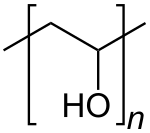
Polyvinyl alcohol (PVOH, PVA, or PVAl) is a water-soluble synthetic polymer. It has the idealized formula [CH2CH(OH)]n. It is used in papermaking, textile warp sizing, as a thickener and emulsion stabilizer in polyvinyl acetate (PVAc) adhesive formulations, in a variety of coatings, and 3D printing. It is colourless (white) and odorless. It is commonly supplied as beads or as solutions in water.[3][4] Without an externally added crosslinking agent, PVA solution can be gelled through repeated freezing-thawing, yielding highly strong, ultrapure, biocompatible hydrogels which have been used for a variety of applications such as vascular stents, cartilages, contact lenses, etc.[5]
| |

| |
| Names | |
|---|---|
| Other names
PVOH; Poly(Ethenol), Ethenol, homopolymer; PVA; Polyviol; Vinol; Alvyl; Alcotex; Covol; Gelvatol; Lemol; Mowiol; Mowiflex, Alcotex, Elvanol, Gelvatol, Lemol, Nelfilcon A, Polyviol und Rhodoviol
| |
| Identifiers | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.121.648 |
| E number | E1203 (additional chemicals) |
| KEGG | |
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C2H4O)x | |
| Density | 1.19–1.31 g/cm3 |
| Melting point | 200 °C (392 °F; 473 K) |
| log P | 0.26 [1] |
Refractive index (nD)
|
1.477 @ 632 nm[2] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 79.44 °C (174.99 °F; 352.59 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
14,700 mg/kg (mouse) |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Although polyvinyl alcohol is often referred to by the acronym PVA, more generally PVA refers to polyvinyl acetate, which is commonly used as a wood adhesive and sealer.
Uses edit
PVA is used in a variety of medical applications because of its biocompatibility, low tendency for protein adhesion, and low toxicity. Specific uses include cartilage replacements, contact lenses, and eye drops.[6] Polyvinyl alcohol is used as an aid in suspension polymerizations. Its largest application in China is its use as a protective colloid to make PVAc dispersions. In Japan its major use is the production of Vinylon fiber.[7] This fiber is also manufactured in North Korea for self-sufficiency reasons, because no oil is required to produce it. Another application is photographic film.[8]
PVA-based polymers are used widely in additive manufacturing. For example, 3D printed oral dosage forms demonstrate great potential in the pharmaceutical industry. It is possible to create drug-loaded tablets with modified drug-release characteristics where PVA is used as a binder substance.[9]
Medically, PVA-based microparticles have received FDA 510(k) approval to be used as embolisation particles to be used for peripheral hypervascular tumors.[10] It may also used as the embolic agent in a Uterine Fibroid Embolectomy (UFE).[11] In biomedical engineering research, PVA has also been studied for cartilage, orthopaedic applications,[12] and potential materials for vascular graft.[13]
PVA is commonly used in household sponges that absorb more water than polyurethane sponges.
Polyvinyl acetals edit
Polyvinyl acetals are prepared by treating PVA with aldehydes. Butyraldehyde and formaldehyde afford polyvinyl butyral (PVB) and polyvinyl formal (PVF), respectively. Preparation of polyvinyl butyral is the largest use for polyvinyl alcohol in the US and Western Europe.
Preparation edit
Unlike most vinyl polymers, PVA is not prepared by polymerization of the corresponding monomer, since the monomer, vinyl alcohol, is thermodynamically unstable with respect to its tautomerization to acetaldehyde. Instead, PVA is prepared by hydrolysis of polyvinyl acetate,[3] or sometimes other vinyl ester-derived polymers with formate or chloroacetate groups instead of acetate. The conversion of the polyvinyl esters is usually conducted by base-catalysed transesterification with ethanol:
- [CH2CH(OAc)]n + C2H5OH → [CH2CH(OH)]n + C2H5OAc
The properties of the polymer are affected by the degree of transesterification.
Worldwide consumption of polyvinyl alcohol was over one million metric tons in 2006.[7] Large producers include Kuraray and Sekisui Specialty Chemicals, while mainland China has installed a number of very large production facilities in the past decade[clarification needed] and currently accounts for 45% of world capacity.
Structure and properties edit
PVA is an atactic material that exhibits crystallinity. In terms of microstructure, it is composed mainly of 1,3-diol linkages [−CH2−CH(OH)−CH2−CH(OH)−], but a few percent of 1,2-diols [−CH2−CH(OH)−CH(OH)−CH2−] occur, depending on the conditions for the polymerization of the vinyl ester precursor.[3]
Polyvinyl alcohol has excellent film-forming, emulsifying and adhesive properties. It is also resistant to oil, grease and solvents. It has high tensile strength and flexibility, as well as high oxygen and aroma barrier properties. However, these properties are dependent on humidity: water absorbed at higher humidity levels acts as a plasticiser, which reduces the polymer's tensile strength, but increases its elongation and tear strength.
Safety and environmental considerations edit
Polyvinyl alcohol is widely used, thus its toxicity and biodegradation are of interest. Tests showed that fish (guppies) are not harmed, even at a poly(vinyl alcohol) concentration of 500 mg/L of water.[3]
There are several different grades of PVA depending on the degrees of polymerization and hydrolysis, which will affect their physical and chemical properties as well as their biodegradability.[3] Aqueous solutions of PVA degrade faster, which is why PVA grades that are highly water-soluble tend to have a faster biodegradation.[14] Not all PVA grades are readily biodegradable, but studies show that high water-soluble PVA grades such as the ones used in detergents can be readily biodegradable according to OECD screening test conditions.[15]
Orally administered PVA is relatively harmless.[16] The safety of polyvinyl alcohol is based on some of the following observations:[16]
- The acute oral toxicity of polyvinyl alcohol is very low, with LD(50)s in the range of 15-20 g/kg;
- Orally administered PVA is very poorly absorbed from the gastrointestinal tract;
- PVA does not accumulate in the body when administered orally;
- Polyvinyl alcohol is not mutagenic or clastogenic
See also edit
References edit
- ^ "Poly(vinyl alcohol)". ChemSrc.
- ^ Schnepf MJ, Mayer M, Kuttner C, et al. (July 2017). "Nanorattles with tailored electric field enhancement". Nanoscale. 9 (27): 9376–9385. doi:10.1039/C7NR02952G. hdl:10067/1447970151162165141. PMID 28656183.
- ^ a b c d e Hallensleben ML (2000). "Polyvinyl Compounds, Others". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_743. ISBN 978-3527306732.
- ^ Tang X, Alavi S (2011). "Recent Advances in Starch, Polyvinyl Alcohol Based Polymer Blends, Nanocomposites and Their Biodegradability". Carbohydrate Polymers. 85: 7–16. doi:10.1016/j.carbpol.2011.01.030.
- ^ Adelnia, Hossein; Ensandoost, Reza; Shebbrin Moonshi, Shehzahdi; et al. (2022-02-05). "Freeze/thawed polyvinyl alcohol hydrogels: Present, past and future". European Polymer Journal. 164: 110974. doi:10.1016/j.eurpolymj.2021.110974. hdl:10072/417476. ISSN 0014-3057. S2CID 245576810.
- ^ Baker MI, Walsh SP, Schwartz Z, Boyan BD (July 2012). "A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications". Journal of Biomedical Materials Research Part B: Applied Biomaterials. 100 (5): 1451–7. doi:10.1002/jbm.b.32694. PMID 22514196.
- ^ a b SRI Consulting CEH Report Polyvinyl Alcohol, published March 2007, abstract retrieved July 30, 2008.
- ^ Lampman, Steve, ed. (2003). "Effects of Composition, Processing, and Structure on Properties of Engineering Plastics". Characterization and Failure Analysis of Plastics. ASM International. p. 29. doi:10.31399/asm.tb.cfap.t69780028. ISBN 978-0-87170-789-5.
- ^ Xu X, Zhao J, Wang M, et al. (August 2019). "3D Printed Polyvinyl Alcohol Tablets with Multiple Release Profiles". Scientific Reports. 9 (1): 12487. Bibcode:2019NatSR...912487X. doi:10.1038/s41598-019-48921-8. PMC 6713737. PMID 31462744.
- ^ "Contour™ - Brief Summary". www.bostonscientific.com. Retrieved 2023-08-11.
- ^ Siskin GP. Cho KJ (ed.). "Uterine Fibroid Embolization and Imaging". Medscape. WebMD LLC. Archived from the original on 2015-03-04.
- ^ Baker, Maribel I.; Walsh, Steven P.; Schwartz, Zvi; Boyan, Barbara D. (July 2012). "A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications". Journal of Biomedical Materials Research Part B: Applied Biomaterials. 100B (5): 1451–1457. doi:10.1002/jbm.b.32694. PMID 22514196.
- ^ Chaouat, Marc; Le Visage, Catherine; Baille, Wilms E.; Escoubet, Brigitte; Chaubet, Frédéric; Mateescu, Mircea Alexandru; Letourneur, Didier (2008-10-09). "A Novel Cross-linked Poly(vinyl alcohol) (PVA) for Vascular Grafts". Advanced Functional Materials. 18 (19): 2855–2861. doi:10.1002/adfm.200701261. S2CID 42332293.
- ^ Kawai F, Hu X (August 2009). "Biochemistry of microbial polyvinyl alcohol degradation". Applied Microbiology and Biotechnology. 84 (2): 227–37. doi:10.1007/s00253-009-2113-6. PMID 19590867. S2CID 25068302.
- ^ Byrne, Dominic, Boeije, Geert, Croft, Ian, Hüttmann, Gerd, Luijkx, Gerard, Meier, Frank, Parulekar, Yash and Stijntjes, Gerard. "Biodegradability of Polyvinyl Alcohol Based Film Used for Liquid Detergent Capsules: Biologische Abbaubarkeit der für Flüssigwaschmittelkapseln verwendeten Folie auf Polyvinylalkoholbasis." Tenside Surfactants Detergents, vol. 58, no. 2, 2021, pp. 88-96. https://doi.org/10.1515/tsd-2020-2326
- ^ a b DeMerlis, C. C.; Schoneker, D. R. (March 2003). "Review of the oral toxicity of polyvinyl alcohol (PVA)". Food and Chemical Toxicology. 41 (3): 319–326. doi:10.1016/s0278-6915(02)00258-2. ISSN 0278-6915. PMID 12504164. Retrieved 17 January 2024.
