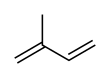
Isoprene, or 2-methyl-1,3-butadiene, is a common volatile organic compound with the formula CH2=C(CH3)−CH=CH2. In its pure form it is a colorless volatile liquid. It is produced by many plants and animals[1] (including humans) and its polymers are the main component of natural rubber. C. G. Williams named the compound in 1860 after obtaining it from the pyrolysis of natural rubber; he correctly deduced the empirical formula C5H8.[2][3]
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Isoprene
| |||
| Preferred IUPAC name
2-Methylbuta-1,3-diene | |||
| Other names
2-Methyl-1,3-butadiene
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.001.040 | ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H8 | |||
| Molar mass | 68.12 g/mol | ||
| Density | 0.681 g/cm3 | ||
| Melting point | −143.95 °C (−227.11 °F; 129.20 K) | ||
| Boiling point | 34.067 °C (93.321 °F; 307.217 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Natural occurrences edit
Isoprene is produced and emitted by many species of trees (major producers are oaks, poplars, eucalyptus, and some legumes). Yearly production of isoprene emissions by vegetation is around 600 million metric tons, half from tropical broadleaf trees and the remainder primarily from shrubs.[4] This is about equivalent to methane emissions and accounts for around one-third of all hydrocarbons released into the atmosphere. In deciduous forests, isoprene makes up approximately 80% of hydrocarbon emissions. While their contribution is small compared to trees, microscopic and macroscopic algae also produce isoprene.[5]
Plants edit
Isoprene is made through the methyl-erythritol 4-phosphate pathway (MEP pathway, also called the non-mevalonate pathway) in the chloroplasts of plants. One of the two end-products of MEP pathway, dimethylallyl pyrophosphate (DMAPP), is cleaved by the enzyme isoprene synthase to form isoprene and diphosphate. Therefore, inhibitors that block the MEP pathway, such as fosmidomycin, also block isoprene formation. Isoprene emission increases dramatically with temperature and maximizes at around 40 °C. This has led to the hypothesis that isoprene may protect plants against heat stress (thermotolerance hypothesis, see below). Emission of isoprene is also observed in some bacteria and this is thought to come from non-enzymatic degradations from DMAPP. Global emission of isoprene by plants is estimated at 350 million tons per year.[6]
Regulation edit
Isoprene emission in plants is controlled both by the availability of the substrate (DMAPP) and by enzyme (isoprene synthase) activity. In particular, light, CO2 and O2 dependencies of isoprene emission are controlled by substrate availability, whereas temperature dependency of isoprene emission is regulated both by substrate level and enzyme activity.
Other organisms edit
Isoprene is the most abundant hydrocarbon measurable in the breath of humans.[7][8][9] The estimated production rate of isoprene in the human body is 0.15 µmol/(kg·h), equivalent to approximately 17 mg/day for a person weighing 70 kg. Isoprene is common in low concentrations in many foods. Many species of soil and marine bacteria, such as Actinomycetota, are capable of degrading isoprene and using it as a fuel source.
Biological roles edit
Isoprene emission appears to be a mechanism that trees use to combat abiotic stresses.[10] In particular, isoprene has been shown to protect against moderate heat stress (around 40 °C). It may also protect plants against large fluctuations in leaf temperature. Isoprene is incorporated into and helps stabilize cell membranes in response to heat stress.
Isoprene also confers resistance to reactive oxygen species.[11] The amount of isoprene released from isoprene-emitting vegetation depends on leaf mass, leaf area, light (particularly photosynthetic photon flux density, or PPFD) and leaf temperature. Thus, during the night, little isoprene is emitted from tree leaves, whereas daytime emissions are expected to be substantial during hot and sunny days, up to 25 μg/(g dry-leaf-weight)/hour in many oak species.[12]
Isoprenoids edit
The isoprene skeleton can be found in naturally occurring compounds called terpenes (also known as isoprenoids), but these compounds do not arise from isoprene itself. Instead, the precursor to isoprene units in biological systems is dimethylallyl pyrophosphate (DMAPP) and its isomer isopentenyl pyrophosphate (IPP). The plural 'isoprenes' is sometimes used to refer to terpenes in general.
Examples of isoprenoids include carotene, phytol, retinol (vitamin A), tocopherol (vitamin E), dolichols, and squalene. Heme A has an isoprenoid tail, and lanosterol, the sterol precursor in animals, is derived from squalene and hence from isoprene. The functional isoprene units in biological systems are dimethylallyl pyrophosphate (DMAPP) and its isomer isopentenyl pyrophosphate (IPP), which are used in the biosynthesis of naturally occurring isoprenoids such as carotenoids, quinones, lanosterol derivatives (e.g. steroids) and the prenyl chains of certain compounds (e.g. phytol chain of chlorophyll). Isoprenes are used in the cell membrane monolayer of many Archaea, filling the space between the diglycerol tetraether head groups. This is thought to add structural resistance to harsh environments in which many Archaea are found.
Similarly, natural rubber is composed of linear polyisoprene chains of very high molecular weight and other natural molecules.[13]
Impact on aerosols edit
After release, isoprene is converted into various species, such as aldehydes, hydroperoxides, organic nitrates, and epoxides[14] by short-lived free radicals (like the hydroxyl radical) and to a lesser extent by ozone.[15] These new species can dissolve into water droplets and contribute to aerosol and haze formation.[16] Secondary organic aerosols formed from this pathway are a current topic of research and may have atmospheric impacts.[17]
While most experts acknowledge that isoprene emission affects aerosol formation, whether isoprene increases or decreases aerosol formation is debated. A second major effect of isoprene on the atmosphere is that in the presence of nitric oxides (NOx) it contributes to the formation of tropospheric (lower atmosphere) ozone, which is one of the leading air pollutants in many countries. Isoprene itself is not normally regarded as a pollutant, as it is a natural plant product. Formation of tropospheric ozone is only possible in presence of high levels of NOx, which comes almost exclusively from industrial activities. Isoprene can have the opposite effect and quench ozone formation under low levels of NOx.
As an example the Blue Ridge Mountains are noted for having a bluish color when seen from a distance. Trees put the "blue" in Blue Ridge, from the isoprene released into the atmosphere.[18] This contributes to the characteristic haze on the mountains and their distinctive color.[19] The similarly named, but geologically district, Blue Mountains of New South Wales also derive their color and name from isoprene and isoprenoids released by Eucalyptus trees.[20]
Industrial production edit
Isoprene is most readily available industrially as a byproduct of the thermal cracking of petroleum naphtha or oil, as a side product in the production of ethylene. About 800,000 metric tons are produced annually. About 95% of isoprene production is used to produce cis-1,4-polyisoprene—a synthetic version of natural rubber.[13]
Natural rubber consists mainly of poly-cis-isoprene with a molecular mass of 100,000 to 1,000,000 g/mol. Typically natural rubber contains a few percent of other materials, such as proteins, fatty acids, resins, and inorganic materials. Some natural rubber sources, called gutta percha, are composed of trans-1,4-polyisoprene, a structural isomer that has similar, but not identical, properties.[13]
See also edit
References edit
- ^ Sharkey TD (1996). "Isoprene synthesis by plants and animals". Endeavour. 20 (2): 74–8. doi:10.1016/0160-9327(96)10014-4. PMID 8690002.
- ^ Williams CG (1860). "On isoprene and caoutchine". Proceedings of the Royal Society of London. 10: 516–519. doi:10.1098/rspl.1859.0101. S2CID 104233421.
- ^ Loadman MJ (2012-12-06). Analysis of Rubber and Rubber-like Polymers. Springer. p. 10. ISBN 9789401144353.
- ^ Guenther A, Karl T, Harley P, Wiedinmyer C, Palmer PI, Geron C (2006). "Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature)". Atmospheric Chemistry and Physics. 6 (11): 3181–3210. Bibcode:2006ACP.....6.3181G. doi:10.5194/acp-6-3181-2006.
- ^ Johnston A, Crombie AT, El Khawand M, Sims L, Whited GM, McGenity TJ, Colin Murrell J (September 2017). "Identification and characterisation of isoprene-degrading bacteria in an estuarine environment". Environmental Microbiology. 19 (9): 3526–3537. doi:10.1111/1462-2920.13842. PMC 6849523. PMID 28654185.
- ^ "Isoprene emissions version 2021". emissions.aeronomie.be. Retrieved 2022-09-26.
- ^ Gelmont D, Stein RA, Mead JF (April 1981). "Isoprene-the main hydrocarbon in human breath". Biochemical and Biophysical Research Communications. 99 (4): 1456–60. doi:10.1016/0006-291X(81)90782-8. PMID 7259787.
- ^ King J, Koc H, Unterkofler K, Mochalski P, Kupferthaler A, Teschl G, et al. (December 2010). "Physiological modeling of isoprene dynamics in exhaled breath". Journal of Theoretical Biology. 267 (4): 626–37. arXiv:1010.2145. Bibcode:2010JThBi.267..626K. doi:10.1016/j.jtbi.2010.09.028. PMID 20869370. S2CID 10267120.
- ^ Williams J, Stönner C, Wicker J, Krauter N, Derstroff B, Bourtsoukidis E, et al. (May 2016). "Cinema audiences reproducibly vary the chemical composition of air during films, by broadcasting scene specific emissions on breath". Scientific Reports. 6: 25464. Bibcode:2016NatSR...625464W. doi:10.1038/srep25464. PMC 4862009. PMID 27160439.
- ^ Sharkey TD, Wiberley AE, Donohue AR (January 2008). "Isoprene emission from plants: why and how". Annals of Botany. 101 (1): 5–18. doi:10.1093/aob/mcm240. PMC 2701830. PMID 17921528.
- ^ Vickers CE, Possell M, Cojocariu CI, Velikova VB, Laothawornkitkul J, Ryan A, et al. (May 2009). "Isoprene synthesis protects transgenic tobacco plants from oxidative stress". Plant, Cell & Environment. 32 (5): 520–31. doi:10.1111/j.1365-3040.2009.01946.x. PMID 19183288.
- ^ Benjamin MT, Sudol M, Bloch L, Winer AM (1996). "Low-emitting urban forests: A taxonomic methodology for assigning isoprene and monoterpene emission rates". Atmospheric Environment. 30 (9): 1437–1452. Bibcode:1996AtmEn..30.1437B. doi:10.1016/1352-2310(95)00439-4.
- ^ a b c Greve HH (2000). "Rubber, 2. Natural". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a23_225. ISBN 978-3527306732.
- ^ Organic Carbon Compounds Emitted By Trees Affect Air Quality, ScienceDaily, Aug. 7, 2009
- ^ IUPAC Subcommittee on Gas Kinetic Data Evaluation – Data Sheet Ox_VOC7, 2007
- ^ A source of haze, ScienceNews, August 6, 2009
- ^ D'Ambro, Emma L.; Schobesberger, Siegfried; Gaston, Cassandra J.; Lopez-Hilfiker, Felipe D.; Lee, Ben H.; Liu, Jiumeng; Zelenyuk, Alla; Bell, David; Cappa, Christopher D.; Helgestad, Taylor; Li, Ziyue (2019-09-05). "Chamber-based insights into the factors controlling epoxydiol (IEPOX) secondary organic aerosol (SOA) yield, composition, and volatility". Atmospheric Chemistry and Physics. 19 (17): 11253–11265. doi:10.5194/acp-19-11253-2019. hdl:10138/305801. ISSN 1680-7316.
- ^ Johnson AW (1998). Invitation To Organic Chemistry. Jones & Bartlett Learning. p. 261. ISBN 978-0-7637-0432-2.
blue mountains chemical terpene.
- ^ "Blue Ridge Parkway, Frequently Asked Questions". National Park Service. 2007. Archived from the original on December 28, 2007. Retrieved December 29, 2007.
- ^ CSIRO. "Beating the eucalypt blues – new ways to model air quality". www.csiro.au. Retrieved 2023-12-10.
Further reading edit
- Budavari S, O'Neil MJ, Smith A, Heckelaman PE, eds. (1989). The Merck Index (11th ed.). Rahway NJ. USA: Merck & Co Inc. ISBN 978-0-911910-28-5.
- Bekkedahl N, Wood LA, Wojciechowski M (1936). "Some physical properties of isoprene". Journal of Research of the National Bureau of Standards. 17 (6): 883. doi:10.6028/jres.017.052.
- Poisson N, Kanakidou M, Crutzen PJ (2000). "Impact of Non-Methane Hydrocarbons on Tropospheric Chemistry and the Oxidizing Power of the Global Troposphere: 3-Dimensional Modelling Results". Journal of Atmospheric Chemistry. 36 (2): 157–230. Bibcode:2000JAtC...36..157P. doi:10.1023/A:1006300616544. S2CID 94217044.
- Claeys M, Graham B, Vas G, Wang W, Vermeylen R, Pashynska V, et al. (February 2004). "Formation of secondary organic aerosols through photooxidation of isoprene". Science. 303 (5661): 1173–6. Bibcode:2004Sci...303.1173C. doi:10.1126/science.1092805. PMID 14976309. S2CID 19268599.
- Pier PA, McDuffie C (1997). "Seasonal isoprene emission rates and model comparisons using whole-tree emissions from white oak". Journal of Geophysical Research: Atmospheres. 102 (D20): 23963–23971. Bibcode:1997JGR...10223963P. doi:10.1029/96JD03786.
- Pöschl U, Von Kuhlmann R, Poisson N, Crutzen PJ (2000). "Development and Intercomparison of Condensed Isoprene Oxidation Mechanisms for Global Atmospheric Modeling". Journal of Atmospheric Chemistry. 37 (1): 29–52. Bibcode:2000JAtC...37...29P. doi:10.1023/A:1006391009798. S2CID 93419825.
- Monson RK, Holland EA (2001). "Biospheric Trace Gas Fluxes and Their Control over Tropospheric Chemistry". Annual Review of Ecology and Systematics. 32: 547–576. doi:10.1146/annurev.ecolsys.32.081501.114136.