Methoxy arachidonyl fluorophosphonate, commonly referred as MAFP, is an irreversible active site-directed enzyme inhibitor that inhibits nearly all serine hydrolases and serine proteases.[1] It inhibits phospholipase A2 and fatty acid amide hydrolase with special potency, displaying IC50 values in the low-nanomolar range. In addition, it binds to the CB1 receptor in rat brain membrane preparations (IC50 = 20 nM),[2] but does not appear to agonize or antagonize the receptor,[3] though some related derivatives do show cannabinoid-like properties.[4]
| |
| Names | |
|---|---|
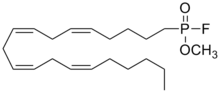
| Preferred IUPAC name
Methyl [(5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraen-1-yl]phosphonofluoridate | |
| Other names
MAFP
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C21H36FO2P | |
| Molar mass | 370.5 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
See also
edit- DIFP – diisopropyl fluorophosphate, a related inhibitor
- IDFP – isopropyl dodecylfluorophosphonate, another related inhibitor with selectivity for FAAH and MAGL
- Activity-based probes
References
edit- ^ Hoover HS, Blankman JL, Niessen S, Cravatt BF (July 2008). "Selectivity of inhibitors of endocannabinoid biosynthesis evaluated by activity-based protein profiling". Bioorg. Med. Chem. Lett. 18 (22): 5838–41. doi:10.1016/j.bmcl.2008.06.091. PMC 2634297. PMID 18657971.
- ^ Deutsch DG, Omeir R, Arreaza G, Salehani D, Prestwich GD, Huang Z, Howlett A (1997). "Methyl arachidonyl fluorophosphonate: a potent irreversible inhibitor of anandamide amidase". Biochem. Pharmacol. 53 (3): 255–60. doi:10.1016/s0006-2952(96)00830-1. PMID 9065728.
- ^ Savinainen JR, Saario SM, Niemi R, Järvinen T, Laitinen JT (2003). "An optimized approach to study endocannabinoid signaling: evidence against constitutive activity of rat brain adenosine A1 and cannabinoid CB1 receptors". Br. J. Pharmacol. 140 (8): 1451–9. doi:10.1038/sj.bjp.0705577. PMC 1574161. PMID 14623770.
- ^ Martin BR, Beletskaya I, Patrick G, Jefferson R, Winckler R, Deutsch DG, Di Marzo V, Dasse O, Mahadevan A, Razdan RK. Cannabinoid properties of methylfluorophosphonate analogs. J Pharmacol Exp Ther. 2000 Sep;294(3):1209-18. PMID 10945879