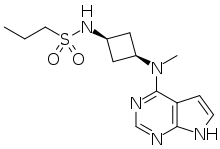
| Formula | C14H21N5O2S |
| Molar mass | 323.42 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Abrocitinib, sold under the brand name Cibinqo, is a medication used to treat atopic dermatitis (eczema).[3] It may be used in cases that do not improve with other treatments.[1] It is taken by mouth.[1]
Common side effects include nausea, headache, herpes simplex, urinary tract infection, acne, impetigo, and low platelets.[1] Other side effects may include infections, sudden cardiac death, pulmonary embolism, and cancer.[1] Safety in pregnancy is unclear.[1] It is a Janus kinase inhibitor.[1]
Abrocitinib was approved for medical use in Europe in 2021 and the United States in 2022.[3][1] In the United Kingdom 4 weeks of treatments cost the NHS about £900 as of 2021.[4] In the United States this amount costs about 4,600 USD as of 2022.[5]
References
edit- ^ a b c d e f g h i j k "Cibinqo- abrocitinib tablet, film coated". DailyMed. 15 February 2022. Archived from the original on 3 March 2022. Retrieved 3 March 2022.
- ^ "Cibinqo Product information". Health Canada. 25 April 2012. Archived from the original on 10 December 2022. Retrieved 30 September 2022.
- ^ a b c d e "Cibinqo EPAR". European Medicines Agency (EMA). 11 October 2021. Archived from the original on 18 December 2021. Retrieved 17 December 2021. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "Abrocitinib". SPS - Specialist Pharmacy Service. 24 January 2018. Archived from the original on 5 July 2022. Retrieved 11 December 2022.
- ^ "Cibinqo". Retrieved 11 December 2022.