 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | de FER a sir ox |
| Trade names | Exjade, Jadenu, others |
| Other names | CGP-72670, ICL-670A, IC L670 |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Iron chelator[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 70% |
| Protein binding | 99% |
| Metabolism | Liver glucuronidation |
| Elimination half-life | 8 to 16 hours |
| Excretion | Fecal (84%) and renal (8%) |
| Identifiers | |
| |
| Chemical and physical data | |
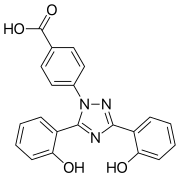
| Formula | C21H15N3O4 |
| Molar mass | 373.368 g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.4±0.1 g/cm3 [2] |
| |
| |
| | |
Deferasirox, sold under the brand name Exjade among others, is a medication used for chronic iron overload in those receiving long-term blood transfusions for conditions such as beta-thalassemia and other chronic anemias.[3] It is generally only when deferoxamine is not sufficient.[3] It is taken by mouth.[4]
Common side effects include kidney problems, nausea, diarrhea, heart burn, rash, itchiness, and liver problems.[3] Other side effects may include metabolic acidosis, bone marrow suppression, and gastrointestinal bleeding.[3][1] It should not be used in people with kidney problems.[1] It is believed to be harmful in pregnancy.[5] It is an iron chelator.[1]
Deferasirox was approved for medical use in the United States in 2005 and Europe in 2006.[6][3] It is on the World Health Organization's List of Essential Medicines.[7] In the United Kingdom a month at a dose of 360 mg per day costs the NHS about £500 as of 2021.[4] In the United States this amount costs about 190 USD.[8] Generic versions were approved in 2020.[9]
References edit
- ^ a b c d "DailyMed - DEFERASIROX- deferasirox tablet, film coated". dailymed.nlm.nih.gov. Archived from the original on 11 January 2022. Retrieved 21 December 2021.
- ^ "Material Safety Data Sheet (MSDS): Deferasirox". ChemSrc. 2018. Archived from the original on 2017-06-12. Retrieved 2021-06-28.
- ^ a b c d e f g "Exjade". Archived from the original on 16 September 2021. Retrieved 21 December 2021.
- ^ a b c BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 1074. ISBN 978-0857114105.
- ^ "Deferasirox Use During Pregnancy". Drugs.com. Archived from the original on 29 November 2020. Retrieved 21 December 2021.
- ^ "Deferasirox Monograph for Professionals". Drugs.com. Archived from the original on 15 August 2020. Retrieved 21 December 2021.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ "Deferasirox Prices and Deferasirox Coupons - GoodRx". GoodRx. Retrieved 21 December 2021.
- ^ "Drugs@FDA: FDA-Approved Drugs". U.S. Food and Drug Administration. Archived from the original on 19 October 2020. Retrieved 15 August 2020.