 | |
| Clinical data | |
|---|---|
| Other names |
|
| AHFS/Drugs.com | Monograph |
| Routes of administration | Injection into a vein |
| Drug class | Nonionic detergent[1] |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| Chemical and physical data | |
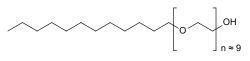
| Formula | C30H62O10 |
| Molar mass | 582.816 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Polidocanol, sold under the brand name Asclera, is a medication used to treat varicose veins including spider veins and reticular veins.[2] It may also be used for hemangiomas, and vascular malformations.[3] It is given by injection into the vein as part of sclerotherapy.[1]
Common side effects include a mild local reaction.[2] Other side effects may include anaphylaxis, blood clots, and tissue necrosis.[2] It is a nonionic detergent.[1]
Polidocanol was approved for medical use in the United States in 2010.[2] In the United States 2 ml of solution costs about 22 USD as of 2021.[4] It is also used topically in cosmetics.[5]
References edit
- ^ a b c "Polidocanol Monograph for Professionals". Drugs.com. Archived from the original on 28 May 2016. Retrieved 28 October 2021.
- ^ a b c d e f "DailyMed - ASCLERA- polidocanol injection, solution". dailymed.nlm.nih.gov. Archived from the original on 24 March 2021. Retrieved 28 October 2021.
- ^ Gao Z, Zhang Y, Li W, Shi C (January 2018). "Effectiveness and safety of polidocanol for the treatment of hemangiomas and vascular malformations: A meta-analysis". Dermatologic Therapy. 31 (1). doi:10.1111/dth.12568. PMID 29082587.
- ^ "Asclera Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 28 October 2021.
- ^ "ADDENDUM to the SCCP opinion on polidocanol (SCCP/1130/07)" (PDF). Archived (PDF) from the original on 14 June 2015. Retrieved 28 October 2021.