Pralmorelin (INN) (brand name GHRP Kaken 100; former developmental code names KP-102, GPA-748, WAY-GPA-748), also known as pralmorelin hydrochloride (JAN) and pralmorelin dihydrochloride (USAN), as well as, notably, growth hormone-releasing peptide 2 (GHRP-2), is a growth hormone secretagogue (GHS) used as a diagnostic agent that is marketed by Kaken Pharmaceutical in Japan in a single-dose formulation for the assessment of growth hormone deficiency (GHD).[1][2][3]
 | |
| Clinical data | |
|---|---|
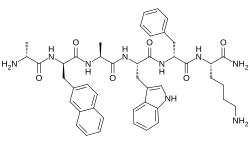
| Other names | D-Alanyl-3-(naphthalen-2-yl)-D-alanyl-L-alanyl-L-tryptophyl-D-phenylalanyl-L-lysinamide |
| Routes of administration | Oral, intravenous |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C45H55N9O6 |
| Molar mass | 817.992 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Pralmorelin is an orally-active, synthetic peptide drug, specifically, an analogue of met-enkephalin, with the amino acid sequence D-Ala-D-(β-naphthyl)-Ala-Trp-D-Phe-Lys-NH2.[2][4] It acts as a ghrelin/growth hormone secretagogue receptor (GHSR) agonist, and was the first of this class of drugs to be introduced clinically.[2][3] Acute administration of the drug markedly increases the levels of plasma growth hormone (GH)[4][5] and reliably induces sensations of hunger and increases food intake in humans.[6]
Pralmorelin was also under investigation for the treatment of GHD and short stature (pituitary dwarfism), and made it to phase II clinical trials for these indications, but was ultimately never marketed for them.[4] This may be because the ability of pralmorelin to increase plasma GH levels is significantly lower in people with GHD relative to healthy individuals.[4]
See also
editReferences
edit- ^ Graul AI, Prous JR (2006). "The Year's New Drugs: A Historical and Research Perspective on the 41 New Products that Reached their First Markets in 2005". Drug News & Perspectives. 19 (1): 33. ISSN 0214-0934.
- ^ a b c Moulin A, Brunel L, Verdié P, Gavara L, Martinez J, Fehrentz JA (2014). "Ghrelin Receptor Ligands: Design and Synthesis of Pseudopeptides and Peptidomimetics". Current Chemical Biology. 7 (3): 254–270. doi:10.2174/2212796807999131128125920. ISSN 2212-7968.
- ^ a b Jameson JL, De Groot LJ (25 February 2015). Endocrinology: Adult and Pediatric: Expert Consult - Online. Elsevier Health Sciences. pp. 1366–. ISBN 978-0-323-32195-2.
- ^ a b c d Adis Editorial (2004). "Pralmorelin: GHRP 2, GPA 748, growth hormone-releasing peptide 2, KP-102 D, KP-102 LN, KP-102D, KP-102LN". Drugs in R&D. 5 (4): 236–239. doi:10.2165/00126839-200405040-00011. PMID 15230633.
- ^ Furuta S, Shimada O, Doi N, Ukai K, Nakagawa T, Watanabe J, Imaizumi M (2004). "General pharmacology of KP-102 (GHRP-2), a potent growth hormone-releasing peptide". Arzneimittel-Forschung. 54 (12): 868–880. doi:10.1055/s-0031-1297042. PMID 15646371. S2CID 28632234.
- ^ Müller TD, Nogueiras R, Andermann ML, Andrews ZB, Anker SD, Argente J, et al. (June 2015). "Ghrelin". Molecular Metabolism. 4 (6): 437–460. doi:10.1016/j.molmet.2015.03.005. PMC 4443295. PMID 26042199.