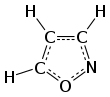
Isoxazole is an electron-rich azole with an oxygen atom next to the nitrogen. It is also the class of compounds containing this ring. Isoxazolyl is the univalent functional group derived from isoxazole.
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,2-Oxazole[1] | |||
| Other names
isoxazole
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.472 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H3NO | |||
| Molar mass | 69.06202 g/mol | ||
| Density | 1.075 g/ml | ||
| Boiling point | 95 °C (203 °F; 368 K) | ||
| Acidity (pKa) | -3.0 (of conjugate acid)[2] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Occurrence
editIsoxazole rings are found in some natural products, such as ibotenic acid and muscimol.
Synthesis
editIsoxazole can be synthesised via a variety of methods.[3][4] Examples include via a 1,3-dipolar cycloaddition of nitrile oxides with alkynes; or the reaction of hydroxylamine with 1,3-diketones or derivatives of propiolic acid.[5]
Photochemistry
editThe photolysis of isoxazole was first reported in 1966.[6] Due to the weak N-O bond, the isoxazole ring tends to collapse under UV irradiation, rearranging to oxazole through azirine intermediate. Meanwhile, the azirine intermediate can react with nucleophiles, especially carboxylic acids. Given the photoreactions, isoxazole group is developed as a native photo-cross-linker for photoaffinity labeling and chemoproteomic studies.[7] [8]
Pharmaceuticals and herbicides
editIsoxazoles also form the basis for a number of drugs,[9] including the COX-2 inhibitor valdecoxib (Bextra) and a neurotransmitter agonist AMPA. A derivative, furoxan, is a nitric oxide donor. An isoxazolyl group is found in many beta-lactamase-resistant antibiotics, such as cloxacillin, dicloxacillin and flucloxacillin. Leflunomide is an isoxazole-derivative drug. Examples of AAS containing the isoxazole ring include danazol and androisoxazole. A number of pesticides are isoxazoles.[10]
See also
editReferences
edit- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 140. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ Zoltewicz, J. A. & Deady, L. W. Quaternization of heteroaromatic compounds. Quantitative aspects. Adv. Heterocycl. Chem. 22, 71-121 (1978)
- ^ Dasa S, Chanda K (2021). "An overview of metal-free synthetic routes to isoxazoles: the privileged scaffold". RSC Adv (11): 32680–32705. doi:10.1039/D1RA04624A. PMC 9042182.
- ^ Morita, Taiki; Yugandar, Somaraju; Fuse, Shinichiro; Nakamura, Hiroyuki (March 2018). "Recent progresses in the synthesis of functionalized isoxazoles". Tetrahedron Letters. 59 (13): 1159–1171. doi:10.1016/j.tetlet.2018.02.020.
- ^ Hossain M, Khan M, Kim S, Le H (2022). "Synthesis of 3,4,5-trisubstituted isoxazoles in water via a [3 + 2]-cycloaddition of nitrile oxides and 1,3-diketones, β-ketoesters, or β-ketoamides". Beilstein J. Org. Chem. (18): 446–458. doi:10.3762/bjoc.18.47. PMC 9039522. PMID 35529890.
- ^ Edwin F. Ullman (1966). "Photochemical Transposition of Ring Atoms in Five-Membered Heterocycles. The Photorearrangement of 3,5-Diphenylisoxazole". J. Am. Chem. Soc. 88 (8): 1844–1845. doi:10.1021/ja00960a066.
- ^ Cheng, K.; Qi, J.; Ren, X.; Zhang, J.; Li, H.; Xiao, H.; Wang, R.; Liu, Z.; Meng, L.; Ma, N.; Sun, H. (2022). "Developing Isoxazole as a Native Photo-Cross-Linker for Photoaffinity Labeling and Chemoproteomics". Angew. Chem. Int. Ed. 61 (47): e202209947. doi:10.1002/anie.202209947.
- ^ Lougee, M.; Pagar, V.; Kim, H.; Pancoe, S.; Chia, W.; Mach, R.; Garcia, B.; Petersson, E. (2022). "Harnessing the Intrinsic Photochemistry of Isoxazoles for the Development of Chemoproteomic Crosslinking Methods". Chem. Comm. 58 (65): 9116–9119. doi:10.1039/D2CC02263J. PMC 9922157.
- ^ Zhu, Jie; Mo, Jun; Lin, Hong-zhi; Chen, Yao; Sun, Hao-peng (2018). "The recent progress of isoxazole in medicinal chemistry". Bioorganic & Medicinal Chemistry. 26 (12): 3065–3075. doi:10.1016/j.bmc.2018.05.013.
- ^ Clemens Lamberth (2018). "Oxazole and Isoxazole Chemistry in Crop Protection". Journal of Heterocyclic Chemistry. 55 (9): 2035–2045. doi:10.1002/jhet.3252.