Rhodium hexafluoride, also rhodium(VI) fluoride, (RhF6) is the inorganic compound of rhodium and fluorine. A black volatile solid,[1] it is a highly reactive material, and a rare example of a rhodium(VI) compound. It is one of seventeen known binary hexafluorides.
| |
| Names | |
|---|---|
| IUPAC name
rhodium(VI) fluoride
| |
| Other names
rhodium hexafluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| F6Rh | |
| Molar mass | 216.91 g/mol |
| Appearance | black crystalline solid[1] |
| Density | 3.71g/mL[2] |
| Melting point | ≈ 70 °C (158 °F; 343 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Synthesis, structure, properties
editRhodium hexafluoride is prepared by reaction of rhodium metal with an excess of elemental fluorine:[3]
- Rh + 3 F2 → RhF6
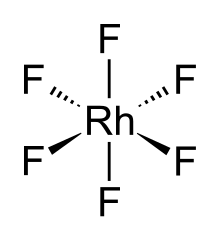
The RhF6 molecule has octahedral molecular geometry. Consistent with its d3 configuration, the six Rh–F bond lengths are equivalent, being 1.824 Å.[2] It crystallises in an orthorhombic space group Pnma with lattice parameters of a = 9.323 Å, b = 8.474 Å, and c = 4.910 Å.
Like some other metal fluorides, RhF6 is highly oxidizing. It attacks glass,[3] and can even react with elemental oxygen.[4]
References
edit- ^ a b c CRC Handbook of Chemistry and Physics, 90th Edition, CRC Press, Boca Raton, Florida, 2009, ISBN 978-1-4200-9084-0, Section 4, Physical Constants of Inorganic Compounds, p. 4-85.
- ^ a b Drews, T.; Supeł, J.; Hagenbach, A.; Seppelt, K. (2006). "Solid State Molecular Structures of Transition Metal Hexafluorides". Inorganic Chemistry. 45 (9): 3782–3788. doi:10.1021/ic052029f. PMID 16634614.
- ^ a b 《无机化学丛书》第九卷:锰分族、铁系、铂系 (in Chinese). 北京: 科学出版社. 1991. p. 478. ISBN 7-03-002238-6.
- ^ Riedel, Sebastian; Kaupp, Martin (2009). "The highest oxidation states of the transition metal elements" (PDF). Coordination Chemistry Reviews. 253 (5–6). Elsevier: 606–624. doi:10.1016/j.ccr.2008.07.014.[permanent dead link]
Further reading
edit- Gmelins Handbuch der anorganischen Chemie, System Nr. 63, Rhodium, Part B1, pp. 266–268.
External links
edit- Media related to Rhodium hexafluoride at Wikimedia Commons
- Rhodium hexafluoride at webelements.com.