A Barr body (named after discoverer Murray Barr)[1] or X-chromatin is an inactive X chromosome. In species with XY sex-determination (including humans), females typically have two X chromosomes,[2] and one is rendered inactive in a process called lyonization. Errors in chromosome separation can also result in male and female individuals with extra X chromosomes. The Lyon hypothesis states that in cells with multiple X chromosomes, all but one are inactivated early in embryonic development in mammals.[3][4] The X chromosomes that become inactivated are chosen randomly, except in marsupials and in some extra-embryonic tissues of some placental mammals, in which the X chromosome from the sperm is always deactivated.[5]

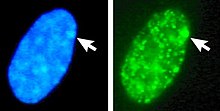
In humans with euploidy, a genotypical female (46, XX karyotype) has one Barr body per somatic cell nucleus, while a genotypical male (46, XY) has none. The Barr body can be seen in the interphase nucleus as a darkly staining small mass in contact with the nucleus membrane. Barr bodies can be seen in neutrophils at the rim of the nucleus.
In humans with more than one X chromosome, the number of Barr bodies visible at interphase is always one fewer than the total number of X chromosomes. For example, people with Klinefelter syndrome (47, XXY) have a single Barr body, and people with a 47, XXX karyotype have two Barr bodies.
Mechanism edit
Someone with two X chromosomes (such as most human females) has only one Barr body per somatic cell, while someone with one X chromosome (such as most human males) has none.
Mammalian X-chromosome inactivation is initiated from the X inactivation centre or Xic, usually found near the centromere.[6] The center contains twelve genes, seven of which code for proteins, five for untranslated RNAs, of which only two are known to play an active role in the X inactivation process, Xist and Tsix.[6] The centre also appears to be important in chromosome counting: ensuring that random inactivation only takes place when two or more X-chromosomes are present. The provision of an extra artificial Xic in early embryogenesis can induce inactivation of the single X found in male cells.[6]
The roles of Xist and Tsix appear to be antagonistic. The loss of Tsix expression on the future inactive X chromosome results in an increase in levels of Xist around the Xic. Meanwhile, on the future active X Tsix levels are maintained; thus the levels of Xist remain low.[7] This shift allows Xist to begin coating the future inactive chromosome, spreading out from the Xic.[2] In non-random inactivation this choice appears to be fixed and current evidence suggests that the maternally inherited gene may be imprinted.[3] Variations in Xi frequency have been reported with age, pregnancy, the use of oral contraceptives, fluctuations in menstrual cycle and neoplasia.[8]
It is thought that this constitutes the mechanism of choice, and allows downstream processes to establish the compact state of the Barr body. These changes include histone modifications, such as histone H3 methylation (i.e. H3K27me3 by PRC2 which is recruited by Xist)[9] and histone H2A ubiquitination,[10] as well as direct modification of the DNA itself, via the methylation of CpG sites.[11] These changes help inactivate gene expression on the inactive X-chromosome and to bring about its compaction to form the Barr body.
Reactivation of a Barr body is also possible, and has been seen in breast cancer patients.[12] One study showed that the frequency of Barr bodies in breast carcinoma were significantly lower than in healthy controls, indicating reactivation of these once inactivated X chromosomes.[12]
See also edit
- X-inactivation
- Sex-determination system
- Nuclear sexing, a genetic sex determination technique
- Demethylation
- Acetylation
- Xist
- Tsix (gene)
References edit
Links to full text articles are provided where access is free, in other cases only the abstract has been linked.
- ^ Barr, M. L.; Bertram, E. G. (1949). "A Morphological Distinction between Neurones of the Male and Female, and the Behaviour of the Nucleolar Satellite during Accelerated Nucleoprotein Synthesis". Nature. 163 (4148): 676–677. Bibcode:1949Natur.163..676B. doi:10.1038/163676a0. PMID 18120749. S2CID 4093883.
- ^ a b Lyon, M. F. (2003). "The Lyon and the LINE hypothesis". Seminars in Cell & Developmental Biology. 14 (6): 313–318. doi:10.1016/j.semcdb.2003.09.015. PMID 15015738.
- ^ a b Brown, C.J., Robinson, W.P., (1997), XIST Expression and X-Chromosome Inactivation in Human Preimplantation Embryos Am. J. Hum. Genet. 61, 5–8 (Full Text PDF)
- ^ Lyon, M. F. (1961). "Gene Action in the X-chromosome of the Mouse (Mus musculus L.)". Nature. 190 (4773): 372–373. Bibcode:1961Natur.190..372L. doi:10.1038/190372a0. PMID 13764598. S2CID 4146768.
- ^ Lee, J. T. (2003). "X-chromosome inactivation: a multi-disciplinary approach". Seminars in Cell & Developmental Biology. 14 (6): 311–312. doi:10.1016/j.semcdb.2003.09.025. PMID 15015737.
- ^ a b c Rougeulle, C.; Avner, P. (2003). "Controlling X-inactivation in mammals: what does the centre hold?". Seminars in Cell & Developmental Biology. 14 (6): 331–340. doi:10.1016/j.semcdb.2003.09.014. PMID 15015740.
- ^ Lee, J. T.; Davidow, L. S.; Warshawsky, D. (1999). "Tisx, a gene antisense to Xist at the X-inactivation centre". Nat. Genet. 21 (4): 400–404. doi:10.1038/7734. PMID 10192391. S2CID 30636065.
- ^ Sharma, Deepti (January 10, 2018). "Deciphering the Role of the Barr Body in Malignancy". Sultan Qaboos University Medical Journal. 17 (4): 389–397. doi:10.18295/squmj.2017.17.04.003. PMC 5766293. PMID 29372079.
- ^ Heard, E.; Rougeulle, C.; Arnaud, D.; Avner, P.; Allis, C. D. (2001). "Methylation of Histone H3 at Lys-9 Is an Early Mark on the X Chromosome during X Inactivation". Cell. 107 (6): 727–738. doi:10.1016/S0092-8674(01)00598-0. PMID 11747809. S2CID 10124177.
- ^ de Napoles, M.; Mermoud, J.E.; Wakao, R.; Tang, Y.A.; Endoh, M.; Appanah, R.; Nesterova, T.B.; Silva, J.; Otte, A.P.; Vidal, M.; Koseki, H.; Brockdorff, N. (2004). "Polycomb Group Proteins Ring1A/B Link Ubiquitylation of Histone H2A to Heritable Gene Silencing and X Inactivation". Dev. Cell. 7 (5): 663–676. doi:10.1016/j.devcel.2004.10.005. PMID 15525528.
- ^ Chadwick, B.P.; Willard, H.F. (2003). "Barring gene expression after XIST: maintaining faculative heterochromatin on the inactive X.". Seminars in Cell & Developmental Biology. 14 (6): 359–367. doi:10.1016/j.semcdb.2003.09.016. PMID 15015743.
- ^ a b Natekar, Prashant E.; DeSouza, Fatima M. (2008). "Reactivation of inactive X chromosome in buccal smear of carcinoma of breast". Indian Journal of Human Genetics. 14 (1): 7–8. doi:10.4103/0971-6866.42320. ISSN 0971-6866. PMC 2840782. PMID 20300284.
Further reading edit
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. (2002). Molecular Biology of the Cell, Fourth Edition. Garland Science. pp. 428–429. ISBN 978-0-8153-4072-0. (Web Edition, Free access)
- Turnpenny & Ellard: Emery's Elements of Medical Genetics 13E (http://www.studentconsult.com/content/default.cfm?ISBN=9780702029172&ID=HC006029 Archived 2020-04-13 at the Wayback Machine)