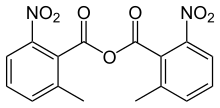
2-Methyl-6-nitrobenzoic anhydride is an organic acid anhydride also known as the Shiina reagent,[1][2] having a structure wherein carboxylic acids undergo intermolecular dehydration condensation. It was developed in 2002 by Prof. Isamu Shiina (Tokyo University of Science, Japan).[3] The compound is often abbreviated MNBA.
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methyl-6-nitrobenzoic anhydride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.156.789 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H12N2O7 | |
| Molar mass | 344.279 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Abstract edit
The reagent is used for synthetic reactions wherein medium- and large-sized lactones are formed from hydroxycarboxylic acids via intramolecular ring closure (Shiina macrolactonization).[4][5] The reaction proceeds at room temperature under basic or neutral conditions. This reagent can be used not only for macrolactonization but also for esterification, amidation, and peptide coupling.
See also edit
References edit
- ^ "Named Reagents". OChemOnline. Archived from the original on 2017-09-04.
- ^ Yahata, K.; Ye, N.; Iso, K.; Naini, S. R.; Yamashita, S.; Ai, Y.; Kishi, Y. (2017). "Unified Synthesis of Right Halves of Halichondrins A–C". J. Org. Chem. 82 (17): 8792–8807. doi:10.1021/acs.joc.7b01283. PMID 28741352.
- ^ Shiina, I.; Ibuka, R.; Kubota, M. (2002). "A New Condensation Reaction for the Synthesis of Carboxylic Esters from Nearly Equimolar Amounts of Carboxylic Acids and Alcohols Using 2-Methyl-6-nitrobenzoic Anhydride". Chem. Lett. 31 (3): 286. doi:10.1246/cl.2002.286.
- ^ Shiina, I.; Kubota, M.; Oshiumi, H.; Hashizume, M. (2004). "An Effective Use of Benzoic Anhydride and Its Derivatives for the Synthesis of Carboxylic Esters and Lactones: A Powerful and Convenient Mixed Anhydride Method Promoted by Basic Catalysts". J. Org. Chem. 69 (6): 1822–30. doi:10.1021/jo030367x. PMID 15058924.
- ^ Shiina, I. (2014). "An Adventurous Synthetic Journey with MNBA from Its Reaction Chemistry to the Total Synthesis of Natural Products". Bull. Chem. Soc. Jpn. 87 (2): 196–233. doi:10.1246/bcsj.20130216.
External links edit
- 2-Methyl-6-nitrobenzoic Anhydride (MNBA) doi:10.1002/047084289X.rn00956
- Enantioselective Total Synthesis of Octalactin A Using Asymmetric Aldol Reactions and a Rapid Lactonization To Form a Medium-Sized Ring doi:10.1002/chem.200500417
- Total Synthesis of Iejimalide B. An Application of the Shiina Macrolactonization doi:10.1021/ol702129w