ATP-binding cassette super-family G member 2 is a protein that in humans is encoded by the ABCG2 gene.[6][7] ABCG2 has also been designated as CDw338 (cluster of differentiation w338). ABCG2 is a translocation protein used to actively pump drugs and other compounds against their concentration gradient using the bonding and hydrolysis of ATP as the energy source.[1]
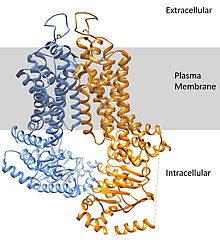
ABCG2 forms into a homodimer to assume its active transport conformation. The dimer weighs approximately 144 kDa. The expression of this transport protein is highly conserved throughout the animal kingdom, pointing to its importance.[8]
Substrate binding with compounds occurs in the large central cavity. ABCG2 can bind to a broad range of compounds but binds strongest to flat, polycyclic chemicals with lots of hydrophobic character.[1]
Function
editThe membrane-associated protein encoded by this gene is included in the superfamily of ATP-binding cassette (ABC) transporters. ABC proteins transport various molecules across extra- and intra-cellular membranes. The active transport of chemicals requires a source of energy to catalyze the conformational changes the protein undergoes. The nucleotide-binding domains (NBDs) found towards the N-terminus allow binding to ATP molecules. The NBD and the transmembrane domain (TMD) are the most conserved region of the transporter in various animal groups, highlighting the importance of these regions for overall protein function.[8] Additionally, many ABC transporters have conserved NBD regions showing the strict conformation needed to bind ATP molecules.[1]
ABC genes are divided into seven distinct subfamilies (ABC1, MDR/TAP, MRP, ALD, OABP, GCN20, White). This protein is a member of the White subfamily. Alternatively referred to as the breast cancer resistance protein (BCRP), this protein functions as a xenobiotic transporter which may play a role in multi-drug resistance to chemotherapeutic agents including mitoxantrone and camptothecin analogues.[8] Early observations of significant ABCG2-mediated resistance to anthracyclines were subsequently attributed mutations encountered in vitro but not in nature or the clinic. Significant expression of this protein has been observed in the placenta,[9] and it has been shown to have a role in protecting the fetus from xenobiotics in the maternal circulation.[10]
The transporter has been shown to play protective roles in blocking absorption at the apical membrane of the intestine, and at the blood–testis barrier,[10] the blood–brain barrier,[10] and the membranes of hematopoietic progenitor and other stem cells. At the apical membranes of the liver and kidney, it enhances excretion of xenobiotics. In the lactating mammary gland, it has a role on excreting vitamins such as riboflavin and biotin into milk.[10] Xenobiotic toxins compete for the substrate binding domain of ABCG2 potentially causing toxins to concentrate in the breast milk.[8] In the kidney and gastrointestinal tract, it has a role in urate excretion.
The protein also carries the Jr(a) antigen, which defines the Junior blood group system.[11]
Interactive pathway map
edit| Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
Fluorouracil (5-FU) Activity edit
|
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
Irinotecan Pathway edit
|
Inhibition
editIt is inhibited by some calcium channel blockers such as amlodipine, felodipine and nifedipine.[12] The fungal toxin fumitremorgin C (FTC) inhibits the protein but has neurotoxic side effects. A synthetic tetracyclic analog of FTC called Ko-143 inhibits ABCG2.[13]
See also
editReferences
edit- ^ a b c d Taylor NM, Manolaridis I, Jackson SM, Kowal J, Stahlberg H, Locher KP (June 2017). "Structure of the human multidrug transporter ABCG2". Nature. 546 (7659): 504–509. Bibcode:2017Natur.546..504T. doi:10.1038/nature22345. hdl:20.500.11850/233014. PMID 28554189. S2CID 4461745.
- ^ a b c GRCh38: Ensembl release 89: ENSG00000118777 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000029802 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Allikmets R, Gerrard B, Hutchinson A, Dean M (October 1996). "Characterization of the human ABC superfamily: isolation and mapping of 21 new genes using the expressed sequence tags database". Human Molecular Genetics. 5 (10): 1649–1655. doi:10.1093/hmg/5.10.1649. PMID 8894702.
- ^ Doyle LA, Yang W, Abruzzo LV, Krogmann T, Gao Y, Rishi AK, Ross DD (December 1998). "A multidrug resistance transporter from human MCF-7 breast cancer cells". Proceedings of the National Academy of Sciences of the United States of America. 95 (26): 15665–15670. Bibcode:1998PNAS...9515665D. doi:10.1073/pnas.95.26.15665. PMC 28101. PMID 9861027.
- ^ a b c d Robey RW, To KK, Polgar O, Dohse M, Fetsch P, Dean M, Bates SE (January 2009). "ABCG2: a perspective". Advanced Drug Delivery Reviews. 61 (1): 3–13. doi:10.1016/j.addr.2008.11.003. PMC 3105088. PMID 19135109.
- ^ "Entrez Gene: ABCG2 ATP-binding cassette, sub-family G (WHITE), member 2".
- ^ a b c d Vlaming ML, Lagas JS, Schinkel AH (January 2009). "Physiological and pharmacological roles of ABCG2 (BCRP): recent findings in Abcg2 knockout mice". Advanced Drug Delivery Reviews. 61 (1): 14–25. doi:10.1016/j.addr.2008.08.007. PMID 19118589.
- ^ Kniffin CL (2013). "OMIM entry # 614490 – BLOOD GROUP, JUNIOR SYSTEM; JR". Online Mendelian Inheritance in Man. Retrieved 1 September 2019.
- ^ Ghosh S, Sircar M (October 2008). "Calcium channel blocker overdose: experience with amlodipine". Indian Journal of Critical Care Medicine. 12 (4). Jaypee Brothers Medical Publishing: 190–193. doi:10.4103/0972-5229.45080. PMC 2738322. PMID 19742263.
- ^ Jackson SM, Manolaridis I, Kowal J, Zechner M, Taylor NM, Bause M, et al. (April 2018). "Structural basis of small-molecule inhibition of human multidrug transporter ABCG2". Nature Structural & Molecular Biology. 25 (4): 333–340. doi:10.1038/s41594-018-0049-1. hdl:20.500.11850/256191. PMID 29610494. S2CID 4617388.
Further reading
edit- Hazai E, Bikádi Z (April 2008). "Homology modeling of breast cancer resistance protein (ABCG2)". Journal of Structural Biology. 162 (1): 63–74. doi:10.1016/j.jsb.2007.12.001. PMID 18249138.
- Abbott BL (January 2006). "ABCG2 (BCRP): a cytoprotectant in normal and malignant stem cells". Clinical Advances in Hematology & Oncology. 4 (1): 63–72. PMID 16562373.
- Schmitz G, Langmann T, Heimerl S (October 2001). "Role of ABCG1 and other ABCG family members in lipid metabolism". Journal of Lipid Research. 42 (10): 1513–1520. doi:10.1016/S0022-2275(20)32205-7. PMID 11590207.
- Ejendal KF, Hrycyna CA (October 2002). "Multidrug resistance and cancer: the role of the human ABC transporter ABCG2". Current Protein & Peptide Science. 3 (5): 503–511. doi:10.2174/1389203023380521. PMID 12369998.
- Doyle L, Ross DD (October 2003). "Multidrug resistance mediated by the breast cancer resistance protein BCRP (ABCG2)". Oncogene. 22 (47): 7340–7358. doi:10.1038/sj.onc.1206938. PMID 14576842. S2CID 9648841.
- Sugimoto Y, Tsukahara S, Ishikawa E, Mitsuhashi J (August 2005). "Breast cancer resistance protein: molecular target for anticancer drug resistance and pharmacokinetics/pharmacodynamics". Cancer Science. 96 (8): 457–465. doi:10.1111/j.1349-7006.2005.00081.x. PMC 11158713. PMID 16108826. S2CID 20734576.
- Ishikawa T, Tamura A, Saito H, Wakabayashi K, Nakagawa H (October 2005). "Pharmacogenomics of the human ABC transporter ABCG2: from functional evaluation to drug molecular design". Die Naturwissenschaften. 92 (10): 451–463. Bibcode:2005NW.....92..451I. doi:10.1007/s00114-005-0019-4. PMID 16160819. S2CID 22151149.
- Krishnamurthy P, Schuetz JD (2006). "Role of ABCG2/BCRP in biology and medicine". Annual Review of Pharmacology and Toxicology. 46: 381–410. doi:10.1146/annurev.pharmtox.46.120604.141238. PMID 16402910.
- Robey RW, Polgar O, Deeken J, To KW, Bates SE (March 2007). "ABCG2: determining its relevance in clinical drug resistance". Cancer and Metastasis Reviews. 26 (1): 39–57. doi:10.1007/s10555-007-9042-6. PMID 17323127. S2CID 11293439.
External links
edit- ABCG2+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human ABCG2 genome location and ABCG2 gene details page in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: Q9UNQ0 (Broad substrate specificity ATP-binding cassette transporter ABCG2) at the PDBe-KB.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.




