Nitrosyl chloride is the chemical compound with the formula NOCl. It is a yellow gas that is commonly encountered as a component of aqua regia, a mixture of 3 parts concentrated hydrochloric acid and 1 part of concentrated nitric acid. It is a strong electrophile and oxidizing agent. It is sometimes called Tilden's reagent, after William A. Tilden, who was the first to produce it as a pure compound.[1]
| |

| |
| Names | |
|---|---|
| IUPAC name
Nitrosyl chloride[citation needed]
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.018.430 |
| EC Number |
|
| E number | E919 (glazing agents, ...) |
| MeSH | nitrosyl+chloride |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1069 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| NOCl | |
| Molar mass | 65.459 g mol−1 |
| Appearance | yellow gas |
| Density | 2.872 mg mL−1 |
| Melting point | −59.4 °C (−74.9 °F; 213.8 K) |
| Boiling point | −5.55 °C (22.01 °F; 267.60 K) |
| Reacts | |
| Structure | |
| Dihedral, digonal | |
| Hybridisation | sp2 at N |
| 1.90 D | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
261.68 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
51.71 kJ mol−1 |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | inchem.org |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
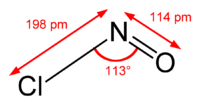
Structure and synthesis edit
The molecule is bent. A double bond exists between N and O (distance = 1.16 Å) and a single bond between N and Cl (distance = 1.96 Å). The O=N–Cl angle is 113°.[2]
Production edit
Nitrosyl chloride can be produced in many ways.
- Combining nitrosylsulfuric acid and HCl affords the compound. This method is used industrially.[3]
- HCl + NOHSO4 → H2SO4 + NOCl
- A more convenient laboratory method involves the (reversible) dehydration of nitrous acid by HCl[4]
- HNO2 + HCl → H2O + NOCl
- By the direct combination of chlorine and nitric oxide; This reaction reverses above 100 °C.
- Cl2 + 2 NO → 2 NOCl
- By reduction of nitrogen dioxide with hydrogen chloride:[5]
- 2NO2 + 4 HCl → 2NOCl + 2H2O + Cl2
Occurrence in aqua regia edit
NOCl also arises from the combination of hydrochloric and nitric acids according to the following reaction:[6]
- HNO3 + 3 HCl → 2[Cl] + 2 H2O + NOCl
In nitric acid, NOCl is readily oxidized into nitrogen dioxide. The presence of NOCl in aqua regia was described by Edmund Davy in 1831.[7]
Reactions edit
NOCl behaves as an electrophile and an oxidant in most of its reactions. With halide acceptors, for example antimony pentachloride, converts to nitrosonium salts:
- NOCl + SbCl5 → [NO]+[SbCl6]−
In a related reaction, sulfuric acid gives nitrosylsulfuric acid, the mixed acid anhydride of nitrous and sulfuric acid:
- ClNO + H2SO4 → ONHSO4 + HCl
NOCl reacts with silver thiocyanate to give silver chloride and the pseudohalogen nitrosyl thiocyanate:
- ClNO + AgSCN → AgCl + ONSCN
Similarly, it reacts with silver cyanide to give nitrosyl cyanide.[8]
Nitrosyl chloride is used to prepare metal nitrosyl complexes. With molybdenum hexacarbonyl, NOCl gives the dinitrosyldichloride complex:[9]
- Mo(CO)6 + 2 NOCl → MoCl2(NO)2 + 6 CO
It dissolves platinum:[10]
- Pt + 6 NOCl → (NO+)2[PtCl6]2- + 4 NO
Applications in organic synthesis edit
Aside from its role in the production of caprolactam, NOCl finds some other uses in organic synthesis. It adds to alkenes to afford α-chloro oximes.[11] The addition of NOCl follows the Markovnikov rule. Ketenes also add NOCl, giving nitrosyl derivatives:
- H2C=C=O + NOCl → ONCH2C(O)Cl
Epoxides react with NOCl to give an α-chloronitritoalkyl derivatives. In the case of propylene oxide, the addition proceeds with high regiochemistry:[12]
It converts amides to N-nitroso derivatives.[13] NOCl converts some cyclic amines to the alkenes. For example, aziridine reacts with NOCl to give ethene, nitrous oxide and hydrogen chloride.
Industrial applications edit
NOCl and cyclohexane react photochemically to give cyclohexanone oxime hydrochloride. This process exploits the tendency of NOCl to undergo photodissociation into NO and Cl radicals. The cyclohexanone oxime is converted to caprolactam, a precursor to nylon-6.[3]
Safety edit
Nitrosyl chloride is very toxic and irritating to the lungs, eyes, and skin.
References edit
- ^ Tilden, William A. (1874). "XXXII.—On aqua regia and the nitrosyl chlorides". J. Chem. Soc. 27: 630–636. doi:10.1039/JS8742700630.
- ^ Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 0-12-352651-5.
- ^ a b Ritz, Josef; Fuchs, Hugo; Kieczka, Heinz; Moran, William C. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a05_031. ISBN 978-3527306732.
- ^ Morton, J. R.; Wilcox, H. W.; Moellerf, Therald; Edwards, Delwin C. (1953). "Nitrosyl Chloride". In Bailar, John C. Jr (ed.). Inorganic Syntheses. Vol. 4. McGraw‐Hill. p. 48. doi:10.1002/9780470132357.ch16. ISBN 9780470132357.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 456. ISBN 978-0-08-037941-8.
- ^ Beckham, L. J.; Fessler, W. A.; Kise, M. A. (1951). "Nitrosyl Chloride". Chemical Reviews. 48 (3): 319–396. doi:10.1021/cr60151a001. PMID 24541207.
- ^ Edmund Davy (1830–1837). "On a New Combination of Chlorine and Nitrous Gas". Abstracts of the Papers Printed in the Philosophical Transactions of the Royal Society of London. 3: 27–29. JSTOR 110250.
- ^ Kirby, G. W. (1977). "Tilden Lecture. Electrophilic C-Nitroso Compounds". Chemical Society Reviews. 6: 1. doi:10.1039/CS9770600001.
- ^ Johnson, B. F. G.; Al-Obadi, K. H. (1970). "Dihalogenodinitrosylmolybdenum and Dihalogenodinitrosyltungsten". Inorganic Syntheses. Vol. 12. pp. 264–266. doi:10.1002/9780470132432.ch47. ISBN 9780470132432.
{{cite book}}:|journal=ignored (help) - ^ Moravek, Richard T. (1986). "Nitrosyl Hexachloroplatinate(IV)". Inorganic Syntheses. Vol. 24. pp. 217–220. doi:10.1002/9780470132555.ch63. ISBN 9780470132555.
- ^ Ohno, M.; Naruse, N.; Terasawa, I. (1969). "7-Cyanoheptanal". Org. Synth. 49: 27. doi:10.15227/orgsyn.049.0027.
- ^ Malinovskii, M. S.; Medyantseva, N. M. (1953). "Olefin Oxides. IX. Condensation of Olefin Oxides with Nitrosyl Chloride". Zhurnal Obshchei Khimii. 23: 84–6. (translated from Russian)
- ^ Van Leusen, A. M.; Strating, J. (1977). "p-Tolylsulfonyldiazomethane". Org. Synth. 57: 95. doi:10.15227/orgsyn.057.0095.
External links edit
- Media related to Nitrosyl chloride at Wikimedia Commons
