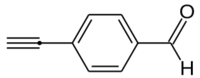
4-Ethynylbenzaldehyde, or p-ethynylbenzaldehyde, is an organic compound with the formula HC2C6H4COH.[2] It is an ethynyl derivative of benzaldehyde, or may also be viewed as a formylated derivative of phenylacetylene.
| |

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
4-Ethynylbenzaldehyde | |
| Other names
p-Ethynylbenzaldehyde
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.155.717 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H6O | |
| Molar mass | 130.146 g·mol−1 |
| Appearance | white or yellow solid |
| Melting point | 89–93 °C (192–199 °F; 362–366 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Preparation
edit4-Ethynylbenzaldehyde may be prepared by the Sonogashira coupling of 4-bromobenzaldehyde with trimethylsilylacetylene to form 4-((trimethylsilyl)ethynyl)benzaldehyde, followed by removal of the trimethylsilyl group with base to form 4-ethynylbenzaldehyde.[3]
Reactions
editThe ethynyl functionality of 4-ethynylbenzaldehyde may undergo a Sonogashira coupling with another molecule of 4-bromobenzaldehyde to form the symmetrical dialdehyde 4,4'-(ethyne-1,2-diyl)dibenzaldehyde.[3]
References
edit- ^ "4-Ethynylbenzaldehyde". ChemSpider. Retrieved 21 October 2023.
- ^ PubChem. "4-Ethynylbenzaldehyde". pubchem.ncbi.nlm.nih.gov. Retrieved 2023-10-22.
- ^ a b Xu, X.; Cai, P.; Chen, H.; Zhou, H.-C.; Huang, N. (28 September 2022). "Three-Dimensional Covalent Organic Frameworks with she Topology". Journal of the American Chemical Society. 144 (40): 18511–18517. doi:10.1021/jacs.2c07733. PMID 36170014. S2CID 252566430.