Portal maintenance status: (May 2019)
|
The Minerals Portal

In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.
The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks.
The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral or may be an aggregate of two or more different types of minerals, spacially segregated into distinct phases.
Some natural solid substances without a definite crystalline structure, such as opal or obsidian, are more properly called mineraloids. If a chemical compound occurs naturally with different crystal structures, each structure is considered a different mineral species. Thus, for example, quartz and stishovite are two different minerals consisting of the same compound, silicon dioxide. (Full article...)
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...)
Selected articles
-
Image 1A ruby crystal from Dodoma Region, Tanzania
Ruby is a pinkish red to blood-red colored gemstone, a variety of the mineral corundum (aluminium oxide). Ruby is one of the most popular traditional jewelry gems and is very durable. Other varieties of gem-quality corundum are called sapphires. Ruby is one of the traditional cardinal gems, alongside amethyst, sapphire, emerald, and diamond. The word ruby comes from ruber, Latin for red. The color of a ruby is due to the element chromium.
Some gemstones that are popularly or historically called rubies, such as the Black Prince's Ruby in the British Imperial State Crown, are actually spinels. These were once known as "Balas rubies".
The quality of a ruby is determined by its color, cut, and clarity, which, along with carat weight, affect its value. The brightest and most valuable shade of red, called blood-red or pigeon blood, commands a large premium over other rubies of similar quality. After color follows clarity: similar to diamonds, a clear stone will command a premium, but a ruby without any needle-like rutile inclusions may indicate that the stone has been treated. Ruby is the traditional birthstone for July and is usually pinker than garnet, although some rhodolite garnets have a similar pinkish hue to most rubies. The world's most valuable ruby to be sold at auction is the Sunrise Ruby. (Full article...) -
Image 2
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the exception of extremely rare native iron deposits, it is the most magnetic of all the naturally occurring minerals on Earth. Naturally magnetized pieces of magnetite, called lodestone, will attract small pieces of iron, which is how ancient peoples first discovered the property of magnetism.
Magnetite is black or brownish-black with a metallic luster, has a Mohs hardness of 5–6 and leaves a black streak. Small grains of magnetite are very common in igneous and metamorphic rocks.
The chemical IUPAC name is iron(II,III) oxide and the common chemical name is ferrous-ferric oxide. (Full article...) -
Image 3

Kaolinite (/ˈkeɪ.ələˌnaɪt, -lɪ-/ KAY-ə-lə-nyte, -lih-; also called kaolin) is a clay mineral, with the chemical composition: Al2Si2O5(OH)4. It is a layered silicate mineral, with one tetrahedral sheet of silica (SiO4) linked through oxygen atoms to one octahedral sheet of alumina (AlO6).
Kaolinite is a soft, earthy, usually white, mineral (dioctahedral phyllosilicate clay), produced by the chemical weathering of aluminium silicate minerals like feldspar. It has a low shrink–swell capacity and a low cation-exchange capacity (1–15 meq/100 g).
Rocks that are rich in kaolinite, and halloysite, are known as kaolin (/ˈkeɪ.əlɪn/) or china clay. In many parts of the world kaolin is colored pink-orange-red by iron oxide, giving it a distinct rust hue. Lower concentrations of iron oxide yield the white, yellow, or light orange colors of kaolin. Alternating lighter and darker layers are sometimes found, as at Providence Canyon State Park in Georgia, United States.
Kaolin is an important raw material in many industries and applications. Commercial grades of kaolin are supplied and transported as powder, lumps, semi-dried noodle or slurry. Global production of kaolin in 2021 was estimated to be 45 million tonnes, with a total market value of $US4.24 billion. (Full article...) -
Image 4
Graphite (/ˈɡræfaɪt/) is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on a large scale (1.3 million metric tons per year in 2022) for uses in pencils, lubricants, and electrodes. Under high pressures and temperatures it converts to diamond. It is a good (but not excellent) conductor of both heat and electricity. (Full article...) -
Image 5

Borax (also referred to as sodium borate, tincal (/ˈtɪŋkəl/) and tincar (/ˈtɪŋkər/)) is a salt (ionic compound), a hydrated or anhydrous borate of sodium, with the chemical formula Na2H20B4O17 (also written as Na2B4O7·10H2O).
It is a colorless crystalline solid that dissolves in water to make a basic solution.
It is commonly available in powder or granular form and has many industrial and household uses, including as a pesticide, as a metal soldering flux, as a component of glass, enamel, and pottery glazes, for tanning of skins and hides, for artificial aging of wood, as a preservative against wood fungus, and as a pharmaceutic alkalizer. In chemical laboratories, it is used as a buffering agent.
The terms tincal and tincar refer to native borax, historically mined from dry lake beds in various parts of Asia. (Full article...) -
Image 6The 423-carat (85 g) blue Logan Sapphire
Sapphire is a precious gemstone, a variety of the mineral corundum, consisting of aluminium oxide (α-Al2O3) with trace amounts of elements such as iron, titanium, cobalt, lead, chromium, vanadium, magnesium, boron, and silicon. The name sapphire is derived from the Latin word sapphirus, itself from the Greek word sappheiros (σάπφειρος), which referred to lapis lazuli. It is typically blue, but natural "fancy" sapphires also occur in yellow, purple, orange, and green colors; "parti sapphires" show two or more colors. Red corundum stones also occur, but are called rubies rather than sapphires. Pink-colored corundum may be classified either as ruby or sapphire depending on locale. Commonly, natural sapphires are cut and polished into gemstones and worn in jewelry. They also may be created synthetically in laboratories for industrial or decorative purposes in large crystal boules. Because of the remarkable hardness of sapphires – 9 on the Mohs scale (the third hardest mineral, after diamond at 10 and moissanite at 9.5) – sapphires are also used in some non-ornamental applications, such as infrared optical components, high-durability windows, wristwatch crystals and movement bearings, and very thin electronic wafers, which are used as the insulating substrates of special-purpose solid-state electronics such as integrated circuits and GaN-based blue LEDs. Sapphire is the birthstone for September and the gem of the 45th anniversary. A sapphire jubilee occurs after 65 years. (Full article...) -
Image 7

Tourmaline (/ˈtʊərməlɪn, -ˌliːn/ TOOR-mə-lin, -leen) is a crystalline silicate mineral group in which boron is compounded with elements such as aluminium, iron, magnesium, sodium, lithium, or potassium. This gemstone comes in a wide variety of colors.
The name is derived from the Sinhalese tōramalli (ටෝරමල්ලි), which refers to the carnelian gemstones. (Full article...) -
Image 8

Turquoise is an opaque, blue-to-green mineral that is a hydrous phosphate of copper and aluminium, with the chemical formula CuAl6(PO4)4(OH)8·4H2O. It is rare and valuable in finer grades and has been prized as a gemstone for millennia due to its hue.
Like most other opaque gems, turquoise has been devalued by the introduction of treatments, imitations, and synthetics into the market. The robin egg blue or sky blue color of the Persian turquoise mined near the modern city of Nishapur, Iran, has been used as a guiding reference for evaluating turquoise quality. (Full article...) -
Image 9
Mineralogy applies principles of chemistry, geology, physics and materials science to the study of minerals
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...) -
Image 10
Opal is a hydrated amorphous form of silica (SiO2·nH2O); its water content may range from 3% to 21% by weight, but is usually between 6% and 10%. Due to its amorphous property, it is classified as a mineraloid, unlike crystalline forms of silica, which are considered minerals. It is deposited at a relatively low temperature and may occur in the fissures of almost any kind of rock, being most commonly found with limonite, sandstone, rhyolite, marl, and basalt.
The name opal is believed to be derived from the Sanskrit word upala (उपल), which means 'jewel', and later the Greek derivative opállios (ὀπάλλιος).
There are two broad classes of opal: precious and common. Precious opal displays play-of-color (iridescence); common opal does not. Play-of-color is defined as "a pseudo chromatic optical effect resulting in flashes of colored light from certain minerals, as they are turned in white light." The internal structure of precious opal causes it to diffract light, resulting in play-of-color. Depending on the conditions in which it formed, opal may be transparent, translucent, or opaque, and the background color may be white, black, or nearly any color of the visual spectrum. Black opal is considered the rarest, while white, gray, and green opals are the most common. (Full article...) -
Image 11Halite from the Wieliczka salt mine, Małopolskie, Poland
Halite (/ˈhælaɪt, ˈheɪlaɪt/ HAL-yte, HAY-lyte), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride (NaCl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, pink, red, orange, yellow or gray depending on inclusion of other materials, impurities, and structural or isotopic abnormalities in the crystals. It commonly occurs with other evaporite deposit minerals such as several of the sulfates, halides, and borates. The name halite is derived from the Ancient Greek word for "salt", ἅλς (háls). (Full article...) -
Image 12
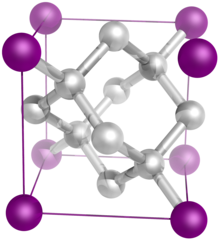
The diamond crystal structure belongs to the face-centered cubic lattice, with a repeated two-atom pattern.
In crystallography, a crystal system is a set of point groups (a group of geometric symmetries with at least one fixed point). A lattice system is a set of Bravais lattices. Space groups are classified into crystal systems according to their point groups, and into lattice systems according to their Bravais lattices. Crystal systems that have space groups assigned to a common lattice system are combined into a crystal family.
The seven crystal systems are triclinic, monoclinic, orthorhombic, tetragonal, trigonal, hexagonal, and cubic. Informally, two crystals are in the same crystal system if they have similar symmetries (though there are many exceptions). (Full article...) -
Image 13

Gypsum is a soft sulfate mineral composed of calcium sulfate dihydrate, with the chemical formula CaSO4·2H2O. It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, drywall and blackboard or sidewalk chalk. Gypsum also crystallizes as translucent crystals of selenite. It forms as an evaporite mineral and as a hydration product of anhydrite. The Mohs scale of mineral hardness defines gypsum as hardness value 2 based on scratch hardness comparison.
Fine-grained white or lightly tinted forms of gypsum known as alabaster have been used for sculpture by many cultures including Ancient Egypt, Mesopotamia, Ancient Rome, the Byzantine Empire, and the Nottingham alabasters of Medieval England. (Full article...) -
Image 14

Crystal structure of table salt (sodium in purple, chlorine in green)
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions, or molecules in a crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns that repeat along the principal directions of three-dimensional space in matter.
The smallest group of particles in the material that constitutes this repeating pattern is the unit cell of the structure. The unit cell completely reflects the symmetry and structure of the entire crystal, which is built up by repetitive translation of the unit cell along its principal axes. The translation vectors define the nodes of the Bravais lattice.
The lengths of the principal axes, or edges, of the unit cell and the angles between them are the lattice constants, also called lattice parameters or cell parameters. The symmetry properties of the crystal are described by the concept of space groups. All possible symmetric arrangements of particles in three-dimensional space may be described by the 230 space groups.
The crystal structure and symmetry play a critical role in determining many physical properties, such as cleavage, electronic band structure, and optical transparency. (Full article...) -
Image 15
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the chemically stable form of carbon at room temperature and pressure, but diamond is metastable and converts to it at a negligible rate under those conditions. Diamond has the highest hardness and thermal conductivity of any natural material, properties that are used in major industrial applications such as cutting and polishing tools. They are also the reason that diamond anvil cells can subject materials to pressures found deep in the Earth.
Because the arrangement of atoms in diamond is extremely rigid, few types of impurity can contaminate it (two exceptions are boron and nitrogen). Small numbers of defects or impurities (about one per million of lattice atoms) can color a diamond blue (boron), yellow (nitrogen), brown (defects), green (radiation exposure), purple, pink, orange, or red. Diamond also has a very high refractive index and a relatively high optical dispersion.
Most natural diamonds have ages between 1 billion and 3.5 billion years. Most were formed at depths between 150 and 250 kilometres (93 and 155 mi) in the Earth's mantle, although a few have come from as deep as 800 kilometres (500 mi). Under high pressure and temperature, carbon-containing fluids dissolved various minerals and replaced them with diamonds. Much more recently (hundreds to tens of million years ago), they were carried to the surface in volcanic eruptions and deposited in igneous rocks known as kimberlites and lamproites.
Synthetic diamonds can be grown from high-purity carbon under high pressures and temperatures or from hydrocarbon gases by chemical vapor deposition (CVD). Imitation diamonds can also be made out of materials such as cubic zirconia and silicon carbide. Natural, synthetic, and imitation diamonds are most commonly distinguished using optical techniques or thermal conductivity measurements. (Full article...) -
Image 16Beachy Head is a part of the extensive Southern England Chalk Formation.
Chalk is a soft, white, porous, sedimentary carbonate rock. It is a form of limestone composed of the mineral calcite and originally formed deep under the sea by the compression of microscopic plankton that had settled to the sea floor. Chalk is common throughout Western Europe, where deposits underlie parts of France, and steep cliffs are often seen where they meet the sea in places such as the Dover cliffs on the Kent coast of the English Channel.
Chalk is mined for use in industry, such as for quicklime, bricks and builder's putty, and in agriculture, for raising pH in soils with high acidity. It is also used for "blackboard chalk" for writing and drawing on various types of surfaces, although these can also be manufactured from other carbonate-based minerals, or gypsum. (Full article...) -
Image 17Quartz crystal cluster from Brazil
Quartz is a hard, crystalline mineral composed of silica (silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon–oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical formula of SiO2. Quartz is, therefore, classified structurally as a framework silicate mineral and compositionally as an oxide mineral. Quartz is the second most abundant mineral in Earth's continental crust, behind feldspar.
Quartz exists in two forms, the normal α-quartz and the high-temperature β-quartz, both of which are chiral. The transformation from α-quartz to β-quartz takes place abruptly at 573 °C (846 K; 1,063 °F). Since the transformation is accompanied by a significant change in volume, it can easily induce microfracturing of ceramics or rocks passing through this temperature threshold.
There are many different varieties of quartz, several of which are classified as gemstones. Since antiquity, varieties of quartz have been the most commonly used minerals in the making of jewelry and hardstone carvings, especially in Europe and Asia.
Quartz is the mineral defining the value of 7 on the Mohs scale of hardness, a qualitative scratch method for determining the hardness of a material to abrasion. (Full article...) -
Image 18A lustrous crystal of zircon perched on a tan matrix of calcite from the Gilgit District of Pakistan
Zircon (/ˈzɜːrkɒn, -kən/) is a mineral belonging to the group of nesosilicates and is a source of the metal zirconium. Its chemical name is zirconium(IV) silicate, and its corresponding chemical formula is ZrSiO4. An empirical formula showing some of the range of substitution in zircon is (Zr1–y, REEy)(SiO4)1–x(OH)4x–y. Zircon precipitates from silicate melts and has relatively high concentrations of high field strength incompatible elements. For example, hafnium is almost always present in quantities ranging from 1 to 4%. The crystal structure of zircon is tetragonal crystal system. The natural color of zircon varies between colorless, yellow-golden, red, brown, blue, and green.
The name derives from the Persian zargun, meaning "gold-hued". This word is changed into "jargoon", a term applied to light-colored zircons. The English word "zircon" is derived from Zirkon, which is the German adaptation of this word. Yellow, orange, and red zircon is also known as "hyacinth", from the flower hyacinthus, whose name is of Ancient Greek origin. (Full article...) -
Image 19
Beryl (/ˈbɛrəl/ BERR-əl) is a mineral composed of beryllium aluminium silicate with the chemical formula Be3Al2Si6O18. Well-known varieties of beryl include emerald and aquamarine. Naturally occurring hexagonal crystals of beryl can be up to several meters in size, but terminated crystals are relatively rare. Pure beryl is colorless, but it is frequently tinted by impurities; possible colors are green, blue, yellow, pink, and red (the rarest). It is an ore source of beryllium. (Full article...) -
Image 20

Cinnabar (/ˈsɪnəˌbɑːr/; from Ancient Greek κιννάβαρι (kinnábari)), or cinnabarite (/ˌsɪnəˈbɑːraɪt/), also known as mercurblende is the bright scarlet to brick-red form of mercury(II) sulfide (HgS). It is the most common source ore for refining elemental mercury and is the historic source for the brilliant red or scarlet pigment termed vermilion and associated red mercury pigments.
Cinnabar generally occurs as a vein-filling mineral associated with volcanic activity and alkaline hot springs. The mineral resembles quartz in symmetry and it exhibits birefringence. Cinnabar has a mean refractive index near 3.2, a hardness between 2.0 and 2.5, and a specific gravity of approximately 8.1. The color and properties derive from a structure that is a hexagonal crystalline lattice belonging to the trigonal crystal system, crystals that sometimes exhibit twinning.
Cinnabar has been used for its color since antiquity in the Near East, including as a rouge-type cosmetic, in the New World since the Olmec culture, and in China since as early as the Yangshao culture, where it was used in coloring stoneware.
Associated modern precautions for the use and handling of cinnabar arise from the toxicity of the mercury component, which was recognized as early as ancient Rome. (Full article...) -
Image 21

Garnets ( /ˈɡɑːrnɪt/) are a group of silicate minerals that have been used since the Bronze Age as gemstones and abrasives.
All species of garnets possess similar physical properties and crystal forms, but differ in chemical composition. The different species are pyrope, almandine, spessartine, grossular (varieties of which are hessonite or cinnamon-stone and tsavorite), uvarovite and andradite. The garnets make up two solid solution series: pyrope-almandine-spessartine (pyralspite), with the composition range [Mg,Fe,Mn]3Al2(SiO4)3; and uvarovite-grossular-andradite (ugrandite), with the composition range Ca3[Cr,Al,Fe]2(SiO4)3. (Full article...) -
Image 22Brazilian trigonal hematite crystal
Hematite (/ˈhiːməˌtaɪt, ˈhɛmə-/), also spelled as haematite, is a common iron oxide compound with the formula, Fe2O3 and is widely found in rocks and soils. Hematite crystals belong to the rhombohedral lattice system which is designated the alpha polymorph of Fe
2O
3. It has the same crystal structure as corundum (Al
2O
3) and ilmenite (FeTiO
3). With this it forms a complete solid solution at temperatures above 950 °C (1,740 °F).
Hematite naturally occurs in black to steel or silver-gray, brown to reddish-brown, or red colors. It is mined as an important ore mineral of iron. It is electrically conductive. Hematite varieties include kidney ore, martite (pseudomorphs after magnetite), iron rose and specularite (specular hematite). While these forms vary, they all have a rust-red streak. Hematite is not only harder than pure iron, but also much more brittle. Maghemite is a polymorph of hematite (γ-Fe
2O
3) with the same chemical formula, but with a spinel structure like magnetite.
Large deposits of hematite are found in banded iron formations. Gray hematite is typically found in places that have still, standing water or mineral hot springs, such as those in Yellowstone National Park in North America. The mineral can precipitate in the water and collect in layers at the bottom of the lake, spring, or other standing water. Hematite can also occur in the absence of water, usually as the result of volcanic activity.
Clay-sized hematite crystals can also occur as a secondary mineral formed by weathering processes in soil, and along with other iron oxides or oxyhydroxides such as goethite, which is responsible for the red color of many tropical, ancient, or otherwise highly weathered soils. (Full article...) -
Image 23

Corundum is a crystalline form of aluminium oxide (Al2O3) typically containing traces of iron, titanium, vanadium, and chromium. It is a rock-forming mineral. It is a naturally transparent material, but can have different colors depending on the presence of transition metal impurities in its crystalline structure. Corundum has two primary gem varieties: ruby and sapphire. Rubies are red due to the presence of chromium, and sapphires exhibit a range of colors depending on what transition metal is present. A rare type of sapphire, padparadscha sapphire, is pink-orange.
The name "corundum" is derived from the Tamil-Dravidian word kurundam (ruby-sapphire) (appearing in Sanskrit as kuruvinda).
Because of corundum's hardness (pure corundum is defined to have 9.0 on the Mohs scale), it can scratch almost all other minerals. It is commonly used as an abrasive on sandpaper and on large tools used in machining metals, plastics, and wood. Emery, a variety of corundum with no value as a gemstone, is commonly used as an abrasive. It is a black granular form of corundum, in which the mineral is intimately mixed with magnetite, hematite, or hercynite.
In addition to its hardness, corundum has a density of 4.02 g/cm3 (251 lb/cu ft), which is unusually high for a transparent mineral composed of the low-atomic mass elements aluminium and oxygen. (Full article...) -
Image 24Malachite from the Democratic Republic of the Congo
Malachite is a copper carbonate hydroxide mineral, with the formula Cu2CO3(OH)2. This opaque, green-banded mineral crystallizes in the monoclinic crystal system, and most often forms botryoidal, fibrous, or stalagmitic masses, in fractures and deep, underground spaces, where the water table and hydrothermal fluids provide the means for chemical precipitation. Individual crystals are rare, but occur as slender to acicular prisms. Pseudomorphs after more tabular or blocky azurite crystals also occur. (Full article...) -
Image 25Deep green isolated fluorite crystal resembling a truncated octahedron, set upon a micaceous matrix, from Erongo Mountain, Erongo Region, Namibia (overall size: 50 mm × 27 mm, crystal size: 19 mm wide, 30 g)
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite.
Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glases, so fluorite is useful in making apochromatic lenses, making it valuable, particularly in photographic optics. Fluorite optics are also usable in the far-ultraviolet and mid-infrared ranges, where conventional glasses are too opaque for use. Fluorite also has low dispersion, and a high refractive index for its density, which can make it useful for some specialized purposes in optics. (Full article...)
Selected mineralogist
-
Image 1Karl Hugo Strunz (24 February 1910 – 19 April 2006) was a German mineralogist. He is best known for creating the Nickel-Strunz classification, the ninth edition of which was published together with Ernest Henry Nickel. (Full article...)
-
Image 2Ralph Early Grim (February 25, 1902 – August 19, 1989) was an American geologist and scientist, often referred to as the "Father of Mineralogy" because he made many discoveries during his investigations of clay materials. He was one of the most outstanding mineralogists of his time and was well-known throughout the world in the field of clay science and technology. Grim's career spanned over 60 years and he received many honors and awards in the field of mineralogy. Some of the textbooks he wrote have been standard university textbooks in mineralogy for many years. (Full article...)
-
Image 3Robert Minard Garrels (August 24, 1916 – March 8, 1988) was an American geochemist. Garrels applied experimental physical chemistry data and techniques to geology and geochemistry problems. The book Solutions, Minerals, and Equilibria co-authored in 1965 by Garrels and Charles L. Christ revolutionized aqueous geochemistry.
Garrels earned a bachelor's degree in geology from the University of Michigan in 1937. He went on to earn an M.S. degree from Northwestern University in 1939, his thesis work was on iron ores of Newfoundland in 1938. His Ph.D. was awarded in 1941 based on lab studies of complex formation between lead and chloride ions in aqueous solution. (Full article...) -
Image 4Bust of Shen at the Beijing Ancient Observatory
Shen Kuo (Chinese: 沈括; 1031–1095) or Shen Gua, courtesy name Cunzhong (存中) and pseudonym Mengqi (now usually given as Mengxi) Weng (夢溪翁), was a Chinese polymath, scientist, and statesman of the Song dynasty (960–1279). Shen was a master in many fields of study including mathematics, optics, and horology. In his career as a civil servant, he became a finance minister, governmental state inspector, head official for the Bureau of Astronomy in the Song court, Assistant Minister of Imperial Hospitality, and also served as an academic chancellor. At court his political allegiance was to the Reformist faction known as the New Policies Group, headed by Chancellor Wang Anshi (1021–1085).
In his Dream Pool Essays or Dream Torrent Essays (夢溪筆談; Mengxi Bitan) of 1088, Shen was the first to describe the magnetic needle compass, which would be used for navigation (first described in Europe by Alexander Neckam in 1187). Shen discovered the concept of true north in terms of magnetic declination towards the north pole, with experimentation of suspended magnetic needles and "the improved meridian determined by Shen's [astronomical] measurement of the distance between the pole star and true north". This was the decisive step in human history to make compasses more useful for navigation, and may have been a concept unknown in Europe for another four hundred years (evidence of German sundials made circa 1450 show markings similar to Chinese geomancers' compasses in regard to declination). (Full article...) -
Image 5

Solvay Conference on Physics in Brussels 1951. Left to right, sitting: Crussaro, N.P. Allen, Cauchois, Borelius, Bragg, Moller, Sietz, Hollomon, Frank; middle row: Rathenau,(nl) Koster, Rudberg,(sv), Flamache, Goche, Groven, Orowan, Burgers, Shockley, Guinier, C.S. Smith, Dehlinger, Laval, Henriot; top row: Gaspart, Lomer, Cottrell, Homes, Curien
Hubert Curien (30 October 1924 – 6 February 2005) was a French physicist and a key figure in European science politics, as the President of CERN Council (1994–1996), the first chairman of the European Space Agency (ESA) (1981–1984), and second President of the Academia Europæa and a President of Fondation de France. (Full article...) -
Image 6Peggy-Kay Hamilton (1922–1959) was born in Illinois in 1922 and was an American Research Associate in Mineralogy in the Department of Geology at Columbia University. One of Hamilton's first research breakthroughs was developing Research Project 49, otherwise known as the study of clay minerals. In her later research years, her focus shifted and led to her becoming involved full time in the study of uranium.
Hamilton achieved success in the fields of geology and mineralogy; according to her frequent research partner and friend Paul F. Kerr, Hamilton was held in high regard by both students at Columbia University as well as professional colleagues at multiple scientific research institutions. (Full article...) -
Image 7

Alexander Evgenyevich Fersman (Russian: Александр Евгеньевич Ферсман; 8 November 1883 – 20 May 1945) was a prominent Soviet Russian geochemist and mineralogist, and a member of the Soviet Academy of Sciences (1919–1945). (Full article...) -
Image 8
Axel Hamberg (17 January 1863 – 28 June 1933) was a Swedish mineralogist, geographer and explorer. (Full article...) -
Image 9

Carl Linnaeus (23 May 1707 – 10 January 1778), also known after ennoblement in 1761 as Carl von Linné, was a Swedish biologist and physician who formalised binomial nomenclature, the modern system of naming organisms. He is known as the "father of modern taxonomy". Many of his writings were in Latin; his name is rendered in Latin as Carolus Linnæus and, after his 1761 ennoblement, as Carolus a Linné.
Linnaeus was the son of a curate and he was born in Råshult, the countryside of Småland, in southern Sweden. He received most of his higher education at Uppsala University and began giving lectures in botany there in 1730. He lived abroad between 1735 and 1738, where he studied and also published the first edition of his Systema Naturae in the Netherlands. He then returned to Sweden where he became professor of medicine and botany at Uppsala. In the 1740s, he was sent on several journeys through Sweden to find and classify plants and animals. In the 1750s and 1760s, he continued to collect and classify animals, plants, and minerals, while publishing several volumes. By the time of his death in 1778, he was one of the most acclaimed scientists in Europe. (Full article...) -
Image 10
Johan Gottlieb Gahn (19 August 1745 – 8 December 1818) was a Swedish chemist and metallurgist who isolated manganese in 1774.
Gahn studied in Uppsala 1762 – 1770 and became acquainted with chemists Torbern Bergman and Carl Wilhelm Scheele. 1770 he settled in Falun, where he introduced improvements in copper smelting, and participated in building up several factories, including those for vitriol, sulfur and red paint. (Full article...) -
Image 11
Nils Gustaf Nordenskiöld (October 12, 1792 – February 2, 1866) was a Finnish mineralogist and traveller. He was the father of Adolf Erik Nordenskiöld, a mineralogist and polar explorer (Full article...) -
Image 12Bertram Eugene Warren (June 28, 1902 – June 27, 1991) was an American crystallographer. His studies of X-rays provided much knowledge and understanding of both crystalline and non-crystalline materials. He also worked on changing amorphous solids to a crystalline state. (Full article...)
-
Image 13

Gustav Adolph Kenngott, c. 1860
Gustav Adolph Kenngott (January 6, 1818 – March 7, 1897) was a German mineralogist. (Full article...) -
Image 14Sjur Aasmundsen Sexe (14 August 1808 – 17 February 1888) was a Norwegian mineralogist and educator. (Full article...)
-
Image 15Friedrich Klockmann (12 April 1858, Schwerin – 17 November 1937, Aachen) was a German geologist and mineralogist.
He studied geology and mineralogy at the Bergakademie in Clausthal and at the University of Rostock, receiving his doctorate in 1881. Following graduation he worked for the Prussian Geological Survey, based in Berlin. From 1887 he taught classes in mineralogy and geology at the academy in Clausthal, and in 1892 attained the title of professor. From 1899 he worked as a professor at the technical university in Aachen, where in 1917/18 he served as academic rector. (Full article...) -
Image 16Alexander Sadebeck (26 June 1843 in Breslau – 9 December 1879 in Hamburg) was a German geologist and mineralogist. He was a brother of botanist Richard Sadebeck (1839–1905).
He studied mineralogy and geology at the University of Berlin as a pupil of Gustav Rose. In 1865 he received his doctorate with a dissertation on Upper Jurassic formations in Pomerania. In 1872 he was appointed professor of mineralogy and geology at the University of Kiel. (Full article...) -
Image 17Leonard James Spencer CBE FRS (7 July 1870 – 14 April 1959) was a British geologist. He was an Honorary member of the Royal Geological Society of Cornwall, and also a recipient of its Bolitho Medal. He was president of the Mineralogical Society of Great Britain and Ireland from 1936 to 1939. In mineralogy, Spencer was an original investigator who described several new minerals, including miersite, tarbuttite and parahopeite. He also did important work as a curator, editor and bibliographer. He was the third person to receive the Roebling Medal, the highest award of the Mineralogical Society of America. In 1937, he was awarded the Murchison Medal of the Geological Society of London. He wrote at least 146 articles for the Encyclopædia Britannica Eleventh Edition.
His daughter, Penelope Spencer became a successful free-style dancer and choreographer. (Full article...) -
Image 18

John Scouler
John Scouler (31 December 1804 – 13 November 1871) was a Scottish naturalist. (Full article...) -
Image 19Warren D. Huff (born April 16, 1937) is Professor of Geology at the University of Cincinnati (UC). He specializes in clay mineralogy, researching the remnants of ancient volcanic eruptions.
He received his BA from Harvard College, 1959 and his PhD from University of Cincinnati, 1963. He has served as assistant professor at UC 1963-1969, associate professor 1969-1985, full professor 1985 to date. He teaches introductory geology sequence. (Full article...) -
Image 20
Auguste Bravais (French pronunciation: [oɡyst bʁavɛ]; 23 August 1811, Annonay, Ardèche – 30 March 1863, Le Chesnay, France) was a French physicist known for his work in crystallography, the conception of Bravais lattices, and the formulation of Bravais law. Bravais also studied magnetism, the northern lights, meteorology, geobotany, phyllotaxis, astronomy, statistics and hydrography.
He studied at the Collège Stanislas in Paris before joining the École Polytechnique in 1829, where he was a classmate of groundbreaking mathematician Évariste Galois, whom Bravais actually beat in a scholastic mathematics competition. Towards the end of his studies he became a naval officer, and sailed on the Finistere in 1832 as well as the Loiret afterwards. He took part in hydrographic work along the Algerian Coast. He participated in the Recherche expedition and helped the Lilloise in Spitzbergen and Lapland. (Full article...) -
Image 21
Johann Gottlob Lehmann (4 August 1719 – 22 January 1767) was a German mineralogist and geologist noted for his work and research contributions to the geologic record leading to the development of stratigraphy. (Full article...) -
Image 22

Auguste Michel-Lévy (7 August 1844 – 27 September 1911) was a French geologist. He was born in Paris. (Full article...) -
Image 23

James Nicol
James Nicol FRSE FGS (12 August 1810 – 8 April 1879) was a Scottish geologist. (Full article...) -
Image 24Edward Sydney Simpson (11 March 1875 – 30 August 1939) was an Australian mineralogist and geochemist.
Simpson was born in Woollahra, New South Wales to an Irish father and English mother. He was educated at Sydney Grammar School and the University of Sydney where he graduated B.E. with honours, in 1895 and D.Sc. in 1919. (Full article...) -
Image 25Friedrich Martin Berwerth (16 November 1850, Schäßburg – 22 September 1918, Vienna) was an Austrian mineralogist and petrographer known for his work with meteorites.
He was a student at the Universities of Vienna, Graz and Heidelberg, and following graduation, worked as an assistant to Gustav Tschermak von Seysenegg at the Mineralogisch-Petrographischen Institut in Vienna (1874). Later the same year, he began work as an assistant at the Imperial Hofmineralien Cabinet. In 1888 he became a curator, followed by titles of leiter (1895) and director (1904) of the mineralogy-petrography department. (Full article...)
Related portals
Get involved
For editor resources and to collaborate with other editors on improving Wikipedia's Minerals-related articles, see WikiProject Rocks and minerals.
General images
-
Image 1Black andradite, an end-member of the orthosilicate garnet group. (from Mineral)
-
Image 2Mohs hardness kit, containing one specimen of each mineral on the ten-point hardness scale (from Mohs scale)
-
Image 4Red cinnabar (HgS), a mercury ore, on dolomite. (from Mineral)
-
Image 5Asbestiform tremolite, part of the amphibole group in the inosilicate subclass (from Mineral)
-
Image 7Hübnerite, the manganese-rich end-member of the wolframite series, with minor quartz in the background (from Mineral)
-
Image 8Mohs Scale versus Absolute Hardness (from Mineral)
-
Image 9Pink cubic halite (NaCl; halide class) crystals on a nahcolite matrix (NaHCO3; a carbonate, and mineral form of sodium bicarbonate, used as baking soda). (from Mineral)
-
Image 10Epidote often has a distinctive pistachio-green colour. (from Mineral)
-
Image 11When minerals react, the products will sometimes assume the shape of the reagent; the product mineral is termed a pseudomorph of (or after) the reagent. Illustrated here is a pseudomorph of kaolinite after orthoclase. Here, the pseudomorph preserved the Carlsbad twinning common in orthoclase. (from Mineral)
-
Image 15Sphalerite crystal partially encased in calcite from the Devonian Milwaukee Formation of Wisconsin (from Mineral)
-
Image 16Gypsum desert rose (from Mineral)
-
Image 18Muscovite, a mineral species in the mica group, within the phyllosilicate subclass (from Mineral)
-
Image 19Schist is a metamorphic rock characterized by an abundance of platy minerals. In this example, the rock has prominent sillimanite porphyroblasts as large as 3 cm (1.2 in). (from Mineral)
-
Image 21Diamond is the hardest natural material, and has a Mohs hardness of 10. (from Mineral)
-
Image 22Perfect basal cleavage as seen in biotite (black), and good cleavage seen in the matrix (pink orthoclase). (from Mineral)
-
Image 24An example of elbaite, a species of tourmaline, with distinctive colour banding. (from Mineral)
-
Image 26Native gold. Rare specimen of stout crystals growing off of a central stalk, size 3.7 x 1.1 x 0.4 cm, from Venezuela. (from Mineral)
In the news
- 3 May 2024 –
- Panama bans First Quantum Minerals from extracting copper following the closure of its Cobre Panamá mine last year. (Reuters) (The Globe and Mail)
Did you know ...?
- ... that while bultfonteinite (pictured) was discovered as early as 1903, the mineral wasn't described until 1932?
- ... that the minerals armalcolite, pyroxferroite and tranquillityite were discovered in lunar rocks?
- ... that taaffeite, one of the world's rarest gemstones, is the first mineral known to contain both beryllium and magnesium as essential components?
- ... that the newly discovered mineral krotite likely was one of the earliest minerals formed in the Solar System?
Subcategories
Topics
| Overview | ||
|---|---|---|
| Common minerals | ||
Ore minerals, mineral mixtures and ore deposits | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ores |
| ||||||||
| Deposit types | |||||||||
| Borates | |||||
|---|---|---|---|---|---|
| Carbonates | |||||
| Oxides |
| ||||
| Phosphates | |||||
| Silicates | |||||
| Sulfides | |||||
| Other |
| ||||
| Crystalline | |||||||
|---|---|---|---|---|---|---|---|
| Cryptocrystalline | |||||||
| Amorphous | |||||||
| Miscellaneous | |||||||
| Notable varieties |
| ||||||
| Oxide minerals |
| ||||
|---|---|---|---|---|---|
| Silicate minerals | |||||
| Other | |||||
Gemmological classifications by E. Ya. Kievlenko (1980), updated | |||||||||
| Jewelry stones |
| ||||||||
| Jewelry-Industrial stones |
| ||||||||
| Industrial stones |
| ||||||||
Mineral identification | |
|---|---|
| "Special cases" ("native elements and organic minerals") |
|
|---|---|
| "Sulfides and oxides" |
|
| "Evaporites and similars" |
|
| "Mineral structures with tetrahedral units" (sulfate anion, phosphate anion, silicon, etc.) |
|
Associated Wikimedia
The following Wikimedia Foundation sister projects provide more on this subject:
-
Commons
Free media repository -
Wikibooks
Free textbooks and manuals -
Wikidata
Free knowledge base -
Wikinews
Free-content news -
Wikiquote
Collection of quotations -
Wikisource
Free-content library -
Wikiversity
Free learning tools -
Wiktionary
Dictionary and thesaurus