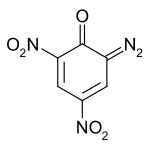
Diazodinitrophenol (DDNP) was the first diazo compound produced; it was subsequently used to make dyes and explosives. It forms yellow crystals in pure form; however, the color of impure forms may vary from dark yellow to green to dark brown.[1] It is soluble in acetic acid, acetone, concentrated hydrochloric acid, like most non-polar solvents and is slightly soluble in water.[2]
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
6-Diazo-2,4-dinitrocyclohexa-2,4-dien-1-one | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| Abbreviations | DDNP | ||
| ChemSpider | |||
| ECHA InfoCard | 100.022.849 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H2N4O5 | |||
| Molar mass | 210.105 g·mol−1 | ||
| Insoluble | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
A solution of cold sodium hydroxide may be used to destroy it.[3] DDNP may be desensitized by immersing it in water, as it does not react in water at normal temperature. It is less sensitive to impact but more powerful than mercury fulminate and almost as powerful as lead azide.[4] The sensitivity of DDNP to friction is much less than that of mercury fulminate and lead azide.
DDNP is used with other materials to form priming mixtures, particularly where a high sensitivity to flame or heat is desired. DDNP is often used as an initiating explosive in propellant primer devices and is a substitute for lead styphnate in what are termed "green" or "non-toxic" (lead free) priming explosive compositions. Lead free primers have been judged as inadequate for service use in firearms due to weak and uneven initiation compared to lead based primers.[5]
History
editDiazodinitrophenol was first prepared in 1858 by the German chemist Peter Griess.[6] It was among the first diazo compounds and for a long time thereafter it was used as a starting material for dyes.[7] Although Griess had mentioned in 1859 that diazodinitrophenol exploded upon heating,[8] it was not until 1892 that diazodinitrophenol was first used as an explosive – when Wilhelm Will[9] and Friedrich Lenze, German chemists at the Militär-Versuchsamt (military research office) in Spandau, Germany, began to investigate azides as potential initiators of explosives.[10] The research was conducted secretly. After a fatal accident, the work was discontinued.[11]
References
edit- ^ Robert Matyáš and Jiří Pachman, Primary Explosives (Berlin, Germany: Springer Verlag, 2013), Chapter 6: Diazodinitrophenol, pp. 157-166; see p. 159.
- ^ Tadeusz Urbański with Marian Jurecki and Sylvia Laverton, trans., Chemistry and Technology of Explosives (Oxford, England: Permagon Press, 1967), vol. 3, pp. 201-204; see especially p. 202.
- ^ (Urbański, 1967), p. 202.
- ^ (Urbański, 1967), p. 204.
- ^ Courtney, Elya; Courtney, Amy; Peter David Summer; Courtney, Michael (2014). "Performance testing of lead free primers: Blast waves, velocity variations, and environmental testing". arXiv:1410.6390 [physics.pop-ph].
- ^ Peter Griess (1858) "Vorläufige Notiz über die Einwirkung von salpetriger Säure auf Amidinitro- und Aminitrophenylsäure," (Preliminary notice of the reaction of nitrous acid with picramic acid and aminonitrophenol), Annalen der Chemie und Pharmacie, 106 : 123-125.
- ^ L. V. Clark (1933) "Diazodinitrophenol, a detonating explosive," Industrial & Engineering Chemistry, 25 (6) : 663-669.
- ^ P. Griess (1859) "On new nitrogenous derivatives of the phenyl- and benzoyl-series," Proceedings of the Royal Society of London, 9 : 594-597; see especially p. 595.
- ^ For biographical information about Wilhelm Will, see Wilhelm Will in German Wikipedia (in German).
- ^ See:
- (Urbański, 1967), p. 202.
- H. Kast, Spreng- und Zündstoffe [Explosives and Detonators] (Braunschweig, Germany: Friedrich Vieweg & Sohn, 1921), pp. 433, 426.
- ^ See:
- Guy B. Taylor and W. C. Cope (1917) "Initial priming substances for high explosives," Technical Paper 162, Bureau of Mines, U. S. Dept. of the Interior, 32 pages; see p. 7.
- Will, W. (1914) "Der Fortschritt in der Auslösung der explosiblen Systeme und sein Einfluss auf die Sprengstoffindustrie" (Progress in the triggering of explosive systems and its influence on the explosives industry), Zeitschrift für das gesamte Schieß- und Sprengstoffwesen, 9 : 52-53.