This article needs additional citations for verification. (November 2016) |
Enrofloxacin, sold under the brand name Baytril, among others, is a fluoroquinolone antibiotic used for the treatment of animals.[1] It is a bactericidal agent.[1]
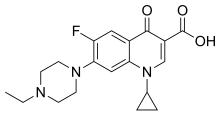 | |
| Clinical data | |
|---|---|
| Trade names | Baytril, others |
| AHFS/Drugs.com | International Drug Names |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, subcutaneous, intramuscular |
| ATCvet code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 80% in dogs, 65-75% in sheep [4] |
| Metabolism | Kidney and non-kidney[4] |
| Elimination half-life | 4–5 hours in dogs, 6 hours in cats, 1.5 - 4.5 hours in sheep |
| Excretion | Bile duct (70%); kidney (30%)[5] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.131.355 |
| Chemical and physical data | |
| Formula | C19H22FN3O3 |
| Molar mass | 359.401 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 219 to 221 °C (426 to 430 °F) |
| |
| |
| (verify) | |
The bactericidal activity of enrofloxacin is concentration-dependent, with susceptible bacteria cell death occurring within 20–30 minutes of exposure. Enrofloxacin has demonstrated a significant post-antibiotic effect for both Gram-negative and Gram-positive bacteria and is active in both stationary and growth phases of bacterial replication. Enrofloxacin is partially deethylated by CYP450 into the active metabolite ciprofloxacin, which is also a fluoroquinolone antibiotic.
In September 2005, the FDA withdrew approval of enrofloxacin for use in water to treat flocks of poultry, as the practice was noted to promote the evolution of fluoroquinolone-resistant strains of the bacterium Campylobacter, a human pathogen.[6] Enrofloxacin is available as a fixed-dose combination medication with silver sulfadiazine for the treatment of canine otitis externa.[7] It is available as a generic medication.
Activity and susceptibility data edit
Enrofloxacin is a synthetic antibacterial agent from the class of the fluoroquinolone carboxylic acid derivatives. It has antibacterial activity against a broad spectrum of Gram-negative and Gram-positive bacteria. It is effective against:
- Pseudomonas aeruginosa
- Klebsiella
- Escherichia coli
- Enterobacter
- Campylobacter
- Shigella
- Salmonella
- Aeromonas
- Haemophilus
- Proteus
- Yersinia
- Serratia
- Vibrio
- Brucella
- Chlamydia trachomatis
- Staphylococcus (including penicillinase-producing and methicillin-resistant strains)
- Mycoplasma
- Mycobacterium
Variable activity against:
Ineffective against:
The following data represent minimum inhibitory concentration ranges for a few medically significant bacterial pathogens:
- Escherichia coli - 0.022 - 0.03 µg/ml
- Staphylococcus aureus - 0.0925 - 64 µg/ml
- Pseudomonas aeruginosa - 0.05 µg/ml
Adverse effects/warnings edit
Enrofloxacin was banned for poultry use in the United States in 2005.[8]
Overdosage/acute toxicity edit
It is unlikely that an acute overdose of either compound would result in symptoms more serious than either anorexia or vomiting, but the adverse effects noted above could occur. Dogs receiving 10 times the labeled dosage rate of enrofloxacin for at least 14 days developed only vomiting and anorexia. Death did occur in some dogs when fed 25 times the labeled rate for 11 days, however.
- Oral LD50: greater than 5000 mg/kg
- Dermal LD50: greater than 2000 mg/kg
- Inhalation LD50: greater than 3547 mg/m3 (4-hour exposure)
- Eye effects: irritant; reversible in less than 7 days. In cats, it can produce sudden onset blindness when administered by injection, as it is retinotoxic.[citation needed]
Degradation edit
The brown rot fungus Gloeophyllum striatum can degrade the fluoroquinolone enrofloxacin using hydroxyl radicals.[9]
References edit
- ^ a b c "Baytril- enrofloxacin injection, solution". DailyMed. 13 October 2022. Retrieved 13 April 2023.
- ^ "Baytril- enrofloxacin injection, solution". DailyMed. 22 March 2023. Retrieved 13 April 2023.
- ^ "Baytril- enrofloxacin tablet, chewable". DailyMed. 25 November 2021. Retrieved 13 April 2023.
- ^ a b Plumb DC. "Enrofloxacin". Veterinary Drug Handbook (fifth ed.).
- ^ "Baytril: Excretion and Elimination". Bayer HealthCare AG. Archived from the original on 6 January 2014. Retrieved 6 January 2014.
- ^ "Enrofloxacin for Poultry". U.S. Food and Drug Administration (FDA). Archived from the original on 10 February 2007. Retrieved 7 March 2007.
- ^ "Baytril Otic- enrofloxacin, silver sulfadiazine emulsion". DailyMed. 27 March 2023. Retrieved 13 April 2023.
- ^ Morgan D, Kaufman M (30 April 2005). "Lawmakers' Help for Drug Firm Tests Limits". Washington Post.
FDA Calls Efforts For Bayer Illegal
- ^ Wetzstein HG, Schmeer N, Karl W (November 1997). "Degradation of the fluoroquinolone enrofloxacin by the brown rot fungus Gloeophyllum striatum: identification of metabolites". Applied and Environmental Microbiology. 63 (11): 4272–81. Bibcode:1997ApEnM..63.4272W. doi:10.1128/AEM.63.11.4272-4281.1997. PMC 168747. PMID 9361414.