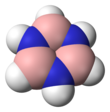
Borazine, also known as borazole, is an inorganic compound with the chemical formula B3H6N3. In this cyclic compound, the three BH units and three NH units alternate. The compound is isoelectronic and isostructural with benzene. For this reason borazine is sometimes referred to as “inorganic benzene”. Like benzene, borazine is a colourless liquid[2] with an aromatic odor.

| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,3,5,2,4,6-Triazatriborinane (only preselected[1]) | |||
| Other names
Cyclotriborazaneborazol
Inorganic benzene Borazole | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.169.303 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| B3H6N3 | |||
| Molar mass | 80.50 g/mol | ||
| Appearance | Colorless liquid | ||
| Density | 0.81 g/cm3 | ||
| Melting point | −58 °C (−72 °F; 215 K) | ||
| Boiling point | 53 °C (127 °F; 326 K) (55 °C at 105 Pa) | ||
| -49.6·10−6 cm3/mol | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Synthesis
editThe compound was reported in 1926 by the chemists Alfred Stock and Erich Pohland by a reaction of diborane with ammonia.[3]
Borazine can be synthesized by treating diborane and ammonia in a 1:2 ratio at 250–300 °C with a conversion of 50%.
- 3 B2H6 + 6 NH3 → 2 B3H6N3 + 12 H2
An alternative more efficient route begins with sodium borohydride and ammonium sulfate:[4]
- 6 NaBH4 + 3 (NH4)2SO4 → 2 B3N3H6 + 3 Na2SO4 + 18 H2
In a two-step process to borazine, boron trichloride is first converted to trichloroborazine:
- 3 BCl3 + 3 NH4Cl → Cl3B3H3N3 + 9 HCl
The B-Cl bonds are subsequently converted to B-H bonds:
- 2 Cl3B3H3N3 + 6 NaBH4 → 2 B3H6N3 + 3 B2H6 + 6 NaCl
Structure
editBorazine is isoelectronic with benzene and has similar connectivity, so it is sometimes referred to as "inorganic benzene". This comparison is not rigorously valid due to the electronegativity difference between boron and nitrogen. X-ray crystallographic structural determinations show that the bond lengths within the borazine ring are all equivalent at 1.429 Å, a property shared by benzene.[5] However, the borazine ring does not form a perfect hexagon. The bond angle is 117.1° at the boron atoms and 122.9° at the nitrogens, giving the molecule the D3h symmetry point group.
The electronegativity of boron (2.04 on the Pauling scale) compared to that of nitrogen (3.04) and also the electron deficiency on the boron atom and the lone pair on nitrogen favor alternative mesomer structures for borazine.
Boron behaves as a Lewis acid and nitrogen behaves as a Lewis base.
Aromaticity
editDue to its similarities to benzene, there have been a number of computational and experimental analyses of borazine's aromaticity. The number of pi electrons in borazine obeys the 4n + 2 rule, and the B-N bond lengths are equal, which suggests the compound may be aromatic. The electronegativity difference between boron and nitrogen, however, creates an unequal sharing of charge which results in bonds with greater ionic character, and thus it is expected to have poorer delocalization of electrons than the all-carbon analog. Borazine, with a standard enthalpy change of formation ΔfH of −531 kJ/mol, is thermally very stable.
Natural bond orbitals (NBO)
editNatural bond orbital (NBO) analysis suggests weak aromaticity in borazine.[6] In the NBO model, B-N bonds in the ring are slightly displaced from the nuclear axes, and B and N have large differences in charge. Natural chemical shielding (NCS) analysis provides some further evidence for aromaticity based on a contribution of the B-N π bond to magnetic shielding. Computations based on NBO orbitals show that this π bond allows for weak ring current which somewhat counteracts a magnetic field simulated at the center of the borazine ring. A small ring current does suggest some delocalization.
Electron localization function (ELF)
editTopological analysis of bonding in borazine by the electron localization function (ELF) indicates that borazine can be described as a π aromatic compound. However, the bonding in borazine is less delocalized than in benzene based on a difference in bifurcation values of the electron basins. Larger bifurcation values indicate better electron delocalization, and it is argued that when this bifurcation value is greater than 0.70, the delocalization is sufficient to designate a compound aromatic.[7] For benzene, this value is 0.91, but the borazine π system bifurcates at the ELF value 0.682.[8] This is caused by the difference in electronegativity between B and N, which produces a weaker bond interaction than the C-C interaction in benzene, leading to increased localization of electrons on the B-H and N-H units. The bifurcation value is slightly below the limit of 0.70 which suggests moderate aromaticity.
Reactivity
editHydrolysis
editBorazine hydrolyzes readily, yielding boric acid, ammonia, and hydrogen.
Polymerization
editHeating borazine at 70 °C expels hydrogen with formation of polyborazylene:
- n B3N3H6 → 1/n[B3N3H4]n
With hydrogen halides and halogens
editWith hydrogen chloride it forms an adduct.
- B3N3H6 + 3 HCl → B3N3H9Cl3
- Addition reaction of borazine with hydrogen chloride
- B3N3H9Cl3 + NaBH4 → (BH4N)3
- Reduction with sodium borohydride
The addition reaction with bromine does not require a catalyst. Borazines undergo nucleophilic attack at boron and electrophilic attack at nitrogen.
Ceramic precursor
editBoron nitride can be prepared by heating polyborazylene to 1000 °C.[4]
Borazines are also starting materials for other potential ceramics such as boron carbonitrides. Borazine can also be used as a precursor to grow hexagonal boron nitride (h-BN) thin films and single layers on catalytic surfaces such as copper,[9] platinum,[10] nickel[11] iron[12] and many more, with chemical vapor deposition (CVD).
Polyborazylene has been proposed as a recycled hydrogen storage medium for hydrogen fuel cell vehicle applications, using a "single pot" process for digestion and reduction to recreate ammonia borane.[13]
Among other B-N type compounds mixed amino-nitro substituted borazines have been predicted to outperform carbon based explosives such as CL-20.[14][15]
Related compounds
edit(C
2H
2B
2N
2) is a six-membered aromatic ring with two carbon atoms, two nitrogen atoms, and two boron atoms in opposing pairs.[16][17]
1,2-Dihydro-1,2-azaborine (C
4BNH
6) is a six-membered ring with four carbon atoms, one nitrogen atom, and one boron atom.
See also
editFurther reading
edit- Nelson, Ralph; et al. (September 1, 1967). "Selected values of electric dipole moments for molecules in the gas phase" (PDF). American National Bureau of Standards (10): 17.
References
edit- ^ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 968. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ Duward Shriver; Peter Atkins (2010). Inorganic Chemistry (Fifth ed.). New York: W. H. Freeman and Company. p. 328. ISBN 978-1429218207.
- ^ Stock A, Pohland E (October 1926). "Borwasserstoffe, VIII. Zur Kenntnis des B2H6 und des B5H11" [Boric acid solution, VIII Regarding knowledge of B2H6 and B5H11]. Berichte (in German). 59 (9): 2210–2215. doi:10.1002/cber.19260590906.
- ^ a b Wideman, Thomas; Fazen, Paul J.; Lynch, Anne T.; Su, Kai; Remsen, Edward E.; Sneddon, Larry G. (1998). "Borazine, Polyborazylene, β-Vinylborazine, and Poly(β-Vinylborazine)". Borazine, Polyborazylene, β‐Vinylborazine, and Poly(β‐Vinylborazine). Inorganic Syntheses. Vol. 32. pp. 232–242. doi:10.1002/9780470132630.ch39. ISBN 9780470132630.
- ^ Boese R, Maulitz AH, Stellberg P (1994). "Solid-State Borazine: Does it Deserve to be Entitled "Inorganic Benzene"?". Chemische Berichte. 127 (10): 1887–1889. doi:10.1002/cber.19941271011.
- ^ Shen W, Li M, Li F, Wang S (2007). "Theoretical study of borazine and its derivatives". Inorg. Chim. Acta. 360 (2): 619–624. doi:10.1016/j.ica.2006.08.028.
- ^ Santos JC, Tiznado W, Contreras R, Fuentealba P (January 2004). "Sigma-pi separation of the electron localization function and aromaticity". The Journal of Chemical Physics. 120 (4): 1670–3. Bibcode:2004JChPh.120.1670S. doi:10.1063/1.1635799. hdl:10533/175272. PMID 15268298.
- ^ Islas R, Chamorro E, Robles J, Heine T, Santos JC, Merino G (2007). "Borazine: to be or not to be aromatic". Struct. Chem. 18 (6): 833–839. doi:10.1007/s11224-007-9229-z. S2CID 95098134.
- ^ Kidambi PR, Blume R, Kling J, Wagner JB, Baehtz C, Weatherup RS, et al. (November 2014). "In Situ Observations during Chemical Vapor Deposition of Hexagonal Boron Nitride on Polycrystalline Copper". Chemistry of Materials. 26 (22): 6380–6392. doi:10.1021/cm502603n. PMC 4311958. PMID 25673919.
- ^ Kim G, Jang AR, Jeong HY, Lee Z, Kang DJ, Shin HS (April 2013). "Growth of high-crystalline, single-layer hexagonal boron nitride on recyclable platinum foil". Nano Letters. 13 (4): 1834–9. Bibcode:2013NanoL..13.1834K. doi:10.1021/nl400559s. PMID 23527543.
- ^ Chatterjee S, Luo Z, Acerce M, Yates DM, Johnson AT, Sneddon LG (2011-10-25). "Chemical Vapor Deposition of Boron Nitride Nanosheets on Metallic Substrates via Decaborane/Ammonia Reactions". Chemistry of Materials. 23 (20): 4414–4416. doi:10.1021/cm201955v. ISSN 0897-4756.
- ^ Caneva S, Weatherup RS, Bayer BC, Brennan B, Spencer SJ, Mingard K, et al. (March 2015). "Nucleation control for large, single crystalline domains of monolayer hexagonal boron nitride via Si-doped Fe catalysts". Nano Letters. 15 (3): 1867–75. Bibcode:2015NanoL..15.1867C. doi:10.1021/nl5046632. PMC 4358078. PMID 25664483.
- ^ Davis BL, Dixon DA, Garner EB, Gordon JC, Matus MH, Scott B, Stephens FH (2009). "Efficient regeneration of partially spent ammonia borane fuel". Angewandte Chemie. 48 (37): 6812–6. doi:10.1002/anie.200900680. OSTI 960522. PMID 19514023.
- ^ Koch EC, Klapötke TM (2012). "Boron-Based High Explosives". Propellants, Explosives, Pyrotechnics. 37 (3): 335–344. doi:10.1002/prep.201100157.
- ^ Kervyn S, Fenwick O, Di Stasio F, Shin YS, Wouters J, Accorsi G, et al. (June 2013). "Polymorphism, fluorescence, and optoelectronic properties of a borazine derivative". Chemistry: A European Journal. 19 (24): 7771–9. doi:10.1002/chem.201204598. PMID 23616404. S2CID 9774352.
- ^ Srivastava AK, Misra N (2015). "Introducing "carborazine" as a novel heterocyclic aromatic species". New Journal of Chemistry. 39 (4): 2483–2488. doi:10.1039/c4nj02089h. S2CID 94792421.
- ^ Bonifazi D, Fasano F, Lorenzo-Garcia MM, Marinelli D, Oubaha H, Tasseroul J (October 2015). "Boron-nitrogen doped carbon scaffolding: organic chemistry, self-assembly and materials applications of borazine and its derivatives". Chemical Communications. 51 (83): 15222–36. doi:10.1039/C5CC06611E. PMID 26411675.
Further reading
edit- Sneddon LG, Mirabelli MG, Lynch AT, Fazen PJ, Su K, Beck JS (1991). "Polymeric precursors to boron based ceramics" (PDF). Pure Appl. Chem. 63 (3): 407–410. doi:10.1351/pac199163030407. S2CID 53629042.
- Jeon JK, Uchimaru Y, Kim DP (August 2004). "Synthesis of novel amorphous boron carbonitride ceramics from the borazine derivative copolymer via hydroboration". Inorganic Chemistry. 43 (16): 4796–8. doi:10.1021/ic035254a. PMID 15285647.
- Paetzold P (1991). "New perspectives in boron-nitrogen chemistry - I" (PDF). Pure Appl. Chem. 63 (3): 345–350. doi:10.1351/pac199163030345. S2CID 53659373.
- Islas R (2007). "Borazine: to be or not to be aromatic". Structural Chemistry. 18 (6): 833–839. doi:10.1007/s11224-007-9229-z. S2CID 95098134.
External links
editMedia related to borazine at Wikimedia Commons