In enzymology, tryptophan 2,3-dioxygenase (EC 1.13.11.11) is a heme enzyme that catalyzes the oxidation of L-tryptophan (L-Trp) to N-formyl-L-kynurenine, as the first and rate-limiting step of the kynurenine pathway.
| TDO2 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | TDO2, TDO, TO, TPH2, TRPO, tryptophan 2,3-dioxygenase, HYPTRP | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 191070 MGI: 1928486 HomoloGene: 4132 GeneCards: TDO2 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
- L-tryptophan + O2 ⇌ N-formyl-L-kynurenine
Tryptophan 2,3-dioxygenase plays a central role in the physiological regulation of tryptophan flux in the human body, as part of the overall biological process of tryptophan metabolism. TDO catalyses the first and rate-limiting step of tryptophan degradation along the kynurenine pathway and thereby regulates systemic tryptophan levels.[5] In humans, tryptophan 2,3-dioxygenase is encoded by the TDO2 gene.[6]
Function edit
| Tryptophan 2,3-dioxygenase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
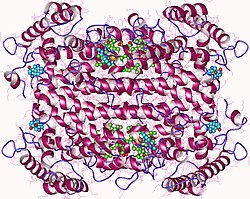
| Crystal structure of the tryptophan 2,3-dioxygenase from xanthomonas campestris | |||||||||
| Identifiers | |||||||||
| EC no. | 1.13.11.11 | ||||||||
| CAS no. | 9014-51-1 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
This enzyme belongs to the family of oxidoreductases, specifically those acting on single donors with O2 as oxidant and incorporation of two atoms of oxygen into the substrate (oxygenases). This family includes tryptophan 2,3-dioxygenase (TDO, also sometimes referred to as tryptophan oxygenase and L-tryptophan pyrrolase) and the closely related indoleamine 2,3-dioxygenase enzyme (IDO).[7][8] Both TDO and IDO contain one noncovalently bound heme per monomer; TDO is usually tetrameric, whereas IDO is monomeric.
Tryptophan 2,3-dioxygenase was initially discovered in the 1930s[9] and is found in both eukaryotes and prokaryotes. Expression of tryptophan 2,3-dioxygenase in mammals is normally restricted to the liver, but it has been identified in the brain and epididymis of some species, and, in some tissues, its production can be induced in response to stimuli.[8] TDO from rat was the first to be expressed recombinantly (in E. coli).[10] Human TDO has also been expressed.[11][12]
The same family of enzymes also includes an indole 2,3-dioxygenase from Shewanella oneidensis[13] and PrnB, the second enzyme in the pyrrolnitrin biosynthesis pathway from Pseudomonas fluorescens,[14] although dioxygenase activity has not been demonstrated for either as yet. In 2007, a new enzyme with the ability to catalyze L-tryptophan dioxygenation, IDO2, was identified.[15]
Structure edit
Tryptophan 2,3-dioxygenase is a heme-containing cytosolic enzyme encoded by gene TDO2.[5] Crystallographic studies of Xanthomonas campestris TD)[13] and Ralstonia metallidurans TDO)[16] have revealed that their structures are essentially identical and are intimately associated homotetrameric enzymes.[17] They are best described as a dimer of dimers because the N terminal residues of each monomer form part of the substrate binding site in an adjacent monomer. The proteins are completely helical, and a flexible loop, involved in L-tryptophan binding, is observed just outside the active-site pocket. This loop appears to be substrate-binding induced, as it is observed only in crystals grown in the presence of L-tryptophan.[17]
There are two TDO structures available with substrate (tryptophan) bound.[17],[18]
Mechanism edit
Early proposals for the mechanism of tryptophan oxidation were presented by Sono and Dawson.[19] This suggested a base-catalysed abstraction mechanism, involving only the ferrous (FeII) heme. It is assumed that TDO and IDO react by the same mechanism, although there is no concrete evidence for that. In IDO, a ferryl heme (FeIV) has been identified during turnover.[20][21] Mechanistic proposals have therefore been adjusted to include the formation of ferryl heme during the mechanism.[22] TDO is assumed to react in the same way, but a ferryl heme has not been observed in TDO. See also discussion of mechanism for indoleamine 2,3-dioxygenase.
Clinical significance edit
It has been shown that tryptophan 2,3-dioxygenase is expressed in a significant proportion of human tumors.[5] In the same study, tryptophan 2,3-dioxygenase expression by tumors prevented their rejection by immunized mice. A tryptophan 2,3-dioxygenase inhibitor developed by the group restored the ability of these mice to reject tryptophan 2,3-dioxygenase-expressed tumors, demonstrating that tryptophan 2,3-dioxygenase inhibitors display potential in cancer therapy.
Another study showed that tryptophan 2,3-dioxygenase is potentially involved in the metabolic pathway responsible for anxiety-related behavior.[23] Generating mice deficient for tryptophan 2,3-dioxygenase and comparing them to the wild type, the group found that the tryptophan 2,3-dioxygenase-deficient mice showed increased plasma levels not only of tryptophan, but also of serotonin and 5-HIAA in the hippocampus and midbrain. A variety of tests, such as elevated plus maze and open-field tests showed anxiolytic modulation in these knock-out mice, the findings demonstrating a direct link between tryptophan 2,3-dioxygenase and tryptophan metabolism and anxiety-related behavior under physiological conditions.
See also edit
References edit
- ^ a b c ENSG00000151790 GRCh38: Ensembl release 89: ENSG00000262635, ENSG00000151790 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000028011 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ a b c Pilotte L, Larrieu P, Stroobant V, Colau D, Dolusic E, Frédérick R, De Plaen E, Uyttenhove C, Wouters J, Masereel B, Van den Eynde BJ (February 2012). "Reversal of tumoral immune resistance by inhibition of tryptophan 2,3-dioxygenase". Proceedings of the National Academy of Sciences of the United States of America. 109 (7): 2497–2502. Bibcode:2012PNAS..109.2497P. doi:10.1073/pnas.1113873109. PMC 3289319. PMID 22308364.
- ^ "Entrez Gene: TDO2 tryptophan 2,3-dioxygenase".
- ^ Efimov I, Basran J, Thackray SJ, Handa S, Mowat CG, Raven EL (April 2011). "Structure and reaction mechanism in the heme dioxygenases". Biochemistry. 50 (14): 2717–2724. doi:10.1021/bi101732n. PMC 3092302. PMID 21361337.
- ^ a b Thackray SJ, Bruckmann C, Mowat CG, Forouhar F, Chapman SK, Tong L (2008). "Indoleamine 2,3-dioxygenase and tryptophan 2,3-dioxygenase". Handbook of Metalloproteins. doi:10.1002/0470028637.met223.
- ^ Kotake Y.; Masayama I. Z. (1936). "Über den Mechanismus der Kynureninbildung aus Tryptophan". Z. Physiol. Chem. 243: 237–244. doi:10.1515/bchm2.1936.243.6.237.
- ^ Ren S, Liu H, Licad E, Correia MA (September 1996). "Expression of rat liver tryptophan 2,3-dioxygenase in Escherichia coli: structural and functional characterization of the purified enzyme". Archives of Biochemistry and Biophysics. 333 (1): 96–102. doi:10.1006/abbi.1996.0368. PMID 8806758.
- ^ Batabyal D, Yeh SR (December 2007). "Human tryptophan dioxygenase: a comparison to indoleamine 2,3-dioxygenase". Journal of the American Chemical Society. 129 (50): 15690–15701. doi:10.1021/ja076186k. PMID 18027945.
- ^ Basran J, Rafice SA, Chauhan N, Efimov I, Cheesman MR, Ghamsari L, Raven EL (April 2008). "A kinetic, spectroscopic, and redox study of human tryptophan 2,3-dioxygenase". Biochemistry. 47 (16): 4752–4760. doi:10.1021/bi702393b. PMID 18370401.
- ^ a b Forouhar F, Anderson JL, Mowat CG, Vorobiev SM, Hussain A, Abashidze M, Bruckmann C, Thackray SJ, Seetharaman J, Tucker T, Xiao R, Ma LC, Zhao L, Acton TB, Montelione GT, Chapman SK, Tong L (January 2007). "Molecular insights into substrate recognition and catalysis by tryptophan 2,3-dioxygenase". Proceedings of the National Academy of Sciences of the United States of America. 104 (2): 473–478. Bibcode:2007PNAS..104..473F. doi:10.1073/pnas.0610007104. PMC 1766409. PMID 17197414.
- ^ De Laurentis W, Khim L, Anderson JL, Adam A, Johnson KA, Phillips RS, Chapman SK, van Pee KH, Naismith JH (October 2007). "The second enzyme in pyrrolnitrin biosynthetic pathway is related to the heme-dependent dioxygenase superfamily". Biochemistry. 46 (43): 12393–12404. doi:10.1021/bi7012189. PMC 3326534. PMID 17924666.
- ^ Ball HJ, Sanchez-Perez A, Weiser S, Austin CJ, Astelbauer F, Miu J, McQuillan JA, Stocker R, Jermiin LS, Hunt NH (July 2007). "Characterization of an indoleamine 2,3-dioxygenase-like protein found in humans and mice". Gene. 396 (1): 203–213. doi:10.1016/j.gene.2007.04.010. PMID 17499941.
- ^ Zhang Y, Kang SA, Mukherjee T, Bale S, Crane BR, Begley TP, Ealick SE (January 2007). "Crystal structure and mechanism of tryptophan 2,3-dioxygenase, a heme enzyme involved in tryptophan catabolism and in quinolinate biosynthesis". Biochemistry. 46 (1): 145–155. doi:10.1021/bi0620095. PMID 17198384.
- ^ a b c Thackray SJ, Mowat CG, Chapman SK (December 2008). "Exploring the mechanism of tryptophan 2,3-dioxygenase". Biochemical Society Transactions. 36 (Pt 6): 1120–1123. doi:10.1042/bst0361120. PMC 2652831. PMID 19021508.
- ^ Lewis-Ballester A, Forouhar F, Kim SM, Lew S, Wang Y, Karkashon S, Seetharaman J, Batabyal D, Chiang BY, Hussain M, Correia MA, Yeh SR, Tong L (October 2016). "Molecular basis for catalysis and substrate-mediated cellular stabilization of human tryptophan 2,3-dioxygenase". Scientific Reports. 6: 35169. Bibcode:2016NatSR...635169L. doi:10.1038/srep35169. PMC 5071832. PMID 27762317.
- ^ Sono M, Roach MP, Coulter ED, Dawson JH (November 1996). "Heme-Containing Oxygenases". Chemical Reviews. 96 (7): 2841–2888. doi:10.1021/cr9500500. PMID 11848843.
- ^ Lewis-Ballester A, Batabyal D, Egawa T, Lu C, Lin Y, Marti MA, Capece L, Estrin DA, Yeh SR (October 2009). "Evidence for a ferryl intermediate in a heme-based dioxygenase". Proceedings of the National Academy of Sciences of the United States of America. 106 (41): 17371–17376. Bibcode:2009PNAS..10617371L. doi:10.1073/pnas.0906655106. PMC 2765089. PMID 19805032.
- ^ Yanagisawa S, Yotsuya K, Hashiwaki Y, Horitani M, Sugimoto H, Shiro Y, Appelman EH, Ogura T (2010). "Identification of the Fe-O2 and the Fe=O heme species for indoleamine 2,3-dioxygenase during catalytic turnover". Chem Lett. 39: 36–37. doi:10.1246/cl.2010.36.
- ^ Basran J, Efimov I, Chauhan N, Thackray SJ, Krupa JL, Eaton G, Griffith GA, Mowat CG, Handa S, Raven EL (October 2011). "The mechanism of formation of N-formylkynurenine by heme dioxygenases". Journal of the American Chemical Society. 133 (40): 16251–16257. doi:10.1021/ja207066z. PMC 3210546. PMID 21892828.
- ^ Kanai M, Funakoshi H, Takahashi H, Hayakawa T, Mizuno S, Matsumoto K, Nakamura T (March 2009). "Tryptophan 2,3-dioxygenase is a key modulator of physiological neurogenesis and anxiety-related behavior in mice". Molecular Brain. 2 (8): 8. doi:10.1186/1756-6606-2-8. PMC 2673217. PMID 19323847.
Further reading edit
- Comings DE, Muhleman D, Dietz GW, Donlon T (February 1991). "Human tryptophan oxygenase localized to 4q31: possible implications for alcoholism and other behavioral disorders". Genomics. 9 (2): 301–308. doi:10.1016/0888-7543(91)90257-F. PMID 2004780.
- Comings DE, Muhleman D, Dietz G, Sherman M, Forest GL (September 1995). "Sequence of human tryptophan 2,3-dioxygenase (TDO2): presence of a glucocorticoid response-like element composed of a GTT repeat and an intronic CCCCT repeat". Genomics. 29 (2): 390–396. doi:10.1006/geno.1995.9990. PMID 8666386.
- Dick R, Murray BP, Reid MJ, Correia MA (August 2001). "Structure--function relationships of rat hepatic tryptophan 2,3-dioxygenase: identification of the putative heme-ligating histidine residues". Archives of Biochemistry and Biophysics. 392 (1): 71–78. doi:10.1006/abbi.2001.2420. PMID 11469796.
- Kudo Y, Boyd CA, Sargent IL, Redman CW (March 2003). "Decreased tryptophan catabolism by placental indoleamine 2,3-dioxygenase in preeclampsia". American Journal of Obstetrics and Gynecology. 188 (3): 719–726. doi:10.1067/mob.2003.156. PMID 12634647.
- Nabi R, Serajee FJ, Chugani DC, Zhong H, Huq AH (February 2004). "Association of tryptophan 2,3 dioxygenase gene polymorphism with autism". American Journal of Medical Genetics. Part B, Neuropsychiatric Genetics. 125B (1): 63–68. doi:10.1002/ajmg.b.20147. PMID 14755447. S2CID 26302464.
- Guillemin GJ, Smythe G, Takikawa O, Brew BJ (January 2005). "Expression of indoleamine 2,3-dioxygenase and production of quinolinic acid by human microglia, astrocytes, and neurons". Glia. 49 (1): 15–23. doi:10.1002/glia.20090. PMID 15390107. S2CID 31823904.
- Baharvand H, Hashemi SM, Kazemi Ashtiani S, Farrokhi A (2006). "Differentiation of human embryonic stem cells into hepatocytes in 2D and 3D culture systems in vitro". The International Journal of Developmental Biology. 50 (7): 645–652. doi:10.1387/ijdb.052072hb. PMID 16892178.
- Batabyal D, Yeh SR (December 2007). "Human tryptophan dioxygenase: a comparison to indoleamine 2,3-dioxygenase". Journal of the American Chemical Society. 129 (50): 15690–15701. doi:10.1021/ja076186k. PMID 18027945.
- Gupta R, Fu R, Liu A, Hendrich MP (2007). "EPR and Mossbauer spectroscopy show inequivalent hemes in tryptophan dioxygenase". Journal of the American Chemical Society. 132 (3): 1098–1109. doi:10.1021/ja908851e. PMC 4251817. PMID 20047315.
- Allegri G, Ragazzi E, Bertazzo A, Costa CV, Rocchi R (2003). "Tryptophan Metabolism Along the Kynurenine Pathway in Rats". Developments in Tryptophan and Serotonin Metabolism. Advances in Experimental Medicine and Biology. Vol. 527. pp. 481–496. doi:10.1007/978-1-4615-0135-0_56. ISBN 978-1-4613-4939-6. PMID 15206766.
- Chung LW, Li X, Sugimoto H, Shiro Y, Morokuma K (September 2008). "Density functional theory study on a missing piece in understanding of heme chemistry: the reaction mechanism for indoleamine 2,3-dioxygenase and tryptophan 2,3-dioxygenase". Journal of the American Chemical Society. 130 (37): 12299–12309. doi:10.1021/ja803107w. PMID 18712870.
- Allegri G, Ragazzi E, Bertazzo A, Biasiolo M, Costa CV (2003). "Tryptophan Metabolism in Rabbits". Developments in Tryptophan and Serotonin Metabolism. Advances in Experimental Medicine and Biology. Vol. 527. pp. 473–479. doi:10.1007/978-1-4615-0135-0_55. ISBN 978-1-4613-4939-6. PMID 15206765.




